In the title compound, [Cu(NCS)(C20H14N4)]SCN, the Cu atom is four-coordinated by a tridentate chelating 4′-(2-pyridyl)-2,2′:6′,2′′-terpyridine ligand and one isothiocyanate group in a square-planar coordination geometry. The sum of angles around the CuII center is 359.1°.
The title compound, [Cu(C8H4O4)(C21H15N3)]·C8H6O4, consists of polymeric chains in which the terpyridine heterocycle chelates to the Cu atom in an N,N′,N′′-tridentate manner; the Cu atoms are also covalently bonded through the two singly-bonded carboxyl O atoms of the dicarboxylate dianions to give rise to a chain structure. An additional terephthalic acid molecule is hydrogen bonded to the chains. The asymmetric unit contains halves of two different half-molecule terephthalate ions; in each case, the complete ions are generated by inversion symmetry.
The title compound, {[Zn(C8H4O4)(C21H15N3)]·H2O}n, is a one-dimensional polymeric complex bridged by centrosymmetric dicarboxylate dianions. Each Zn atom is five-coordinate, bonded to two carboxyl O atoms from the dicarboxylate dianions and to three N atoms from the heterocycle, which chelates to the Zn atom in a κ3N,N′,N′′ manner.
The Ag atoms and the ligands of the dication in the title compound, [Ag2(C20H14N4)2](PF6)2, are disordered; the dication lies on a center of symmetry. The major component is chelated by two N atoms of the heterocycle and is coordinated by the N atom of the pyridyl substituent of the other heterocycle; the geometry is trigonal planar distorted towards tetrahedral owing to a weak Ag F interaction. The minor component is chelated by three N atoms and is also coordinated by the N atom of the pyridyl substituent of the inversion-related heterocycle; its geometry is octahedral owing to two Ag
F interaction. The minor component is chelated by three N atoms and is also coordinated by the N atom of the pyridyl substituent of the inversion-related heterocycle; its geometry is octahedral owing to two Ag F interactions. One anion lies on a special position of site symmetry 2 and the other on a special position of site symmetry
F interactions. One anion lies on a special position of site symmetry 2 and the other on a special position of site symmetry 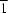 . The disorder in the centrosymmetric dication is best described in terms of the Ag atom being disordered over two sites and the C5H2N ring of the heterocycle being rotated by about 180°; the two dications are superimposed in the crystal structure.
. The disorder in the centrosymmetric dication is best described in terms of the Ag atom being disordered over two sites and the C5H2N ring of the heterocycle being rotated by about 180°; the two dications are superimposed in the crystal structure.
 F interaction. The minor component is chelated by three N atoms and is also coordinated by the N atom of the pyridyl substituent of the inversion-related heterocycle; its geometry is octahedral owing to two Ag
F interaction. The minor component is chelated by three N atoms and is also coordinated by the N atom of the pyridyl substituent of the inversion-related heterocycle; its geometry is octahedral owing to two Ag F interactions. One anion lies on a special position of site symmetry 2 and the other on a special position of site symmetry
F interactions. One anion lies on a special position of site symmetry 2 and the other on a special position of site symmetry In the title complex, [PdCl2(C12H9NS)2], the Pd atom lies on a center of inversion and is coordinated by two chloride anions and two S atoms from two phenothiazine ligands, forming a square-planar PdCl2S2 geometry. In the crystal structure, a one-dimensional polymer structure is constructed via N—H Cl and N—H
Cl and N—H N hydrogen bonds.
N hydrogen bonds.
 Cl and N—H
Cl and N—H N hydrogen bonds.
N hydrogen bonds. In the title complex, [Cu2I2(C12H8N2)2]·C2H3N, two CuI atoms are bridged by two I atoms and each CuI atom is chelated by a 1,10-phenanthroline ligand to give four-coordinate tetrahedral geometry.
The Cu atom in the title compound, [Cu(NCS)2(H2O)2], exists in a trans-N2O2Cu square-planar geometry and the molecule lies on a crystallographic mirror plane. A weak intermolecular Cu S interaction of 3.0185 (2) Å is also observed.
S interaction of 3.0185 (2) Å is also observed.
 S interaction of 3.0185 (2) Å is also observed.
S interaction of 3.0185 (2) Å is also observed.The title compound, [Ni2(C3H2O4)2(C4H4N2)(H2O)2]n, has been prepared from the hydrothermal reaction of nickel(II) chloride, malonic acid and pyrazine. The NiII atoms are linked via coordinated malonates, forming a two-dimensional network with cavities of 6.14 × 5.60 Å. These sheet structures are further connected into a three-dimensional network by bridging pyrazine ligands which have inversion symmetry, the mean distance between the layers being 7.39 Å. The coordination geometry around the NiII atom is a tetragonally elongated octahedron, with pyrazine N and aqua O atoms at the apical positions.

 journal menu
journal menu

















