The asymmetric unit of the title compound, [Ru(OH)(NH3)4(NO)][Pd(NO2)4]·H2O, consists of one complex [Ru(NO)(NH3)4OH]2+ cation, two half [Pd(NO2)4]2- complex anions, each anion located on an inversion centre, and a water molecule of crystallization. The crystal structure exhibits an extensive hydrogen-bonding network.
The structure of monoclinic (C2/c) cadmium bis[dihydrogenphosphate(I)], [Cd(H2PO2)2], consists of layers located at x = 0 and 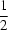 . The Cd cation has a distorted octahedral coordination and dihydrogenphosphate(I) anions act as tridentate bridging ligands. The Cd atoms are on sites of twofold symmetry, while all other atoms are in general positions. The compound is isotypic with Ca(H2PO2)2 [Goedkoop & Loopstra (1959). Ned. Tijdschr. Natuurkd. 25, 29-41].
. The Cd cation has a distorted octahedral coordination and dihydrogenphosphate(I) anions act as tridentate bridging ligands. The Cd atoms are on sites of twofold symmetry, while all other atoms are in general positions. The compound is isotypic with Ca(H2PO2)2 [Goedkoop & Loopstra (1959). Ned. Tijdschr. Natuurkd. 25, 29-41].
The title compound, (C8H20N)[Nb(C6H4S2)2(C6H4S3)], is the major product of the reaction of (Et4N)4[Nb2S4(NCS)8] and disodium benzene-1,2-dithiolate. The coordination environment of the Nb atom contains seven S atoms, viz. four S atoms from two benzene-1,2-dithiolate ligands and three S atoms from 2-disulfanylbenzenethiolate. The Nb-S distances are in the range 2.4391 (6)-2.5932 (7) Å.

 journal menu
journal menu












