
The α-polymorph of
ortho-ethoxy-
trans-cinnamic acid (OETCA) undergoes a reversible single-crystal-to-single-crystal phase transformation at 333 K. The new high-temperature polymorph (α′-OETCA) is stable between 333 and 393 K with three molecules in the asymmetric unit (
Z′ = 3), space group
P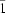
. Unlike the other polymorphs (and solvate) of OETCA recently reported, two of the molecules in α′-OETCA deviate significantly from planarity. This conformational change results in the corrugated sheet-type structure of α′-OETCA. The sheets are made up of ribbons, each composed of
hydrogen-bonded pairs (
via the —COOH groups), which are further connected by CH

O interactions. When exposed to UV radiation the α′-OETCA polymorph can be stabilized below 333 K with
ca 8% of the monomer converted into the photodimer. The crystal structures of α′-OETCA are reported at two temperatures above the phase transition point (at 345 and 375 K) as well as the stabilized forms at 173 and 293 K. A mechanism for the phase transition involving a cooperative conformational transformation coupled with a shift of layers of OETCA molecules is proposed. The α′-OETCA polymorph is also an example of a cinnamic acid derivative where two different potentially photoreactive environments exist in one crystal in which each unit cell has two non-centrosymmetric predimer sites and one centrosymmetric predimer site.
Supporting information
CCDC references: 241455; 241456; 241457; 241458
For all compounds, data collection: SMART-NT (Bruker, 1998); cell refinement: SAINT+(Bruker, 1999a); data reduction: SAINT+(Bruker, 1999a); program(s) used to solve structure: SHELXTL (Bruker, 1999b); program(s) used to refine structure: SHELXTL (Bruker, 1999b); molecular graphics: PLATON (Spek, 2003), SHELXTL (Bruker, 1999b), SCHAKAL97 (Keller, 1997) and DS ViewerPro (Accelrys, 2002); software used to prepare material for publication: SHELXTL (Bruker, 1999b).
(almer60) (
E)-3-(2-Ethoxyphenyl)propenoic acid
top
Crystal data top
| C11H12O3 | Z = 6 |
| Mr = 192.21 | F(000) = 612 |
| Triclinic, P1 | Dx = 1.231 Mg m−3 |
| Hall symbol: -P 1 | Melting point: 134 - 135 °C K |
| a = 8.7336 (4) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 10.9590 (7) Å | Cell parameters from 665 reflections |
| c = 17.2709 (10) Å | θ = 2.7–21.6° |
| α = 91.232 (4)° | µ = 0.09 mm−1 |
| β = 92.703 (4)° | T = 345 K |
| γ = 109.472 (4)° | Prismic, colourless |
| V = 1555.53 (15) Å3 | 1.00 × 0.34 × 0.31 mm |
Data collection top
Bruker SMART 1K
diffractometer | 2689 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.000 |
| Graphite monochromator | θmax = 25.0°, θmin = 1.2° |
| phi and ω scans | h = −10→10 |
| 4937 measured reflections | k = −12→12 |
| 4937 independent reflections | l = 0→20 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.049 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.152 | w = 1/[σ2(Fo2) + (0.0807P)2 + 0.0096P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.02 | (Δ/σ)max < 0.001 |
| 4937 reflections | Δρmax = 0.16 e Å−3 |
| 386 parameters | Δρmin = −0.12 e Å−3 |
| 0 restraints | Extinction correction: SHELXL, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.0085 (17) |
Crystal data top
| C11H12O3 | γ = 109.472 (4)° |
| Mr = 192.21 | V = 1555.53 (15) Å3 |
| Triclinic, P1 | Z = 6 |
| a = 8.7336 (4) Å | Mo Kα radiation |
| b = 10.9590 (7) Å | µ = 0.09 mm−1 |
| c = 17.2709 (10) Å | T = 345 K |
| α = 91.232 (4)° | 1.00 × 0.34 × 0.31 mm |
| β = 92.703 (4)° | |
Data collection top
Bruker SMART 1K
diffractometer | 2689 reflections with I > 2σ(I) |
| 4937 measured reflections | Rint = 0.000 |
| 4937 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.049 | 0 restraints |
| wR(F2) = 0.152 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.02 | Δρmax = 0.16 e Å−3 |
| 4937 reflections | Δρmin = −0.12 e Å−3 |
| 386 parameters | |
Special details top
Experimental. The following alerts are triggered when using the ACTA-C CIF validation software. PLAT029_ALERT_3_A _diffrn_measured_fraction_theta_full Low ······. 0.90 PLAT063_ALERT_3_B Crystal Probably too Large for Beam Size ······. 1.00 mm REFLT03_ALERT_3_C Reflection count < 95% complete From the CIF: _diffrn_reflns_theta_max 25.00 From the CIF: _diffrn_reflns_theta_full 0.00 From the CIF: _reflns_number_total 4937 TEST2: Reflns within _diffrn_reflns_theta_max Count of symmetry unique reflns 5485 Completeness (_total/calc) 90.01% PLAT022_ALERT_3_C Ratio Unique / Expected Reflections too Low ···. 0.90 —————- Author response: These issues are addressed in the paper associated with this structure: "Two crystals with different orientations were used (size: 1.0 x 0.34 x 0.33 and 1.0 x 0.34 x 0.28 mm respectively; collimator size: 0.8 mm) and a data set at each temperature was collected for each crystal. Large crystals were used to counteract the affect of sublimation at 375 K by allowing quick data collection times (about 1.5 h); smaller crystals were found to have almost disappeared by the time the data collection run was over; alternative methods of preventing sublimation such as coating the crystals with epoxy glue (Hashizume et al., 2003) or sealing in capillaries (Botoshansky et al., 1998) were avoided as it was thought that they might interfere with the phase transition. Two crystals in two different orientations were necessary as the heating device physically restricted the amount of data collected on each crystal to about 60% completeness to 25 ° Theta, for a triclinic unit cell, while collecting two data sets on two crystals mounted in different orientations ensured a data completeness of about 90% to 25 ° theta." See article for further details PLAT305_ALERT_2_A Isolated Hydrogen Atom (Outside Bond Range ??) H1C PLAT355_ALERT_3_A Long O—H Bond (0.82 A) O11B - H1B ··· 1.24 A ng. PLAT355_ALERT_3_A Long O—H Bond (0.82 A) O12A - H1A ··· 1.17 A ng. PLAT417_ALERT_2_B Short Inter D—H.·H—D H1A.. H1A.. 1.95 A ng. PLAT420_ALERT_2_B D—H Without Acceptor O11B - H1B ··· ? PLAT430_ALERT_2_B Short Inter D···A Contact O11C.. O12B.. 2.64 A ng. PLAT041_ALERT_1_C Calc. and Rep. SumFormula Strings Differ ···. ? PLAT042_ALERT_1_C Calc. and Rep. MoietyFormula Strings Differ ···. ? PLAT045_ALERT_1_C Calculated and Reported Z Differ by ············ 0.33 Ratio ——— Author response: From article associated with this structure: >From article "··· the hydrogen bond data was obtained by locating the carboxylic hydrogen atom positions in the difference map and then fixing them to their parent atom. In contrast to the usual O—H bond length of about 0.82 Å (the default for SHELX at 345 K), O—H lengths in the a' polymorph at 345/375 K are on average about 1.21/1.28 Å long or about 46/48% of the O···O distance (average 2.63/2.64 Å) respectively i.e. they are almost in the middle of the hydrogen bond being shared almost equally between two carboxylic oxygen atoms." We chose to use located positions for the carboxylic H atoms, rather than fix them using AFIX in SHELX, as this may more accurately describe the hydrogen bonding situation. The located hydrogen atoms being in the middle of the carboxylic acid bond does cause some problems with platons builtin CIF check criteria, which is not able to correctly connect the molecules leading to other errors related to an incorrect calculation of the number of molecules in the unit cell. |
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| C1A | 1.0179 (3) | 0.0351 (2) | 0.22241 (13) | 0.0789 (6) | |
