Single crystals of the LaMg2 cubic Laves phase were synthesized by arc melting. The binary LaMg2 compound has been shown to adopt the MgCu2-type structure. The coordination sphere of the rare earth metal, adopting a site symmetry of 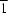 , consists only of 12 Mg atoms. The site symmetry of the alkaline earth metal is
, consists only of 12 Mg atoms. The site symmetry of the alkaline earth metal is 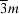 , giving rise to superimposed distorted MgLa6 and MgMg6 octahedra.
, giving rise to superimposed distorted MgLa6 and MgMg6 octahedra.
(NH4)2[Co(H2O)6]3(HPO3)4 is a member of the isotypic (NH4)2[T(H2O)6]3(HPO3)4 family, where T = Co, Ni or Mg. The structure of (NH4)2[Co(H2O)6]3(HPO3)4 is composed of the units [Co(H2O)6]2+, (HPO3)2- and NH4+, which interact via an intricate network of hydrogen bonds. One of the Co atoms is located at a site of 2/m symmetry, whereas the other central atoms, viz. the second Co, the two P and ammonium N atom, are located at sites of m symmetry.
(NH4)2[Ni(H2O)6]3(HPO3)4 is a member of the isotypic (NH4)2[T(H2O)6]3(HPO3)4 family, where T = Co, Ni or Mg. The structure of (NH4)2[Ni(H2O)6]3(HPO3)4 is composed of the units [Ni(H2O)6]2+, (HPO3)2- and NH4+, which interact via an intricate network of hydrogen bonds. One of the Ni atoms is located at a site of 2/m symmetry, whereas the other central atoms, viz. the second Ni, the two P and ammonium N atom, are located at sites of m symmetry.

 journal menu
journal menu












