In the title compound, [Na6(NO3)2(H2O)13][Ni(C14H18N2O8)]2, the edta-like tetraanionic unit uses two N and four O atoms to chelate to the Ni atom so that the [Ni(C14H18N2O8)]2- portion of the structure has the Ni atom in a cis-N2O4Ni octahedral geometry. The [Na6(NO3)2H2O)13]4+ portion exists as a chain consisting of edge-sharing NaO6 octahedra; there are four independent Na atoms in the asymmetric unit. The vertices are derived from the O atoms of water molecules, the O atom of the nitrate group as well as the O atoms of the [Ni(C14H18N2O8)]2- unit. The layer structure is consolidated by hydrogen bonds into a three-dimensional network. Two of the Na atoms and three of the water O atoms lie on special positions of site symmetry 2.
Cucurbit[6]uril crystallizes with bis(μ-oxo-κ2O:O)bis[aquaoxodichloromolybdenum(VI)] as the 10.5-hydrate, with the formula [Mo2O4Cl4(H2O)2]·(C36H36N24O12)·10.5H2O. The molybdenum-bearing component interacts indirectly with two cryptands through the two coordinated water molecules, each outer-sphere-coordination cryptand forming only one hydrogen bond with each water molecule. The asymmetric unit has two cryptands, both of which lie on special positions of 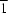 site symmetry. One of them encapsulates a water molecule that is disordered over two general positions, whereas the other encapsulates one that lies on a centre of inversion. The solvent water molecules interact with the dinuclear complex and with both cucurbituril molecules to form a three-dimensional network structure.
site symmetry. One of them encapsulates a water molecule that is disordered over two general positions, whereas the other encapsulates one that lies on a centre of inversion. The solvent water molecules interact with the dinuclear complex and with both cucurbituril molecules to form a three-dimensional network structure.
In the crystal structure of the title compound, [Zn{K(C30H30N20O10)(H2O)}Cl(H2O)][ZnCl4]·5H2O, the [(H2O)3ClZn]+ cation links adjacent [(C30H30N20O10)(H2O)K]+ cations into a polycationic chain. The chain is surrounded by water molecules and disordered [ZnCl4]2− anions and they interact to form a hydrogen-bonded layer structure. One water molecule is encapsulated by the cryptand. The cryptand and the encapsulated water molecule, as well as the K and Zn atoms, one coordinated water molecule and one free water molecule, lie on special positions of site symmetry m.
In the crystal structure of the title compound, [Zn2(C7H5O2)3(OH)], the two Zn atoms are bridged by the carboxyl groups of three benzoate anions. Adjacent dinuclear units are linked into a chain through the hydroxy groups to confer tetrahedral geometries on the metal atoms.
In the crystal structure of the title compound, [Zn2(C8H4O4)2(C2H6OS)2]·5C2H6OS, four independent carboxylate units of the 1,4-benzenedicarboxylate groups chelate to two Zn atoms to give rise to —O—C—O—Zn Zn— rings, the four O atoms around each zinc atom forming a square, above which is a dimethyl sulfoxide (DMSO) molecule and below which is the other Zn atom. The bridging mode of the 1,4-benzenedicarboxylate groups, one of which lies on a general position and the other two on inversion centers, leads to a layer structure having DMSO molecules between the layers. The uncoordinated solvent molecules account for about 80% of the volume of the unit cell.
Zn— rings, the four O atoms around each zinc atom forming a square, above which is a dimethyl sulfoxide (DMSO) molecule and below which is the other Zn atom. The bridging mode of the 1,4-benzenedicarboxylate groups, one of which lies on a general position and the other two on inversion centers, leads to a layer structure having DMSO molecules between the layers. The uncoordinated solvent molecules account for about 80% of the volume of the unit cell.
 Zn— rings, the four O atoms around each zinc atom forming a square, above which is a dimethyl sulfoxide (DMSO) molecule and below which is the other Zn atom. The bridging mode of the 1,4-benzenedicarboxylate groups, one of which lies on a general position and the other two on inversion centers, leads to a layer structure having DMSO molecules between the layers. The uncoordinated solvent molecules account for about 80% of the volume of the unit cell.
Zn— rings, the four O atoms around each zinc atom forming a square, above which is a dimethyl sulfoxide (DMSO) molecule and below which is the other Zn atom. The bridging mode of the 1,4-benzenedicarboxylate groups, one of which lies on a general position and the other two on inversion centers, leads to a layer structure having DMSO molecules between the layers. The uncoordinated solvent molecules account for about 80% of the volume of the unit cell.The hydrothermally prepared title compound, {[Cu3Mo2(NO3)2O8(C10H8N2)3]·4H2O}n, is a hybrid organic/inorganic polymer containing a 16-atom ring built up from molybdate anions, nitrate ions and copper cations. This inorganic network is crosslinked by infinite linear 4,4′-bpy-bridged copper chains (4,4′-bpy is 4,4'-bipyridine) to form channels along the c axis containing the disordered non-coordinated water molecules.

 journal menu
journal menu