| C2A | 1.0129 (3) | 0.0300 (2) | 0.30283 (13) | 0.0813 (6) | |
| C3A | 1.1498 (3) | 0.0399 (3) | 0.34824 (15) | 0.1015 (8) | |
| H3A | 1.1450 | 0.0370 | 0.4019 | 0.122* | |
| C4A | 1.2939 (3) | 0.0541 (3) | 0.31491 (19) | 0.1150 (9) | |
| H4A | 1.3863 | 0.0606 | 0.3461 | 0.138* | |
| C5A | 1.3024 (3) | 0.0586 (3) | 0.2361 (2) | 0.1148 (9) | |
| H5A | 1.4000 | 0.0675 | 0.2136 | 0.138* | |
| C6A | 1.1666 (3) | 0.0502 (2) | 0.19071 (16) | 0.1007 (8) | |
| H6A | 1.1737 | 0.0546 | 0.1372 | 0.121* | |
| C11A | 0.8700 (3) | 0.0252 (2) | 0.17502 (13) | 0.0836 (7) | |
| H11A | 0.7784 | 0.0199 | 0.2019 | 0.100* | |
| C12A | 0.8519 (3) | 0.0230 (2) | 0.09937 (14) | 0.0918 (7) | |
| H12A | 0.9419 | 0.0294 | 0.0709 | 0.110* | |
| C13A | 0.6983 (3) | 0.0109 (2) | 0.05687 (15) | 0.0866 (7) | |
| C21A | 0.8489 (3) | 0.0112 (3) | 0.41270 (13) | 0.1027 (8) | |
| H21E | 0.9250 | 0.0890 | 0.4381 | 0.123* | |
| H21F | 0.8722 | −0.0631 | 0.4328 | 0.123* | |
| C22A | 0.6795 (3) | 0.0018 (3) | 0.42765 (15) | 0.1280 (10) | |
| H22G | 0.6576 | 0.0757 | 0.4075 | 0.192* | |
| H22H | 0.6660 | −0.0005 | 0.4825 | 0.192* | |
| H22I | 0.6052 | −0.0758 | 0.4026 | 0.192* | |
| O11A | 0.6952 (2) | 0.01020 (17) | −0.01537 (9) | 0.1090 (6) | |
| O12A | 0.5726 (2) | 0.00053 (17) | 0.09525 (8) | 0.1068 (6) | |
| H1A | 0.4692 | 0.0100 | 0.0526 | 0.242 (19)* | |
| O21A | 0.86409 (18) | 0.01490 (16) | 0.33134 (8) | 0.0961 (5) | |
| C1B | 0.8074 (2) | 0.3527 (2) | 0.13506 (12) | 0.0718 (6) | |
| C2B | 0.8054 (2) | 0.3390 (2) | 0.05442 (12) | 0.0726 (6) | |
| C3B | 0.6699 (3) | 0.3333 (2) | 0.00898 (14) | 0.0944 (7) | |
| H3B | 0.6701 | 0.3246 | −0.0447 | 0.113* | |
| C4B | 0.5329 (3) | 0.3405 (3) | 0.04312 (17) | 0.1079 (9) | |
| H4B | 0.4408 | 0.3357 | 0.0122 | 0.129* | |
| C5B | 0.5317 (3) | 0.3547 (3) | 0.12155 (18) | 0.1101 (9) | |
| H5B | 0.4397 | 0.3604 | 0.1442 | 0.132* | |
| C6B | 0.6668 (3) | 0.3604 (2) | 0.16679 (15) | 0.0947 (7) | |
| H6B | 0.6650 | 0.3697 | 0.2203 | 0.114* | |
| C11B | 0.9519 (3) | 0.35932 (19) | 0.18293 (12) | 0.0738 (6) | |
| H11B | 1.0441 | 0.3651 | 0.1564 | 0.089* | |
| C12B | 0.9684 (3) | 0.3582 (2) | 0.25900 (12) | 0.0845 (7) | |
| H12B | 0.8783 | 0.3531 | 0.2870 | 0.101* | |
| C13B | 1.1174 (3) | 0.3643 (2) | 0.30209 (12) | 0.0803 (6) | |
| C21B | 0.9511 (2) | 0.3152 (2) | −0.05662 (11) | 0.0817 (6) | |
| H21A | 0.9373 | 0.3885 | −0.0828 | 0.098* | |
| H21B | 0.8650 | 0.2372 | −0.0758 | 0.098* | |
| C22B | 1.1141 (3) | 0.3047 (2) | −0.07109 (13) | 0.0971 (8) | |
| H22D | 1.1979 | 0.3837 | −0.0538 | 0.146* | |
| H22E | 1.1208 | 0.2896 | −0.1256 | 0.146* | |
| H22F | 1.1279 | 0.2339 | −0.0432 | 0.146* | |
| O11B | 1.1114 (2) | 0.34873 (16) | 0.37454 (8) | 0.0982 (5) | |
| H1B | 1.2335 | 0.3478 | 0.4136 | 0.251 (18)* | |
| O12B | 1.24646 (19) | 0.38378 (16) | 0.26678 (8) | 0.0981 (5) | |
| O21B | 0.94565 (16) | 0.33197 (15) | 0.02525 (7) | 0.0825 (5) | |
| C1C | 0.2044 (2) | 0.6761 (2) | 0.41880 (12) | 0.0727 (6) | |
| C2C | 0.2171 (2) | 0.6971 (2) | 0.33960 (12) | 0.0746 (6) | |
| C3C | 0.0873 (3) | 0.7034 (2) | 0.29414 (14) | 0.0952 (7) | |
| H3C | 0.0977 | 0.7173 | 0.2414 | 0.114* | |
| C4C | −0.0581 (3) | 0.6893 (3) | 0.32632 (18) | 0.1069 (8) | |
| H4C | −0.1459 | 0.6934 | 0.2951 | 0.128* | |
| C5C | −0.0750 (3) | 0.6694 (2) | 0.40403 (18) | 0.1060 (9) | |
| H5C | −0.1734 | 0.6606 | 0.4257 | 0.127* | |
| C6C | 0.0540 (3) | 0.6626 (2) | 0.44922 (14) | 0.0922 (7) | |
| H6C | 0.0418 | 0.6485 | 0.5018 | 0.111* | |
| C11C | 0.3420 (3) | 0.6665 (2) | 0.46571 (12) | 0.0773 (6) | |
| H11C | 0.4367 | 0.6781 | 0.4400 | 0.093* | |
| C12C | 0.3496 (3) | 0.6434 (2) | 0.54027 (13) | 0.0857 (7) | |
| H12C | 0.2573 | 0.6318 | 0.5679 | 0.103* | |
| C13C | 0.4940 (3) | 0.6350 (2) | 0.58137 (13) | 0.0854 (7) | |
| C21C | 0.3828 (3) | 0.7136 (3) | 0.23053 (12) | 0.0958 (8) | |
| H21C | 0.3737 | 0.7938 | 0.2116 | 0.115* | |
| H21D | 0.2983 | 0.6417 | 0.2040 | 0.115* | |
| C22C | 0.5453 (3) | 0.7060 (3) | 0.21584 (13) | 0.1112 (9) | |
| H22A | 0.6278 | 0.7765 | 0.2434 | 0.167* | |
| H22B | 0.5615 | 0.7114 | 0.1613 | 0.167* | |
| H22C | 0.5518 | 0.6253 | 0.2334 | 0.167* | |
| O11C | 0.4853 (2) | 0.60726 (18) | 0.65238 (9) | 0.1148 (6) | |
| H1C | 0.6111 | 0.6123 | 0.6892 | 0.217 (14)* | |
| O12C | 0.62491 (19) | 0.65652 (16) | 0.54689 (8) | 0.1010 (5) | |
| O21C | 0.36550 (16) | 0.70875 (16) | 0.31198 (8) | 0.0909 (5) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C1A | 0.0921 (16) | 0.0695 (17) | 0.0742 (16) | 0.0259 (13) | 0.0064 (13) | 0.0022 (12) |
| C2A | 0.0849 (16) | 0.0832 (18) | 0.0775 (16) | 0.0308 (13) | 0.0027 (13) | 0.0020 (12) |
| C3A | 0.1018 (18) | 0.121 (2) | 0.0874 (17) | 0.0475 (16) | −0.0076 (15) | −0.0018 (15) |
| C4A | 0.0935 (19) | 0.127 (3) | 0.131 (3) | 0.0472 (17) | −0.0027 (17) | −0.0030 (19) |
| C5A | 0.096 (2) | 0.118 (2) | 0.134 (3) | 0.0394 (17) | 0.0254 (19) | 0.0007 (19) |
| C6A | 0.111 (2) | 0.098 (2) | 0.0949 (19) | 0.0355 (16) | 0.0203 (17) | 0.0012 (14) |
| C11A | 0.1156 (19) | 0.0688 (17) | 0.0641 (15) | 0.0264 (14) | 0.0138 (13) | 0.0033 (12) |
| C12A | 0.118 (2) | 0.0861 (19) | 0.0708 (16) | 0.0311 (14) | 0.0204 (14) | 0.0061 (13) |
| C13A | 0.1121 (19) | 0.0744 (18) | 0.0676 (16) | 0.0236 (14) | 0.0058 (15) | 0.0047 (12) |
| C21A | 0.1171 (19) | 0.133 (2) | 0.0630 (15) | 0.0487 (17) | 0.0043 (13) | 0.0041 (14) |
| C22A | 0.110 (2) | 0.180 (3) | 0.097 (2) | 0.052 (2) | 0.0186 (16) | −0.0015 (19) |
| O11A | 0.1298 (14) | 0.1283 (16) | 0.0600 (10) | 0.0308 (11) | 0.0107 (9) | 0.0005 (9) |
| O12A | 0.1314 (14) | 0.1297 (16) | 0.0580 (10) | 0.0404 (11) | 0.0129 (10) | 0.0105 (9) |
| O21A | 0.0907 (11) | 0.1340 (15) | 0.0635 (10) | 0.0377 (10) | 0.0006 (8) | 0.0044 (9) |
| C1B | 0.0784 (14) | 0.0696 (16) | 0.0700 (14) | 0.0273 (11) | 0.0074 (11) | 0.0065 (11) |
| C2B | 0.0721 (13) | 0.0790 (17) | 0.0686 (14) | 0.0278 (12) | 0.0027 (11) | 0.0071 (11) |
| C3B | 0.0820 (15) | 0.117 (2) | 0.0852 (17) | 0.0354 (15) | −0.0035 (13) | 0.0047 (14) |
| C4B | 0.0764 (16) | 0.131 (3) | 0.118 (2) | 0.0377 (15) | −0.0009 (15) | 0.0089 (18) |
| C5B | 0.0831 (18) | 0.135 (3) | 0.121 (2) | 0.0448 (16) | 0.0245 (17) | 0.0127 (18) |
| C6B | 0.0952 (17) | 0.110 (2) | 0.0860 (17) | 0.0412 (15) | 0.0218 (14) | 0.0110 (13) |
| C11B | 0.0903 (15) | 0.0715 (16) | 0.0603 (13) | 0.0280 (12) | 0.0053 (11) | 0.0020 (11) |
| C12B | 0.1016 (17) | 0.0931 (19) | 0.0617 (15) | 0.0359 (14) | 0.0113 (13) | 0.0012 (12) |
| C13B | 0.1071 (18) | 0.0784 (18) | 0.0566 (15) | 0.0328 (14) | 0.0034 (14) | 0.0012 (11) |
| C21B | 0.0888 (15) | 0.0910 (19) | 0.0603 (14) | 0.0236 (13) | 0.0019 (11) | 0.0028 (11) |
| C22B | 0.0970 (16) | 0.115 (2) | 0.0885 (17) | 0.0465 (15) | 0.0159 (13) | 0.0014 (14) |
| O11B | 0.1233 (13) | 0.1266 (15) | 0.0514 (9) | 0.0503 (11) | 0.0069 (8) | 0.0034 (8) |
| O12B | 0.1034 (12) | 0.1256 (15) | 0.0627 (10) | 0.0333 (10) | 0.0106 (9) | 0.0158 (8) |
| O21B | 0.0809 (9) | 0.1130 (13) | 0.0597 (9) | 0.0413 (8) | 0.0003 (7) | −0.0014 (8) |
| C1C | 0.0783 (14) | 0.0723 (16) | 0.0676 (14) | 0.0254 (12) | 0.0050 (11) | 0.0010 (11) |
| C2C | 0.0722 (14) | 0.0806 (17) | 0.0706 (15) | 0.0254 (12) | 0.0037 (11) | −0.0003 (12) |
| C3C | 0.0892 (16) | 0.119 (2) | 0.0835 (16) | 0.0436 (15) | −0.0038 (14) | 0.0060 (14) |
| C4C | 0.0826 (17) | 0.122 (2) | 0.120 (2) | 0.0399 (15) | −0.0032 (15) | 0.0028 (18) |
| C5C | 0.0841 (17) | 0.110 (2) | 0.131 (2) | 0.0398 (15) | 0.0251 (17) | 0.0108 (18) |
