The crystal structures of the monoclinic and triclinic polymorphs of zoledronic acid, C5H10N2O7P2, have been established from laboratory powder X-ray diffraction data. The molecules in both polymorphs are described as zwitterions, namely 1-(2-hydroxy-2-phosphonato-2-phosphonoethyl)-1H-imidazol-3-ium. Strong intermolecular hydrogen bonds (with donor–acceptor distances of 2.60 Å or less) link the molecules into layers, parallel to the (100) plane in the monoclinic polymorph and to the (1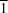 0) plane in the triclinic polymorph. The phosphonic acid groups form the inner side of each layer, while the imidazolium groups lie to the outside of the layer, protruding in opposite directions. In both polymorphs, layers related by translation along [100] interact through weak hydrogen bonds (with donor–acceptor distances greater than 2.70 Å), forming three-dimensional layered structures. In the monoclinic polymorph, there are hydrogen-bonded centrosymmetric dimers linked by four strong O—H
0) plane in the triclinic polymorph. The phosphonic acid groups form the inner side of each layer, while the imidazolium groups lie to the outside of the layer, protruding in opposite directions. In both polymorphs, layers related by translation along [100] interact through weak hydrogen bonds (with donor–acceptor distances greater than 2.70 Å), forming three-dimensional layered structures. In the monoclinic polymorph, there are hydrogen-bonded centrosymmetric dimers linked by four strong O—H O hydrogen bonds, which are not present in the triclinic polymorph.
O hydrogen bonds, which are not present in the triclinic polymorph.
 O hydrogen bonds, which are not present in the triclinic polymorph.
O hydrogen bonds, which are not present in the triclinic polymorph.Afobazole {systematic name: 2-[2-(morpholin-4-yl)ethylsulfanyl]-1H-benzimidazole} is a new anxiolytic drug and Actins, Auzins & Petkune [(2012). Eur. Patent EP10163962] described four polymorphic modifications. In the present study, the crystal structures of two monoclinic polymorphs, 5-ethoxy-2-[2-(morpholin-4-ium-4-yl)ethylsulfanyl]-1H-benzimidazol-3-ium dichloride, C15H23N3O2S2+·2Cl-, (II) and (IV), have been established from laboratory powder diffraction data. The crystal packing and conformation of the dications in (II) and (IV) are different. In (II), there are channels in the [001] direction, which offer atmospheric water molecules an easy way of penetrating into the crystal structure, thus explaining the higher hygroscopicity of (II) compared with (IV).

 journal menu
journal menu