| C6C | 0.0953 (17) | 0.094 (2) | 0.0918 (17) | 0.0351 (14) | 0.0253 (15) | 0.0093 (14) |
| C11C | 0.0937 (15) | 0.0762 (17) | 0.0597 (14) | 0.0242 (12) | 0.0143 (12) | 0.0013 (11) |
| C12C | 0.0906 (16) | 0.099 (2) | 0.0670 (15) | 0.0301 (14) | 0.0104 (12) | 0.0043 (13) |
| C13C | 0.1011 (18) | 0.0911 (19) | 0.0574 (14) | 0.0232 (14) | 0.0057 (13) | 0.0026 (12) |
| C21C | 0.1013 (17) | 0.130 (2) | 0.0623 (15) | 0.0452 (16) | 0.0101 (12) | 0.0192 (13) |
| C22C | 0.0983 (17) | 0.150 (3) | 0.0949 (18) | 0.0500 (17) | 0.0255 (14) | 0.0228 (16) |
| O11C | 0.1112 (13) | 0.1663 (18) | 0.0591 (10) | 0.0343 (11) | 0.0077 (9) | 0.0258 (10) |
| O12C | 0.1043 (12) | 0.1383 (16) | 0.0638 (10) | 0.0442 (11) | 0.0106 (9) | 0.0095 (9) |
| O21C | 0.0812 (10) | 0.1346 (14) | 0.0594 (9) | 0.0389 (9) | 0.0046 (7) | 0.0080 (8) |
Geometric parameters (Å, º) top
| C1A—C2A | 1.393 (3) | C11B—H11B | 0.9300 |
| C1A—C6A | 1.394 (3) | C12B—C13B | 1.449 (3) |
| C1A—C11A | 1.467 (3) | C12B—H12B | 0.9300 |
| C2A—O21A | 1.371 (2) | C13B—O12B | 1.264 (2) |
| C2A—C3A | 1.371 (3) | C13B—O11B | 1.268 (2) |
| C3A—C4A | 1.371 (3) | C21B—O21B | 1.427 (2) |
| C3A—H3A | 0.9300 | C21B—C22B | 1.498 (3) |
| C4A—C5A | 1.367 (3) | C21B—H21A | 0.9700 |
| C4A—H4A | 0.9300 | C21B—H21B | 0.9700 |
| C5A—C6A | 1.366 (3) | C22B—H22D | 0.9600 |
| C5A—H5A | 0.9300 | C22B—H22E | 0.9600 |
| C6A—H6A | 0.9300 | C22B—H22F | 0.9600 |
| C11A—C12A | 1.307 (3) | O11B—H1B | 1.2377 |
| C11A—H11A | 0.9300 | C1C—C2C | 1.395 (3) |
| C12A—C13A | 1.463 (3) | C1C—C6C | 1.400 (3) |
| C12A—H12A | 0.9300 | C1C—C11C | 1.452 (3) |
| C13A—O11A | 1.246 (2) | C2C—O21C | 1.369 (2) |
| C13A—O12A | 1.283 (2) | C2C—C3C | 1.370 (3) |
| C21A—O21A | 1.418 (2) | C3C—C4C | 1.373 (3) |
| C21A—C22A | 1.484 (3) | C3C—H3C | 0.9300 |
| C21A—H21E | 0.9700 | C4C—C5C | 1.371 (3) |
| C21A—H21F | 0.9700 | C4C—H4C | 0.9300 |
| C22A—H22G | 0.9600 | C5C—C6C | 1.364 (3) |
| C22A—H22H | 0.9600 | C5C—H5C | 0.9300 |
| C22A—H22I | 0.9600 | C6C—H6C | 0.9300 |
| O12A—H1A | 1.1705 | C11C—C12C | 1.321 (3) |
| C1B—C6B | 1.394 (3) | C11C—H11C | 0.9300 |
| C1B—C2B | 1.396 (3) | C12C—C13C | 1.448 (3) |
| C1B—C11B | 1.456 (3) | C12C—H12C | 0.9300 |
| C2B—O21B | 1.371 (2) | C13C—O12C | 1.268 (2) |
| C2B—C3B | 1.371 (3) | C13C—O11C | 1.269 (2) |
| C3B—C4B | 1.383 (3) | C21C—O21C | 1.422 (2) |
| C3B—H3B | 0.9300 | C21C—C22C | 1.482 (3) |
| C4B—C5B | 1.361 (3) | C21C—H21C | 0.9700 |
| C4B—H4B | 0.9300 | C21C—H21D | 0.9700 |
| C5B—C6B | 1.367 (3) | C22C—H22A | 0.9600 |
| C5B—H5B | 0.9300 | C22C—H22B | 0.9600 |
| C6B—H6B | 0.9300 | C22C—H22C | 0.9600 |
| C11B—C12B | 1.316 (3) | O11C—H1C | 1.2278 |
| | | |
| C2A—C1A—C6A | 117.3 (2) | C11B—C12B—C13B | 124.5 (2) |
| C2A—C1A—C11A | 119.7 (2) | C11B—C12B—H12B | 117.8 |
| C6A—C1A—C11A | 122.9 (2) | C13B—C12B—H12B | 117.8 |
| O21A—C2A—C3A | 124.0 (2) | O12B—C13B—O11B | 122.7 (2) |
| O21A—C2A—C1A | 115.3 (2) | O12B—C13B—C12B | 119.60 (19) |
| C3A—C2A—C1A | 120.7 (2) | O11B—C13B—C12B | 117.7 (2) |
| C2A—C3A—C4A | 120.3 (2) | O21B—C21B—C22B | 106.92 (16) |
| C2A—C3A—H3A | 119.9 | O21B—C21B—H21A | 110.3 |
| C4A—C3A—H3A | 119.9 | C22B—C21B—H21A | 110.3 |
| C5A—C4A—C3A | 120.3 (3) | O21B—C21B—H21B | 110.3 |
| C5A—C4A—H4A | 119.8 | C22B—C21B—H21B | 110.3 |
| C3A—C4A—H4A | 119.8 | H21A—C21B—H21B | 108.6 |
| C6A—C5A—C4A | 119.6 (2) | C21B—C22B—H22D | 109.5 |
| C6A—C5A—H5A | 120.2 | C21B—C22B—H22E | 109.5 |
| C4A—C5A—H5A | 120.2 | H22D—C22B—H22E | 109.5 |
| C5A—C6A—C1A | 121.7 (2) | C21B—C22B—H22F | 109.5 |
| C5A—C6A—H6A | 119.1 | H22D—C22B—H22F | 109.5 |
| C1A—C6A—H6A | 119.1 | H22E—C22B—H22F | 109.5 |
| C12A—C11A—C1A | 127.6 (2) | C13B—O11B—H1B | 120.3 |
| C12A—C11A—H11A | 116.2 | C2B—O21B—C21B | 118.69 (15) |
| C1A—C11A—H11A | 116.2 | C2C—C1C—C6C | 117.0 (2) |
| C11A—C12A—C13A | 123.8 (2) | C2C—C1C—C11C | 120.26 (18) |
| C11A—C12A—H12A | 118.1 | C6C—C1C—C11C | 122.7 (2) |
| C13A—C12A—H12A | 118.1 | O21C—C2C—C3C | 123.8 (2) |
| O11A—C13A—O12A | 122.8 (2) | O21C—C2C—C1C | 115.22 (19) |
| O11A—C13A—C12A | 118.3 (2) | C3C—C2C—C1C | 121.02 (19) |
| O12A—C13A—C12A | 118.9 (2) | C2C—C3C—C4C | 120.1 (2) |
| O21A—C21A—C22A | 107.8 (2) | C2C—C3C—H3C | 120.0 |
| O21A—C21A—H21E | 110.2 | C4C—C3C—H3C | 120.0 |
| C22A—C21A—H21E | 110.2 | C5C—C4C—C3C | 120.6 (2) |
| O21A—C21A—H21F | 110.2 | C5C—C4C—H4C | 119.7 |
| C22A—C21A—H21F | 110.2 | C3C—C4C—H4C | 119.7 |
| H21E—C21A—H21F | 108.5 | C6C—C5C—C4C | 119.3 (2) |
| C21A—C22A—H22G | 109.5 | C6C—C5C—H5C | 120.3 |
| C21A—C22A—H22H | 109.5 | C4C—C5C—H5C | 120.3 |
| H22G—C22A—H22H | 109.5 | C5C—C6C—C1C | 122.0 (2) |
| C21A—C22A—H22I | 109.5 | C5C—C6C—H6C | 119.0 |
| H22G—C22A—H22I | 109.5 | C1C—C6C—H6C | 119.0 |
| H22H—C22A—H22I | 109.5 | C12C—C11C—C1C | 128.2 (2) |
| C13A—O12A—H1A | 108.7 | C12C—C11C—H11C | 115.9 |
| C2A—O21A—C21A | 118.96 (18) | C1C—C11C—H11C | 115.9 |
| C6B—C1B—C2B | 117.2 (2) | C11C—C12C—C13C | 123.6 (2) |
| C6B—C1B—C11B | 122.2 (2) | C11C—C12C—H12C | 118.2 |
| C2B—C1B—C11B | 120.53 (18) | C13C—C12C—H12C | 118.2 |
| O21B—C2B—C3B | 123.55 (19) | O12C—C13C—O11C | 121.7 (2) |
| O21B—C2B—C1B | 115.65 (18) | O12C—C13C—C12C | 120.2 (2) |
| C3B—C2B—C1B | 120.81 (19) | O11C—C13C—C12C | 118.0 (2) |
| C2B—C3B—C4B | 119.9 (2) | O21C—C21C—C22C | 107.66 (17) |
| C2B—C3B—H3B | 120.1 | O21C—C21C—H21C | 110.2 |
| C4B—C3B—H3B | 120.1 | C22C—C21C—H21C | 110.2 |
| C5B—C4B—C3B | 120.6 (2) | O21C—C21C—H21D | 110.2 |
| C5B—C4B—H4B | 119.7 | C22C—C21C—H21D | 110.2 |
| C3B—C4B—H4B | 119.7 | H21C—C21C—H21D | 108.5 |
| C4B—C5B—C6B | 119.5 (2) | C21C—C22C—H22A | 109.5 |
| C4B—C5B—H5B | 120.3 | C21C—C22C—H22B | 109.5 |
| C6B—C5B—H5B | 120.3 | H22A—C22C—H22B | 109.5 |
| C5B—C6B—C1B | 122.0 (2) | C21C—C22C—H22C | 109.5 |
| C5B—C6B—H6B | 119.0 | H22A—C22C—H22C | 109.5 |
| C1B—C6B—H6B | 119.0 | H22B—C22C—H22C | 109.5 |
| C12B—C11B—C1B | 128.2 (2) | C13C—O11C—H1C | 118.0 |
| C12B—C11B—H11B | 115.9 | C2C—O21C—C21C | 118.80 (16) |
| C1B—C11B—H11B | 115.9 | | |
| | | |
| C6A—C1A—C2A—O21A | −179.91 (18) | C11B—C1B—C6B—C5B | 179.7 (2) |
| C11A—C1A—C2A—O21A | 0.3 (3) | C6B—C1B—C11B—C12B | 8.3 (4) |
| C6A—C1A—C2A—C3A | 0.2 (3) | C2B—C1B—C11B—C12B | −172.1 (2) |
| C11A—C1A—C2A—C3A | −179.6 (2) | C1B—C11B—C12B—C13B | 179.6 (2) |
| O21A—C2A—C3A—C4A | 179.6 (2) | C11B—C12B—C13B—O12B | 6.8 (3) |
| C1A—C2A—C3A—C4A | −0.5 (4) | C11B—C12B—C13B—O11B | −172.8 (2) |
| C2A—C3A—C4A—C5A | 0.1 (4) | C3B—C2B—O21B—C21B | −1.3 (3) |
| C3A—C4A—C5A—C6A | 0.6 (4) | C1B—C2B—O21B—C21B | 178.70 (17) |
| C4A—C5A—C6A—C1A | −0.9 (4) | C22B—C21B—O21B—C2B | −177.41 (17) |
| C2A—C1A—C6A—C5A | 0.5 (4) | C6C—C1C—C2C—O21C | −179.48 (17) |
| C11A—C1A—C6A—C5A | −179.7 (2) | C11C—C1C—C2C—O21C | −0.7 (3) |
| C2A—C1A—C11A—C12A | −177.0 (2) | C6C—C1C—C2C—C3C | −0.3 (3) |
| C6A—C1A—C11A—C12A | 3.2 (4) | C11C—C1C—C2C—C3C | 178.5 (2) |
| C1A—C11A—C12A—C13A | 179.1 (2) | O21C—C2C—C3C—C4C | 179.3 (2) |
| C11A—C12A—C13A—O11A | 179.8 (2) | C1C—C2C—C3C—C4C | 0.2 (4) |
| C11A—C12A—C13A—O12A | −0.7 (4) | C2C—C3C—C4C—C5C | 0.3 (4) |
| C3A—C2A—O21A—C21A | 1.1 (3) | C3C—C4C—C5C—C6C | −0.5 (4) |
| C1A—C2A—O21A—C21A | −178.81 (19) | C4C—C5C—C6C—C1C | 0.4 (4) |
| C22A—C21A—O21A—C2A | 176.8 (2) | C2C—C1C—C6C—C5C | 0.0 (3) |
| C6B—C1B—C2B—O21B | 179.90 (18) | C11C—C1C—C6C—C5C | −178.7 (2) |
| C11B—C1B—C2B—O21B | 0.3 (3) | C2C—C1C—C11C—C12C | −177.9 (2) |
| C6B—C1B—C2B—C3B | −0.1 (3) | C6C—C1C—C11C—C12C | 0.8 (4) |
| C11B—C1B—C2B—C3B | −179.7 (2) | C1C—C11C—C12C—C13C | 179.6 (2) |
| O21B—C2B—C3B—C4B | 179.7 (2) | C11C—C12C—C13C—O12C | 3.6 (4) |
| C1B—C2B—C3B—C4B | −0.3 (3) | C11C—C12C—C13C—O11C | −177.3 (2) |
| C2B—C3B—C4B—C5B | 0.7 (4) | C3C—C2C—O21C—C21C | −7.1 (3) |
| C3B—C4B—C5B—C6B | −0.7 (4) | C1C—C2C—O21C—C21C | 172.03 (18) |
| C4B—C5B—C6B—C1B | 0.3 (4) | C22C—C21C—O21C—C2C | −171.53 (19) |
| C2B—C1B—C6B—C5B | 0.1 (4) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11C—H1C···O12Bi | 1.23 | 1.41 | 2.642 (2) | 178 |
| O11B—H1B···O12Ci | 1.24 | 1.40 | 2.635 (2) | 176 |
| O12A—H1A···O11Aii | 1.17 | 1.49 | 2.626 (3) | 161 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
(almer80) (
E)-3-(2-Ethoxyphenyl)propenoic acid
top
Crystal data top
| C11H12O3 | Z = 6 |
| Mr = 192.21 | F(000) = 612 |
| Triclinic, P1 | Dx = 1.218 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 8.7373 (5) Å | Cell parameters from 797 reflections |
| b = 11.0167 (8) Å | θ = 2.3–20.9° |
| c = 17.295 (1) Å | µ = 0.09 mm−1 |
| α = 91.187 (4)° | T = 375 K |
| β = 92.775 (5)° | Prismic, colourless |
| γ = 108.938 (4)° | 1.00 × 0.34 × 0.31 mm |
| V = 1571.62 (17) Å3 | |
Data collection top
Bruker SMART 1K
diffractometer | 1906 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.000 |
| Graphite monochromator | θmax = 25.0°, θmin = 1.2° |
| phi and ω scans | h = −10→10 |
| 4947 measured reflections | k = −12→12 |
| 4947 independent reflections | l = 0→20 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.049 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.157 | w = 1/[σ2(Fo2) + (0.0788P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 0.88 | (Δ/σ)max < 0.001 |
| 4947 reflections | Δρmax = 0.15 e Å−3 |
| 386 parameters | Δρmin = −0.11 e Å−3 |
| 0 restraints | Extinction correction: SHELXL, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.0062 (13) |
Crystal data top
| C11H12O3 | γ = 108.938 (4)° |
| Mr = 192.21 | V = 1571.62 (17) Å3 |
| Triclinic, P1 | Z = 6 |
| a = 8.7373 (5) Å | Mo Kα radiation |
| b = 11.0167 (8) Å | µ = 0.09 mm−1 |
| c = 17.295 (1) Å | T = 375 K |
| α = 91.187 (4)° | 1.00 × 0.34 × 0.31 mm |
| β = 92.775 (5)° | |
Data collection top
Bruker SMART 1K
diffractometer | 1906 reflections with I > 2σ(I) |
| 4947 measured reflections | Rint = 0.000 |
| 4947 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.049 | 0 restraints |
| wR(F2) = 0.157 | H atoms treated by a mixture of independent and constrained refinement |
| S = 0.88 | Δρmax = 0.15 e Å−3 |
| 4947 reflections | Δρmin = −0.11 e Å−3 |
| 386 parameters | |
Special details top
Experimental. The following alerts are triggered when using the ACTA-C CIF validation software. PLAT029_ALERT_3_A _diffrn_measured_fraction_theta_full Low ······. 0.89 REFLT03_ALERT_3_B Reflection count < 90% complete (theta max?) From the CIF: _diffrn_reflns_theta_max 25.00 From the CIF: _diffrn_reflns_theta_full 0.00 From the CIF: _reflns_number_total 4947 TEST2: Reflns within _diffrn_reflns_theta_max Count of symmetry unique reflns 5534 Completeness (_total/calc) 89.39% PLAT022_ALERT_3_B Ratio Unique / Expected Reflections too Low ···. 0.89 PLAT026_ALERT_3_B Ratio Observed / Unique Reflections too Low ···. 39 Perc. PLAT063_ALERT_3_B Crystal Probably too Large for Beam Size ······. 1.00 mm ———- Author response: These issues are addressed in the paper associated with this structure: "Two crystals with different orientations were used (size: 1.0 x 0.34 x 0.33 and 1.0 x 0.34 x 0.28 mm respectively; collimator size: 0.8 mm) and a data set at each temperature was collected for each crystal. Large crystals were used to counteract the affect of sublimation at 375 K by allowing quick data collection times (about 1.5 h); smaller crystals were found to have almost disappeared by the time the data collection run was over; alternative methods of preventing sublimation such as coating the crystals with epoxy glue (Hashizume et al., 2003) or sealing in capillaries (Botoshansky et al., 1998) were avoided as it was thought that they might interfere with the phase transition. Two crystals in two different orientations were necessary as the heating device physically restricted the amount of data collected on each crystal to about 60% completeness to 25 ° Theta, for a triclinic unit cell, while collecting two data sets on two crystals mounted in different orientations ensured a data completeness of about 90% to 25 ° theta." See article for further details PLAT305_ALERT_2_A Isolated Hydrogen Atom (Outside Bond Range ??) H1C PLAT355_ALERT_3_A Long O—H Bond (0.82 A) O12B - H1B ··· 1.31 A ng. PLAT355_ALERT_3_A Long O—H Bond (0.82 A) O12A - H1A ··· 1.26 A ng. PLAT772_ALERT_2_A Suspect O—H Bond in CIF: O12B –H1B.. 1.31 A ng. PLAT420_ALERT_2_B D—H Without Acceptor O12A - H1A ··· ? PLAT420_ALERT_2_B D—H Without Acceptor O12B - H1B ··· ? PLAT430_ALERT_2_B Short Inter D···A Contact O11B.. O12C.. 2.64 A ng. PLAT041_ALERT_1_C Calc. and Rep. SumFormula Strings Differ ···. ? PLAT042_ALERT_1_C Calc. and Rep. MoietyFormula Strings Differ ···. ? ——— Author response: From article associated with this structure: >From article "··· the hydrogen bond data was obtained by locating the carboxylic hydrogen atom positions in the difference map and then fixing them to their parent atom. In contrast to the usual O—H bond length of about 0.82 Å (the default for SHELX at 345 K), O—H lengths in the a' polymorph at 345/375 K are on average about 1.21/1.28 Å long or about 46/48% of the O···O distance (average 2.63/2.64 Å) respectively i.e. they are almost in the middle of the hydrogen bond being shared almost equally between two carboxylic oxygen atoms." We chose to use located positions for the carboxylic H atoms, rather than fix them using AFIX in SHELX, as this may more accurately describe the hydrogen bonding situation. The located hydrogen atoms being in the middle of the carboxylic acid bond does cause some problems with platons builtin CIF check criteria, which is not able to correctly connect the molecules leading to other errors related to an incorrect calculation of the number of molecules in the unit cell. |
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| C1A | 1.0173 (4) | 0.0345 (3) | 0.22231 (19) | 0.0918 (8) | |
| C2A | 1.0123 (4) | 0.0293 (3) | 0.30236 (19) | 0.0950 (9) | |
| C3A | 1.1485 (5) | 0.0389 (3) | 0.34807 (19) | 0.1192 (11) | |
| H3A | 1.1434 | 0.0352 | 0.4016 | 0.143* | |
| C4A | 1.2923 (5) | 0.0541 (3) | 0.3147 (3) | 0.1342 (13) | |
| H4A | 1.3845 | 0.0606 | 0.3459 | 0.161* | |
| C5A | 1.3023 (5) | 0.0598 (3) | 0.2362 (3) | 0.1317 (13) | |
| H5A | 1.4004 | 0.0705 | 0.2140 | 0.158* | |
| C6A | 1.1658 (5) | 0.0497 (3) | 0.1908 (2) | 0.1167 (11) | |
| H6A | 1.1724 | 0.0530 | 0.1373 | 0.140* | |
| C11A | 0.8693 (4) | 0.0251 (3) | 0.17491 (18) | 0.0988 (10) | |
| H11A | 0.7778 | 0.0199 | 0.2017 | 0.119* | |
| C12A | 0.8523 (5) | 0.0233 (3) | 0.1006 (2) | 0.1088 (10) | |
| H12A | 0.9425 | 0.0304 | 0.0723 | 0.131* | |
| C13A | 0.6961 (5) | 0.0104 (3) | 0.0575 (2) | 0.1000 (10) | |
| C21A | 0.8482 (4) | 0.0113 (3) | 0.41231 (17) | 0.1272 (11) | |
| H21E | 0.9225 | 0.0890 | 0.4375 | 0.153* | |
| H21F | 0.8736 | −0.0617 | 0.4328 | 0.153* | |
| C22A | 0.6799 (4) | 0.0007 (4) | 0.42699 (18) | 0.1505 (14) | |
| H22G | 0.6552 | 0.0724 | 0.4056 | 0.226* | |
| H22H | 0.6671 | 0.0004 | 0.4818 | 0.226* | |
| H22I | 0.6075 | −0.0777 | 0.4031 | 0.226* | |
| O11A | 0.6956 (3) | 0.0099 (2) | −0.01446 (12) | 0.1277 (8) | |
| O12A | 0.5719 (3) | 0.0006 (2) | 0.09493 (11) | 0.1222 (8) | |
| H1A | 0.4401 | 0.0007 | 0.0614 | 0.25 (2)* | |
| O21A | 0.8640 (2) | 0.0145 (2) | 0.33072 (11) | 0.1139 (7) | |
| C1B | 0.8087 (3) | 0.3534 (3) | 0.13578 (17) | 0.0841 (8) | |
| C2B | 0.8061 (3) | 0.3390 (2) | 0.05543 (17) | 0.0844 (8) | |
| C3B | 0.6716 (4) | 0.3333 (3) | 0.00966 (18) | 0.1099 (10) | |
| H3B | 0.6723 | 0.3244 | −0.0439 | 0.132* | |
| C4B | 0.5353 (4) | 0.3409 (3) | 0.0433 (2) | 0.1279 (12) | |
| H4B | 0.4437 | 0.3365 | 0.0122 | 0.154* | |
| C5B | 0.5328 (4) | 0.3548 (3) | 0.1219 (3) | 0.1298 (12) | |
| H5B | 0.4404 | 0.3600 | 0.1442 | 0.156* | |
| C6B | 0.6677 (4) | 0.3611 (3) | 0.16708 (19) | 0.1104 (10) | |
| H6B | 0.6657 | 0.3708 | 0.2205 | 0.132* | |
| C11B | 0.9523 (3) | 0.3591 (2) | 0.18385 (16) | 0.0854 (8) | |
| H11B | 1.0443 | 0.3642 | 0.1574 | 0.103* | |
| C12B | 0.9690 (4) | 0.3580 (3) | 0.25923 (17) | 0.0977 (9) | |
| H12B | 0.8789 | 0.3525 | 0.2872 | 0.117* | |
| C13B | 1.1196 (4) | 0.3646 (3) | 0.30273 (17) | 0.0923 (9) | |
| C21B | 0.9524 (3) | 0.3149 (3) | −0.05520 (14) | 0.0963 (9) | |
| H21A | 0.9373 | 0.3875 | −0.0815 | 0.116* | |
| H21B | 0.8675 | 0.2375 | −0.0743 | 0.116* | |
| C22B | 1.1154 (3) | 0.3049 (3) | −0.06990 (16) | 0.1147 (11) | |
| H22D | 1.1984 | 0.3826 | −0.0517 | 0.172* | |
| H22E | 1.1229 | 0.2920 | −0.1245 | 0.172* | |
| H22F | 1.1296 | 0.2335 | −0.0430 | 0.172* | |
| O11B | 1.1124 (2) | 0.34918 (19) | 0.37500 (11) | 0.1127 (7) | |
| O12B | 1.2469 (3) | 0.38339 (19) | 0.26725 (10) | 0.1123 (7) | |
| H1B | 1.3781 | 0.3768 | 0.3049 | 0.27 (2)* | |
| O21B | 0.9463 (2) | 0.33174 (18) | 0.02649 (10) | 0.0960 (6) | |
| C1C | 0.2034 (3) | 0.6762 (2) | 0.41845 (17) | 0.0830 (8) | |
| C2C | 0.2154 (4) | 0.6967 (3) | 0.33951 (17) | 0.0885 (8) | |
| C3C | 0.0860 (4) | 0.7036 (3) | 0.29377 (18) | 0.1111 (10) | |
| H3C | 0.0964 | 0.7178 | 0.2412 | 0.133* | |
| C4C | −0.0592 (4) | 0.6896 (3) | 0.3261 (3) | 0.1266 (12) | |
| H4C | −0.1471 | 0.6943 | 0.2952 | 0.152* | |
| C5C | −0.0753 (4) | 0.6686 (3) | 0.4034 (3) | 0.1223 (12) | |
| H5C | −0.1739 | 0.6587 | 0.4250 | 0.147* | |
| C6C | 0.0539 (4) | 0.6624 (3) | 0.44855 (18) | 0.1069 (10) | |
| H6C | 0.0419 | 0.6486 | 0.5011 | 0.128* | |
| C11C | 0.3410 (4) | 0.6665 (3) | 0.46511 (16) | 0.0891 (8) | |
| H11C | 0.4356 | 0.6781 | 0.4395 | 0.107* | |
| C12C | 0.3486 (4) | 0.6433 (3) | 0.53937 (17) | 0.0999 (9) | |
| H12C | 0.2561 | 0.6314 | 0.5667 | 0.120* | |
| C13C | 0.4941 (4) | 0.6352 (3) | 0.58122 (18) | 0.0996 (9) | |
| C21C | 0.3819 (3) | 0.7144 (3) | 0.23043 (15) | 0.1158 (11) | |
| H21C | 0.3726 | 0.7942 | 0.2117 | 0.139* | |
| H21D | 0.2981 | 0.6435 | 0.2036 | 0.139* | |
| C22C | 0.5435 (4) | 0.7062 (3) | 0.21622 (17) | 0.1325 (12) | |
| H22A | 0.6254 | 0.7781 | 0.2417 | 0.199* | |
| H22B | 0.5579 | 0.7072 | 0.1615 | 0.199* | |
| H22C | 0.5522 | 0.6279 | 0.2362 | 0.199* | |
| O11C | 0.4849 (2) | 0.6081 (2) | 0.65176 (12) | 0.1326 (9) | |
| O12C | 0.6235 (3) | 0.6569 (2) | 0.54658 (10) | 0.1160 (7) | |
| H1C | 0.7462 | 0.6440 | 0.5835 | 0.201 (15)* | |
| O21C | 0.3641 (2) | 0.70890 (19) | 0.31218 (10) | 0.1068 (7) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C1A | 0.106 (2) | 0.089 (2) | 0.081 (2) | 0.0312 (18) | 0.006 (2) | 0.0055 (17) |
| C2A | 0.098 (2) | 0.102 (3) | 0.087 (2) | 0.0349 (19) | 0.003 (2) | 0.0014 (18) |
| C3A | 0.112 (2) | 0.149 (3) | 0.101 (2) | 0.052 (2) | −0.014 (2) | 0.003 (2) |
| C4A | 0.109 (3) | 0.145 (3) | 0.153 (4) | 0.050 (2) | −0.011 (3) | −0.002 (3) |
| C5A | 0.108 (3) | 0.145 (3) | 0.146 (4) | 0.045 (2) | 0.023 (3) | 0.003 (3) |
| C6A | 0.120 (3) | 0.118 (3) | 0.111 (3) | 0.036 (2) | 0.019 (3) | 0.001 (2) |
| C11A | 0.142 (3) | 0.083 (2) | 0.070 (2) | 0.0325 (19) | 0.013 (2) | 0.0074 (17) |
| C12A | 0.140 (3) | 0.099 (3) | 0.087 (2) | 0.035 (2) | 0.024 (2) | 0.0069 (19) |
| C13A | 0.131 (3) | 0.091 (3) | 0.069 (2) | 0.025 (2) | −0.001 (2) | 0.0042 (18) |
| C21A | 0.143 (3) | 0.171 (3) | 0.076 (2) | 0.062 (2) | 0.007 (2) | 0.004 (2) |
| C22A | 0.124 (3) | 0.212 (4) | 0.121 (3) | 0.061 (3) | 0.022 (2) | 0.003 (3) |
| O11A | 0.1470 (18) | 0.155 (2) | 0.0712 (14) | 0.0362 (14) | 0.0076 (14) | 0.0013 (13) |
| O12A | 0.1454 (19) | 0.151 (2) | 0.0690 (14) | 0.0458 (15) | 0.0129 (14) | 0.0118 (12) |
| O21A | 0.1052 (15) | 0.161 (2) | 0.0768 (14) | 0.0465 (13) | 0.0001 (12) | 0.0059 (12) |
| C1B | 0.088 (2) | 0.089 (2) | 0.078 (2) | 0.0329 (16) | 0.0091 (18) | 0.0096 (16) |
| C2B | 0.081 (2) | 0.095 (2) | 0.077 (2) | 0.0292 (17) | 0.0028 (18) | 0.0045 (16) |
| C3B | 0.090 (2) | 0.146 (3) | 0.095 (2) | 0.043 (2) | −0.009 (2) | 0.0045 (19) |
| C4B | 0.092 (2) | 0.158 (3) | 0.136 (3) | 0.045 (2) | −0.005 (2) | 0.016 (3) |
| C5B | 0.094 (3) | 0.165 (4) | 0.140 (3) | 0.053 (2) | 0.024 (3) | 0.017 (3) |
| C6B | 0.114 (2) | 0.127 (3) | 0.097 (2) | 0.046 (2) | 0.024 (2) | 0.0127 (19) |
| C11B | 0.102 (2) | 0.085 (2) | 0.071 (2) | 0.0313 (17) | 0.0058 (17) | 0.0053 (15) |
| C12B | 0.117 (3) | 0.109 (3) | 0.069 (2) | 0.0397 (19) | 0.0133 (19) | 0.0035 (17) |
| C13B | 0.117 (3) | 0.094 (2) | 0.066 (2) | 0.037 (2) | −0.001 (2) | −0.0010 (17) |
| C21B | 0.101 (2) | 0.110 (3) | 0.071 (2) | 0.0257 (18) | −0.0009 (16) | 0.0052 (16) |
| C22B | 0.111 (2) | 0.138 (3) | 0.105 (2) | 0.053 (2) | 0.0202 (19) | 0.004 (2) |
| O11B | 0.1368 (17) | 0.147 (2) | 0.0601 (13) | 0.0539 (14) | 0.0053 (12) | 0.0068 (12) |
| O12B | 0.1163 (16) | 0.146 (2) | 0.0698 (14) | 0.0353 (14) | 0.0081 (12) | 0.0161 (11) |
| O21B | 0.0953 (13) | 0.1299 (17) | 0.0677 (13) | 0.0447 (11) | −0.0014 (10) | −0.0007 (10) |
| C1C | 0.088 (2) | 0.087 (2) | 0.075 (2) | 0.0294 (16) | 0.0045 (18) | 0.0030 (15) |
| C2C | 0.087 (2) | 0.099 (2) | 0.079 (2) | 0.0297 (17) | −0.0009 (19) | −0.0010 (17) |
| C3C | 0.105 (2) | 0.144 (3) | 0.090 (2) | 0.050 (2) | −0.007 (2) | 0.0064 (19) |
| C4C | 0.101 (3) | 0.141 (3) | 0.143 (3) | 0.048 (2) | −0.005 (2) | 0.003 (3) |
| C5C | 0.095 (3) | 0.129 (3) | 0.146 (3) | 0.038 (2) | 0.021 (3) | 0.009 (3) |
| C6C | 0.108 (2) | 0.115 (3) | 0.102 (2) | 0.039 (2) | 0.022 (2) | 0.0119 (19) |
| C11C | 0.105 (2) | 0.095 (2) | 0.0667 (19) | 0.0320 (17) | 0.0093 (17) | 0.0047 (16) |
| C12C | 0.104 (2) | 0.118 (3) | 0.075 (2) | 0.0309 (19) | 0.0115 (19) | 0.0067 (18) |
| C13C | 0.112 (3) | 0.112 (3) | 0.067 (2) | 0.026 (2) | 0.006 (2) | 0.0026 (18) |
| C21C | 0.120 (2) | 0.158 (3) | 0.075 (2) | 0.052 (2) | 0.0100 (18) | 0.0235 (19) |
| C22C | 0.115 (2) | 0.183 (4) | 0.111 (3) | 0.060 (2) | 0.028 (2) | 0.027 (2) |
| O11C | 0.1280 (17) | 0.194 (2) | 0.0659 (14) | 0.0362 (15) | 0.0078 (13) | 0.0279 (14) |
| O12C | 0.1171 (16) | 0.164 (2) | 0.0708 (14) | 0.0500 (14) | 0.0059 (12) | 0.0120 (12) |
| O21C | 0.0939 (14) | 0.159 (2) | 0.0701 (13) | 0.0436 (12) | 0.0044 (11) | 0.0129 (11) |
Geometric parameters (Å, º) top
| C1A—C2A | 1.389 (3) | C11B—H11B | 0.9300 |
| C1A—C6A | 1.394 (4) | C12B—C13B | 1.463 (4) |
| C1A—C11A | 1.470 (4) | C12B—H12B | 0.9300 |
| C2A—O21A | 1.368 (3) | C13B—O12B | 1.256 (3) |
| C2A—C3A | 1.370 (4) | C13B—O11B | 1.267 (3) |
| C3A—C4A | 1.371 (4) | C21B—O21B | 1.427 (2) |
| C3A—H3A | 0.9300 | C21B—C22B | 1.498 (3) |
| C4A—C5A | 1.366 (4) | C21B—H21A | 0.9700 |
| C4A—H4A | 0.9300 | C21B—H21B | 0.9700 |
| C5A—C6A | 1.367 (4) | C22B—H22D | 0.9600 |
| C5A—H5A | 0.9300 | C22B—H22E | 0.9600 |
| C6A—H6A | 0.9300 | C22B—H22F | 0.9600 |
| C11A—C12A | 1.285 (3) | O12B—H1B | 1.3137 |
| C11A—H11A | 0.9300 | C1C—C2C | 1.392 (3) |
| C12A—C13A | 1.487 (4) | C1C—C6C | 1.394 (3) |
| C12A—H12A | 0.9300 | C1C—C11C | 1.449 (3) |
| C13A—O11A | 1.244 (3) | C2C—O21C | 1.370 (3) |
| C13A—O12A | 1.266 (3) | C2C—C3C | 1.372 (3) |
| C21A—O21A | 1.425 (3) | C3C—C4C | 1.375 (4) |
| C21A—C22A | 1.472 (4) | C3C—H3C | 0.9300 |
| C21A—H21E | 0.9700 | C4C—C5C | 1.366 (4) |
| C21A—H21F | 0.9700 | C4C—H4C | 0.9300 |
| C22A—H22G | 0.9600 | C5C—C6C | 1.362 (4) |
| C22A—H22H | 0.9600 | C5C—H5C | 0.9300 |
| C22A—H22I | 0.9600 | C6C—H6C | 0.9300 |
| O12A—H1A | 1.2645 | C11C—C12C | 1.317 (3) |
| C1B—C2B | 1.394 (3) | C11C—H11C | 0.9300 |
| C1B—C6B | 1.396 (3) | C12C—C13C | 1.461 (4) |
| C1B—C11B | 1.454 (3) | C12C—H12C | 0.9300 |
| C2B—C3B | 1.368 (3) | C13C—O12C | 1.261 (3) |
| C2B—O21B | 1.371 (3) | C13C—O11C | 1.262 (3) |
| C3B—C4B | 1.376 (3) | C21C—O21C | 1.430 (3) |
| C3B—H3B | 0.9300 | C21C—C22C | 1.476 (3) |
| C4B—C5B | 1.366 (4) | C21C—H21C | 0.9700 |
| C4B—H4B | 0.9300 | C21C—H21D | 0.9700 |
| C5B—C6B | 1.364 (4) | C22C—H22A | 0.9600 |
| C5B—H5B | 0.9300 | C22C—H22B | 0.9600 |
| C6B—H6B | 0.9300 | C22C—H22C | 0.9600 |
| C11B—C12B | 1.306 (3) | O12C—H1C | 1.2714 |
| | | |
| C2A—C1A—C6A | 117.3 (3) | C11B—C12B—C13B | 124.5 (3) |
| C2A—C1A—C11A | 119.7 (3) | C11B—C12B—H12B | 117.7 |
| C6A—C1A—C11A | 123.0 (3) | C13B—C12B—H12B | 117.7 |
| O21A—C2A—C3A | 123.7 (3) | O12B—C13B—O11B | 123.7 (3) |
| O21A—C2A—C1A | 115.3 (3) | O12B—C13B—C12B | 119.3 (3) |
| C3A—C2A—C1A | 121.0 (3) | O11B—C13B—C12B | 117.0 (3) |
| C2A—C3A—C4A | 119.8 (3) | O21B—C21B—C22B | 107.3 (2) |
| C2A—C3A—H3A | 120.1 | O21B—C21B—H21A | 110.2 |
| C4A—C3A—H3A | 120.1 | C22B—C21B—H21A | 110.2 |
| C5A—C4A—C3A | 121.0 (4) | O21B—C21B—H21B | 110.2 |
| C5A—C4A—H4A | 119.5 | C22B—C21B—H21B | 110.2 |
| C3A—C4A—H4A | 119.5 | H21A—C21B—H21B | 108.5 |
| C4A—C5A—C6A | 119.0 (3) | C21B—C22B—H22D | 109.5 |
| C4A—C5A—H5A | 120.5 | C21B—C22B—H22E | 109.5 |
| C6A—C5A—H5A | 120.5 | H22D—C22B—H22E | 109.5 |
| C5A—C6A—C1A | 121.9 (3) | C21B—C22B—H22F | 109.5 |
| C5A—C6A—H6A | 119.0 | H22D—C22B—H22F | 109.5 |
| C1A—C6A—H6A | 119.0 | H22E—C22B—H22F | 109.5 |
| C12A—C11A—C1A | 127.3 (3) | C13B—O12B—H1B | 119.2 |
| C12A—C11A—H11A | 116.4 | C2B—O21B—C21B | 118.8 (2) |
| C1A—C11A—H11A | 116.4 | C2C—C1C—C6C | 116.9 (3) |
| C11A—C12A—C13A | 123.5 (3) | C2C—C1C—C11C | 120.2 (3) |
| C11A—C12A—H12A | 118.3 | C6C—C1C—C11C | 122.9 (3) |
| C13A—C12A—H12A | 118.3 | O21C—C2C—C3C | 123.6 (3) |
| O11A—C13A—O12A | 123.6 (3) | O21C—C2C—C1C | 115.0 (3) |
| O11A—C13A—C12A | 117.2 (3) | C3C—C2C—C1C | 121.4 (3) |
| O12A—C13A—C12A | 119.2 (3) | C2C—C3C—C4C | 119.7 (3) |
| O21A—C21A—C22A | 108.0 (3) | C2C—C3C—H3C | 120.2 |
| O21A—C21A—H21E | 110.1 | C4C—C3C—H3C | 120.2 |
| C22A—C21A—H21E | 110.1 | C5C—C4C—C3C | 120.4 (3) |
| O21A—C21A—H21F | 110.1 | C5C—C4C—H4C | 119.8 |
| C22A—C21A—H21F | 110.1 | C3C—C4C—H4C | 119.8 |
| H21E—C21A—H21F | 108.4 | C6C—C5C—C4C | 119.6 (3) |
| C21A—C22A—H22G | 109.5 | C6C—C5C—H5C | 120.2 |
| C21A—C22A—H22H | 109.5 | C4C—C5C—H5C | 120.2 |
| H22G—C22A—H22H | 109.5 | C5C—C6C—C1C | 122.0 (3) |
| C21A—C22A—H22I | 109.5 | C5C—C6C—H6C | 119.0 |
| H22G—C22A—H22I | 109.5 | C1C—C6C—H6C | 119.0 |
| H22H—C22A—H22I | 109.5 | C12C—C11C—C1C | 128.0 (3) |
| C13A—O12A—H1A | 121.5 | C12C—C11C—H11C | 116.0 |
| C2A—O21A—C21A | 119.2 (2) | C1C—C11C—H11C | 116.0 |
| C2B—C1B—C6B | 116.8 (3) | C11C—C12C—C13C | 123.7 (3) |
| C2B—C1B—C11B | 120.9 (3) | C11C—C12C—H12C | 118.1 |
| C6B—C1B—C11B | 122.3 (3) | C13C—C12C—H12C | 118.1 |
| C3B—C2B—O21B | 123.2 (3) | O12C—C13C—O11C | 122.5 (3) |
| C3B—C2B—C1B | 121.3 (3) | O12C—C13C—C12C | 119.6 (3) |
| O21B—C2B—C1B | 115.4 (3) | O11C—C13C—C12C | 117.9 (3) |
| C2B—C3B—C4B | 119.6 (3) | O21C—C21C—C22C | 107.5 (2) |
| C2B—C3B—H3B | 120.2 | O21C—C21C—H21C | 110.2 |
| C4B—C3B—H3B | 120.2 | C22C—C21C—H21C | 110.2 |
| C5B—C4B—C3B | 120.9 (3) | O21C—C21C—H21D | 110.2 |
| C5B—C4B—H4B | 119.6 | C22C—C21C—H21D | 110.2 |
| C3B—C4B—H4B | 119.6 | H21C—C21C—H21D | 108.5 |
| C6B—C5B—C4B | 119.2 (3) | C21C—C22C—H22A | 109.5 |
| C6B—C5B—H5B | 120.4 | C21C—C22C—H22B | 109.5 |
| C4B—C5B—H5B | 120.4 | H22A—C22C—H22B | 109.5 |
| C5B—C6B—C1B | 122.2 (3) | C21C—C22C—H22C | 109.5 |
| C5B—C6B—H6B | 118.9 | H22A—C22C—H22C | 109.5 |
| C1B—C6B—H6B | 118.9 | H22B—C22C—H22C | 109.5 |
| C12B—C11B—C1B | 128.5 (3) | C13C—O12C—H1C | 118.1 |
| C12B—C11B—H11B | 115.8 | C2C—O21C—C21C | 118.8 (2) |
| C1B—C11B—H11B | 115.8 | | |
| | | |
| C6A—C1A—C2A—O21A | 179.9 (2) | C11B—C1B—C6B—C5B | −179.6 (3) |
| C11A—C1A—C2A—O21A | 0.4 (4) | C2B—C1B—C11B—C12B | −171.7 (3) |
| C6A—C1A—C2A—C3A | 0.1 (4) | C6B—C1B—C11B—C12B | 7.9 (5) |
| C11A—C1A—C2A—C3A | −179.4 (3) | C1B—C11B—C12B—C13B | −179.7 (3) |
| O21A—C2A—C3A—C4A | −179.7 (3) | C11B—C12B—C13B—O12B | 6.2 (4) |
| C1A—C2A—C3A—C4A | 0.0 (5) | C11B—C12B—C13B—O11B | −172.9 (3) |
| C2A—C3A—C4A—C5A | 0.0 (5) | C3B—C2B—O21B—C21B | −1.3 (4) |
| C3A—C4A—C5A—C6A | −0.3 (6) | C1B—C2B—O21B—C21B | 179.0 (2) |
| C4A—C5A—C6A—C1A | 0.4 (5) | C22B—C21B—O21B—C2B | −177.7 (2) |
| C2A—C1A—C6A—C5A | −0.4 (5) | C6C—C1C—C2C—O21C | −179.6 (2) |
| C11A—C1A—C6A—C5A | 179.2 (3) | C11C—C1C—C2C—O21C | −1.3 (4) |
| C2A—C1A—C11A—C12A | −177.0 (3) | C6C—C1C—C2C—C3C | 0.3 (4) |
| C6A—C1A—C11A—C12A | 3.4 (5) | C11C—C1C—C2C—C3C | 178.7 (2) |
| C1A—C11A—C12A—C13A | 178.5 (3) | O21C—C2C—C3C—C4C | 179.7 (3) |
| C11A—C12A—C13A—O11A | −180.0 (3) | C1C—C2C—C3C—C4C | −0.3 (5) |
| C11A—C12A—C13A—O12A | 0.0 (5) | C2C—C3C—C4C—C5C | −0.1 (5) |
| C3A—C2A—O21A—C21A | 1.3 (4) | C3C—C4C—C5C—C6C | 0.4 (5) |
| C1A—C2A—O21A—C21A | −178.5 (2) | C4C—C5C—C6C—C1C | −0.3 (5) |
| C22A—C21A—O21A—C2A | 177.4 (3) | C2C—C1C—C6C—C5C | 0.0 (4) |
| C6B—C1B—C2B—C3B | 0.2 (4) | C11C—C1C—C6C—C5C | −178.3 (3) |
| C11B—C1B—C2B—C3B | 179.9 (3) | C2C—C1C—C11C—C12C | −177.6 (3) |
| C6B—C1B—C2B—O21B | 180.0 (2) | C6C—C1C—C11C—C12C | 0.7 (4) |
| C11B—C1B—C2B—O21B | −0.4 (4) | C1C—C11C—C12C—C13C | 179.9 (3) |
| O21B—C2B—C3B—C4B | 179.8 (3) | C11C—C12C—C13C—O12C | 3.3 (5) |
| C1B—C2B—C3B—C4B | −0.5 (4) | C11C—C12C—C13C—O11C | −177.6 (3) |
| C2B—C3B—C4B—C5B | 0.4 (5) | C3C—C2C—O21C—C21C | −7.6 (4) |
| C3B—C4B—C5B—C6B | −0.1 (6) | C1C—C2C—O21C—C21C | 172.4 (2) |
| C4B—C5B—C6B—C1B | −0.2 (5) | C22C—C21C—O21C—C2C | −171.0 (2) |
| C2B—C1B—C6B—C5B | 0.1 (4) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O12C—H1C···O11Bi | 1.27 | 1.38 | 2.640 (3) | 171 |
| O12B—H1B···O11Ci | 1.31 | 1.34 | 2.646 (3) | 170 |
| O12A—H1A···O11Aii | 1.26 | 1.38 | 2.630 (3) | 170 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
(d6h_s) (
E)-3-(2-Ethoxyphenyl)propenoic acid and
trans-2,4-bis(2-ethoxyphenyl)-
trans-1,3-cyclobutane-dicarboxylic acid
top
Crystal data top
| 2.738(C11H12O3)·0.131(C22H24O6) | Z = 2 |
| Mr = 576.62 | F(000) = 612 |
| Triclinic, P1 | Dx = 1.247 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 8.6759 (11) Å | Cell parameters from 781 reflections |
| b = 11.0003 (13) Å | θ = 2.3–21.4° |
| c = 17.299 (2) Å | µ = 0.09 mm−1 |
| α = 92.552 (3)° | T = 293 K |
| β = 92.347 (2)° | Irregular, white |
| γ = 111.155 (3)° | 0.38 × 0.33 × 0.26 mm |
| V = 1535.3 (3) Å3 | |
Data collection top
Bruker SMART 1K
diffractometer | 2192 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.028 |
| Graphite monochromator | θmax = 25.0°, θmin = 1.2° |
| phi and ω scans | h = −10→10 |
| 8251 measured reflections | k = −11→13 |
| 5391 independent reflections | l = −20→14 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.068 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.220 | w = 1/[σ2(Fo2) + (0.1128P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 0.90 | (Δ/σ)max < 0.001 |
| 5391 reflections | Δρmax = 0.19 e Å−3 |
| 391 parameters | Δρmin = −0.18 e Å−3 |
| 0 restraints | Extinction correction: SHELXL, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.008 (2) |
Crystal data top
| 2.738(C11H12O3)·0.131(C22H24O6) | γ = 111.155 (3)° |
| Mr = 576.62 | V = 1535.3 (3) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 8.6759 (11) Å | Mo Kα radiation |
| b = 11.0003 (13) Å | µ = 0.09 mm−1 |
| c = 17.299 (2) Å | T = 293 K |
| α = 92.552 (3)° | 0.38 × 0.33 × 0.26 mm |
| β = 92.347 (2)° | |
Data collection top
Bruker SMART 1K
diffractometer | 2192 reflections with I > 2σ(I) |
| 8251 measured reflections | Rint = 0.028 |
| 5391 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.068 | 0 restraints |
| wR(F2) = 0.220 | H atoms treated by a mixture of independent and constrained refinement |
| S = 0.90 | Δρmax = 0.19 e Å−3 |
| 5391 reflections | Δρmin = −0.18 e Å−3 |
| 391 parameters | |
Special details top
Experimental. The following alerts are triggered when using the ACTA-C CIF validation software. PLAT301_ALERT_3_B Main Residue Disorder ························. 40.00 Perc. PLAT432_ALERT_2_B Short Inter X···Y Contact C3C.. C4D.. 3.05 A ng. PLAT432_ALERT_2_B Short Inter X···Y Contact C4C.. C3D.. 3.07 A ng. PLAT165_ALERT_3_C Nr. of Status R Flagged Non-Hydrogen Atoms ···.. 28 PLAT340_ALERT_3_C Low Bond Precision on C—C bonds (x 1000) Ang ··· 6 PLAT432_ALERT_2_C Short Inter X···Y Contact C3C.. C3D.. 3.13 A ng. —— Authors comment: The assymetric unit consists of a mixture of the monomer and dimer. The short contacts are most probably due to the dimer not being properly optimized for the crystal structure at this point. Coordinates for the dimer molecule at a higher conversion stage was used as a starting point for the refinement. The dimer was also refined isotropically as a rigid body. This is necessary as the dimer is only present in about 4–5% occupancy (4% dimer occupancy = 8% of monomer consumed). PLAT026_ALERT_3_C Ratio Observed / Unique Reflections too Low ···. 41 Perc. ——- Authors comment: The crystal was exposed to 6 h to UV light allowing some reaction to occur. This has most probably caused a certain amount of disorder leading to higher angle reflections having lower intensity than expected·The number of observed reflections has most probably decreased as a consequence. PLAT042_ALERT_1_C Calc. and Rep. MoietyFormula Strings Differ ···. ? —— Authors commment: Our moiety formula is correct as it takes the occupancy of the dimer and monomer molecules into account. |
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| C1A | 1.0151 (6) | 0.0304 (4) | 0.2213 (2) | 0.0733 (12) | 0.869 (3) |
| C2A | 1.0109 (6) | 0.0251 (4) | 0.3015 (2) | 0.0761 (12) | 0.869 (3) |
| C3A | 1.1502 (6) | 0.0346 (4) | 0.3466 (3) | 0.0924 (14) | 0.869 (3) |
| H3A | 1.1462 | 0.0325 | 0.4002 | 0.111* | 0.869 (3) |
| C4A | 1.2944 (6) | 0.0472 (5) | 0.3126 (4) | 0.1040 (16) | 0.869 (3) |
| H4A | 1.3873 | 0.0515 | 0.3430 | 0.125* | 0.869 (3) |
| C5A | 1.3022 (7) | 0.0535 (5) | 0.2333 (4) | 0.1062 (17) | 0.869 (3) |
| H5A | 1.4009 | 0.0647 | 0.2102 | 0.127* | 0.869 (3) |
| C6A | 1.1624 (8) | 0.0431 (5) | 0.1888 (3) | 0.0976 (15) | 0.869 (3) |
| H6A | 1.1673 | 0.0446 | 0.1353 | 0.117* | 0.869 (3) |
| C11A | 0.8643 (5) | 0.0194 (3) | 0.1735 (2) | 0.0738 (12) | 0.869 (3) |
| H11A | 0.7708 | 0.0117 | 0.2003 | 0.089* | 0.869 (3) |
| C12A | 0.8480 (6) | 0.0194 (4) | 0.0976 (2) | 0.0814 (13) | 0.869 (3) |
| H12A | 0.9397 | 0.0272 | 0.0693 | 0.098* | 0.869 (3) |
| C13A | 0.6944 (6) | 0.0077 (4) | 0.0553 (3) | 0.0809 (13) | 0.869 (3) |
| C21A | 0.8468 (7) | 0.0093 (5) | 0.4133 (2) | 0.0951 (14) | 0.869 (3) |
| H21C | 0.9285 | 0.0874 | 0.4391 | 0.114* | 0.869 (3) |
| H21D | 0.8647 | −0.0663 | 0.4326 | 0.114* | 0.869 (3) |
| C22A | 0.6766 (7) | 0.0035 (6) | 0.4279 (3) | 0.1086 (16) | 0.869 (3) |
| H22D | 0.6604 | 0.0785 | 0.4083 | 0.163* | 0.869 (3) |
| H22E | 0.6624 | 0.0026 | 0.4827 | 0.163* | 0.869 (3) |
| H22F | 0.5972 | −0.0745 | 0.4022 | 0.163* | 0.869 (3) |
| O11A | 0.6914 (4) | 0.0087 (3) | −0.01701 (17) | 0.0957 (10) | 0.869 (3) |
| O12A | 0.5711 (5) | −0.0035 (3) | 0.09473 (19) | 0.1040 (11) | 0.869 (3) |
| H1A | 0.4916 | −0.0085 | 0.0657 | 0.156* | 0.869 (3) |
| O21A | 0.8606 (5) | 0.0111 (4) | 0.33080 (16) | 0.0899 (9) | 0.869 (3) |
| C1B | 0.7939 (5) | 0.3471 (4) | 0.1306 (2) | 0.0661 (10) | 0.869 (3) |
| C2B | 0.7929 (5) | 0.3366 (4) | 0.0494 (2) | 0.0657 (10) | 0.869 (3) |
| C3B | 0.6538 (6) | 0.3308 (4) | 0.0043 (3) | 0.0848 (14) | 0.869 (3) |
| H3B | 0.6531 | 0.3218 | −0.0495 | 0.102* | 0.869 (3) |
| C4B | 0.5175 (6) | 0.3384 (5) | 0.0389 (3) | 0.1009 (16) | 0.869 (3) |
| H4B | 0.4255 | 0.3358 | 0.0084 | 0.121* | 0.869 (3) |
| C5B | 0.5164 (7) | 0.3497 (6) | 0.1182 (4) | 0.1037 (16) | 0.869 (3) |
| H5B | 0.4234 | 0.3536 | 0.1414 | 0.124* | 0.869 (3) |
| C6B | 0.6547 (6) | 0.3553 (4) | 0.1632 (3) | 0.0861 (13) | 0.869 (3) |
| H6B | 0.6540 | 0.3648 | 0.2169 | 0.103* | 0.869 (3) |
| C11B | 0.9396 (5) | 0.3533 (3) | 0.1780 (2) | 0.0636 (10) | 0.869 (3) |
| H11B | 1.0333 | 0.3595 | 0.1518 | 0.076* | 0.869 (3) |
| C12B | 0.9532 (5) | 0.3510 (4) | 0.2543 (2) | 0.0736 (12) | 0.869 (3) |
| H12B | 0.8616 | 0.3445 | 0.2822 | 0.088* | 0.869 (3) |
| C13B | 1.1052 (6) | 0.3584 (4) | 0.2963 (2) | 0.0724 (12) | 0.869 (3) |
| C21B | 0.9379 (6) | 0.3138 (4) | −0.0625 (2) | 0.0761 (12) | 0.869 (3) |
| H21E | 0.9277 | 0.3886 | −0.0867 | 0.091* | 0.869 (3) |
| H21F | 0.8477 | 0.2359 | −0.0832 | 0.091* | 0.869 (3) |
| C22B | 1.1002 (6) | 0.3028 (5) | −0.0782 (2) | 0.0921 (14) | 0.869 (3) |
| H22G | 1.1884 | 0.3815 | −0.0589 | 0.138* | 0.869 (3) |
| H22H | 1.1067 | 0.2900 | −0.1331 | 0.138* | 0.869 (3) |
| H22I | 1.1100 | 0.2299 | −0.0528 | 0.138* | 0.869 (3) |
| O11B | 1.0958 (4) | 0.3432 (3) | 0.36910 (15) | 0.0883 (10) | 0.869 (3) |
| H1B | 1.1862 | 0.3480 | 0.3881 | 0.132* | 0.869 (3) |
| O12B | 1.2360 (4) | 0.3790 (3) | 0.26206 (17) | 0.0877 (9) | 0.869 (3) |
| O21B | 0.9329 (4) | 0.3290 (3) | 0.01970 (14) | 0.0751 (8) | 0.869 (3) |
| C1C | 0.2163 (4) | 0.6710 (3) | 0.42390 (19) | 0.0688 (9) | |
| C2C | 0.2291 (4) | 0.6868 (3) | 0.34529 (19) | 0.0743 (10) | |
| C3C | 0.0957 (5) | 0.6907 (4) | 0.2992 (2) | 0.0975 (12) | |
| H3C | 0.1049 | 0.7015 | 0.2464 | 0.117* | |
| C4C | −0.0505 (5) | 0.6785 (4) | 0.3330 (3) | 0.1070 (13) | |
| H4C | −0.1406 | 0.6800 | 0.3026 | 0.128* | |
| C5C | −0.0636 (6) | 0.6640 (4) | 0.4113 (3) | 0.1041 (13) | |
| H5C | −0.1617 | 0.6570 | 0.4338 | 0.125* | |
| C6C | 0.0680 (5) | 0.6598 (3) | 0.4562 (2) | 0.0882 (11) | |
| H6C | 0.0580 | 0.6494 | 0.5091 | 0.106* | |
| C11C | 0.3570 (4) | 0.6642 (3) | 0.47092 (19) | 0.0700 (9) | |
| H11C | 0.4524 | 0.6741 | 0.4451 | 0.084* | |
| C12C | 0.3643 (5) | 0.6455 (3) | 0.54595 (19) | 0.0805 (10) | |
| H12C | 0.2720 | 0.6366 | 0.5739 | 0.097* | |
| C13C | 0.5116 (5) | 0.6382 (3) | 0.58652 (19) | 0.0796 (11) | |
| C21C | 0.3944 (4) | 0.7049 (4) | 0.23477 (18) | 0.0846 (11) | |
| H21A | 0.3861 | 0.7855 | 0.2180 | 0.102* | |
| H21B | 0.3080 | 0.6320 | 0.2068 | 0.102* | |
| C22C | 0.5601 (4) | 0.7008 (4) | 0.2197 (2) | 0.0953 (12) | |
| H22A | 0.6443 | 0.7729 | 0.2481 | 0.143* | |
| H22B | 0.5769 | 0.7067 | 0.1653 | 0.143* | |
| H22C | 0.5662 | 0.6203 | 0.2360 | 0.143* | |
| O11C | 0.4971 (3) | 0.6124 (3) | 0.65788 (13) | 0.1059 (9) | |
| H1C | 0.5846 | 0.6101 | 0.6763 | 0.159* | |
| O12C | 0.6431 (3) | 0.6588 (2) | 0.55318 (13) | 0.0957 (8) | |
| O21C | 0.3774 (3) | 0.6973 (2) | 0.31633 (12) | 0.0854 (7) | |
| C1D | 1.0193 (15) | 0.0446 (10) | 0.2376 (7) | 0.078 (3)* | 0.131 (3) |
| C2D | 1.0034 (18) | 0.0066 (12) | 0.3131 (7) | 0.078 (3)* | 0.131 (3) |
| C3D | 1.129 (2) | −0.0246 (15) | 0.3525 (9) | 0.078 (3)* | 0.131 (3) |
| H3D | 1.1161 | −0.0523 | 0.4026 | 0.094* | 0.131 (3) |
| C4D | 1.271 (2) | −0.0131 (18) | 0.3149 (10) | 0.078 (3)* | 0.131 (3) |
| H4D | 1.3555 | −0.0315 | 0.3409 | 0.094* | 0.131 (3) |
| C5D | 1.2909 (19) | 0.0247 (18) | 0.2406 (10) | 0.078 (3)* | 0.131 (3) |
| H5D | 1.3862 | 0.0306 | 0.2158 | 0.094* | 0.131 (3) |
| C6D | 1.1654 (16) | 0.0541 (14) | 0.2029 (8) | 0.078 (3)* | 0.131 (3) |
| H6D | 1.1792 | 0.0811 | 0.1526 | 0.094* | 0.131 (3) |
| C11D | 0.8894 (12) | 0.0911 (8) | 0.2048 (6) | 0.078 (3)* | 0.131 (3) |
| H11D | 0.7790 | 0.0294 | 0.2146 | 0.094* | 0.131 (3) |
| C12D | 0.8940 (12) | 0.1274 (9) | 0.1198 (5) | 0.078 (3)* | 0.131 (3) |
| H12D | 0.9920 | 0.1235 | 0.0952 | 0.094* | 0.131 (3) |
| C13D | 0.7332 (14) | 0.0555 (10) | 0.0702 (6) | 0.078 (3)* | 0.131 (3) |
| C21D | 0.845 (2) | −0.0164 (18) | 0.4253 (7) | 0.078 (3)* | 0.131 (3) |
| H21G | 0.9355 | 0.0516 | 0.4542 | 0.094* | 0.131 (3) |
| H21H | 0.8485 | −0.1004 | 0.4376 | 0.094* | 0.131 (3) |
| C22D | 0.681 (2) | −0.008 (2) | 0.4465 (8) | 0.078 (3)* | 0.131 (3) |
| H22J | 0.6687 | −0.0181 | 0.5010 | 0.117* | 0.131 (3) |
| H22K | 0.5919 | −0.0756 | 0.4177 | 0.117* | 0.131 (3) |
| H22L | 0.6785 | 0.0758 | 0.4342 | 0.117* | 0.131 (3) |
| O11D | 0.7562 (18) | 0.0505 (13) | −0.0048 (6) | 0.078 (3)* | 0.131 (3) |
| H1D | 0.6673 | 0.0113 | −0.0288 | 0.117* | 0.131 (3) |
| O12D | 0.5962 (13) | 0.0209 (12) | 0.0952 (8) | 0.078 (3)* | 0.131 (3) |
| O21D | 0.8578 (19) | −0.0007 (14) | 0.3443 (7) | 0.078 (3)* | 0.131 (3) |
| C1E | 0.7855 (16) | 0.3239 (11) | 0.1128 (7) | 0.078 (3)* | 0.131 (3) |
| C2E | 0.7950 (19) | 0.3461 (12) | 0.0348 (7) | 0.078 (3)* | 0.131 (3) |
| C3E | 0.669 (2) | 0.3721 (16) | −0.0060 (9) | 0.078 (3)* | 0.131 (3) |
| H3E | 0.6754 | 0.3869 | −0.0584 | 0.094* | 0.131 (3) |
| C4E | 0.533 (2) | 0.3754 (19) | 0.0333 (11) | 0.078 (3)* | 0.131 (3) |
| H4E | 0.4490 | 0.3926 | 0.0066 | 0.094* | 0.131 (3) |
| C5E | 0.522 (2) | 0.3539 (19) | 0.1104 (11) | 0.078 (3)* | 0.131 (3) |
| H5E | 0.4311 | 0.3571 | 0.1362 | 0.094* | 0.131 (3) |
| C6E | 0.6475 (18) | 0.3274 (15) | 0.1497 (9) | 0.078 (3)* | 0.131 (3) |
| H6E | 0.6393 | 0.3115 | 0.2021 | 0.094* | 0.131 (3) |
| C11E | 0.9129 (12) | 0.2741 (8) | 0.1470 (6) | 0.078 (3)* | 0.131 (3) |
| H11E | 1.0244 | 0.3344 | 0.1378 | 0.094* | 0.131 (3) |
| C12E | 0.9060 (12) | 0.2380 (9) | 0.2321 (6) | 0.078 (3)* | 0.131 (3) |
| H12E | 0.8065 | 0.2408 | 0.2558 | 0.094* | 0.131 (3) |
| C13E | 1.0633 (15) | 0.3090 (10) | 0.2832 (6) | 0.078 (3)* | 0.131 (3) |
| C21E | 0.937 (2) | 0.3347 (18) | −0.0810 (7) | 0.078 (3)* | 0.131 (3) |
| H21I | 0.9376 | 0.4155 | −0.1013 | 0.094* | 0.131 (3) |
| H21J | 0.8398 | 0.2637 | −0.1028 | 0.094* | 0.131 (3) |
| C22E | 1.093 (2) | 0.311 (2) | −0.1017 (7) | 0.078 (3)* | 0.131 (3) |
| H22M | 1.0974 | 0.3046 | −0.1571 | 0.117* | 0.131 (3) |
| H22N | 1.0915 | 0.2303 | −0.0813 | 0.117* | 0.131 (3) |
| H22O | 1.1889 | 0.3814 | −0.0799 | 0.117* | 0.131 (3) |
| O11E | 1.0390 (19) | 0.2985 (13) | 0.3569 (6) | 0.078 (3)* | 0.131 (3) |
| H1E | 1.1262 | 0.3378 | 0.3823 | 0.117* | 0.131 (3) |
| O12E | 1.2002 (14) | 0.3558 (11) | 0.2574 (8) | 0.078 (3)* | 0.131 (3) |
| O21E | 0.9371 (19) | 0.3427 (13) | 0.0023 (7) | 0.078 (3)* | 0.131 (3) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C1A | 0.090 (3) | 0.068 (3) | 0.068 (3) | 0.034 (2) | 0.020 (3) | 0.004 (2) |
| C2A | 0.088 (3) | 0.076 (3) | 0.066 (3) | 0.031 (2) | 0.012 (3) | 0.000 (2) |
| C3A | 0.096 (3) | 0.105 (4) | 0.078 (3) | 0.039 (3) | −0.003 (3) | 0.004 (3) |
| C4A | 0.088 (4) | 0.109 (4) | 0.122 (5) | 0.044 (3) | 0.009 (3) | 0.000 (3) |
| C5A | 0.105 (4) | 0.102 (4) | 0.118 (5) | 0.042 (3) | 0.037 (4) | 0.007 (3) |
| C6A | 0.116 (4) | 0.101 (3) | 0.082 (3) | 0.045 (3) | 0.024 (3) | 0.003 (3) |
| C11A | 0.102 (3) | 0.061 (2) | 0.060 (3) | 0.030 (2) | 0.015 (2) | 0.007 (2) |
| C12A | 0.113 (4) | 0.078 (3) | 0.058 (3) | 0.038 (3) | 0.019 (3) | 0.008 (2) |
| C13A | 0.112 (4) | 0.074 (3) | 0.060 (3) | 0.035 (3) | 0.022 (3) | 0.009 (2) |
| C21A | 0.113 (4) | 0.124 (4) | 0.054 (3) | 0.050 (3) | 0.007 (3) | 0.004 (2) |
| C22A | 0.101 (4) | 0.154 (4) | 0.076 (3) | 0.052 (3) | 0.017 (3) | 0.001 (3) |
| O11A | 0.115 (3) | 0.113 (2) | 0.0525 (18) | 0.0311 (19) | 0.0157 (17) | 0.0107 (16) |
| O12A | 0.131 (3) | 0.120 (3) | 0.0630 (19) | 0.046 (2) | 0.023 (2) | 0.0136 (17) |
| O21A | 0.092 (2) | 0.126 (2) | 0.0528 (18) | 0.0407 (17) | 0.0073 (16) | 0.0066 (16) |
| C1B | 0.079 (3) | 0.068 (2) | 0.061 (3) | 0.036 (2) | 0.012 (2) | 0.0075 (19) |
| C2B | 0.076 (3) | 0.073 (3) | 0.053 (2) | 0.033 (2) | 0.007 (2) | 0.0068 (19) |
| C3B | 0.080 (3) | 0.111 (4) | 0.065 (3) | 0.039 (3) | −0.009 (3) | 0.003 (2) |
| C4B | 0.086 (3) | 0.128 (4) | 0.099 (4) | 0.051 (3) | 0.005 (3) | 0.007 (3) |
| C5B | 0.093 (4) | 0.122 (4) | 0.110 (4) | 0.053 (3) | 0.029 (3) | 0.008 (3) |
| C6B | 0.097 (3) | 0.097 (3) | 0.075 (3) | 0.046 (3) | 0.017 (3) | 0.011 (2) |
| C11B | 0.079 (3) | 0.068 (2) | 0.052 (2) | 0.036 (2) | 0.007 (2) | 0.0053 (18) |
| C12B | 0.085 (3) | 0.091 (3) | 0.053 (2) | 0.042 (2) | 0.002 (2) | 0.002 (2) |
| C13B | 0.111 (4) | 0.070 (3) | 0.040 (2) | 0.036 (2) | 0.008 (2) | 0.0067 (19) |
| C21B | 0.088 (3) | 0.094 (3) | 0.046 (2) | 0.032 (2) | 0.003 (2) | 0.003 (2) |
| C22B | 0.099 (3) | 0.126 (4) | 0.067 (3) | 0.057 (3) | 0.021 (3) | 0.006 (3) |
| O11B | 0.108 (3) | 0.115 (2) | 0.0489 (17) | 0.049 (2) | 0.0030 (16) | 0.0063 (15) |
| O12B | 0.102 (2) | 0.112 (2) | 0.0516 (17) | 0.0392 (18) | 0.0158 (17) | 0.0170 (15) |
| O21B | 0.0819 (18) | 0.107 (2) | 0.0458 (16) | 0.0455 (15) | 0.0036 (14) | 0.0015 (14) |
| C1C | 0.075 (2) | 0.074 (2) | 0.062 (2) | 0.0319 (18) | 0.0077 (19) | −0.0015 (17) |
| C2C | 0.078 (2) | 0.087 (2) | 0.055 (2) | 0.0269 (19) | 0.010 (2) | −0.0002 (18) |
| C3C | 0.089 (3) | 0.138 (3) | 0.072 (3) | 0.051 (2) | −0.001 (2) | 0.002 (2) |
| C4C | 0.091 (3) | 0.134 (4) | 0.103 (4) | 0.049 (3) | 0.004 (3) | 0.003 (3) |
| C5C | 0.095 (3) | 0.116 (3) | 0.109 (4) | 0.044 (3) | 0.031 (3) | 0.007 (3) |
| C6C | 0.098 (3) | 0.099 (3) | 0.078 (3) | 0.045 (2) | 0.025 (2) | 0.010 (2) |
| C11C | 0.082 (2) | 0.075 (2) | 0.056 (2) | 0.0313 (18) | 0.0084 (19) | −0.0003 (17) |
| C12C | 0.092 (3) | 0.099 (3) | 0.057 (2) | 0.040 (2) | 0.015 (2) | 0.009 (2) |
| C13C | 0.112 (3) | 0.085 (2) | 0.045 (2) | 0.039 (2) | 0.004 (2) | 0.0030 (18) |
| C21C | 0.090 (3) | 0.115 (3) | 0.055 (2) | 0.043 (2) | 0.0115 (19) | 0.015 (2) |
| C22C | 0.096 (3) | 0.128 (3) | 0.070 (2) | 0.047 (2) | 0.023 (2) | 0.012 (2) |
| O11C | 0.109 (2) | 0.152 (2) | 0.0576 (16) | 0.0455 (17) | 0.0068 (14) | 0.0249 (15) |
| O12C | 0.111 (2) | 0.130 (2) | 0.0551 (15) | 0.0534 (17) | 0.0189 (15) | 0.0098 (14) |
| O21C | 0.0802 (16) | 0.131 (2) | 0.0475 (13) | 0.0408 (14) | 0.0061 (12) | 0.0076 (13) |
Geometric parameters (Å, º) top
| C1A—C6A | 1.380 (5) | C12C—C13C | 1.462 (5) |
| C1A—C2A | 1.394 (5) | C12C—H12C | 0.9300 |
| C1A—C11A | 1.482 (6) | C13C—O12C | 1.251 (4) |
| C2A—O21A | 1.377 (4) | C13C—O11C | 1.280 (4) |
| C2A—C3A | 1.380 (6) | C21C—O21C | 1.428 (3) |
| C3A—C4A | 1.368 (6) | C21C—C22C | 1.488 (4) |
| C3A—H3A | 0.9300 | C21C—H21A | 0.9700 |
| C4A—C5A | 1.380 (6) | C21C—H21B | 0.9700 |
| C4A—H4A | 0.9300 | C22C—H22A | 0.9600 |
| C5A—C6A | 1.375 (6) | C22C—H22B | 0.9600 |
| C5A—H5A | 0.9300 | C22C—H22C | 0.9600 |
| C6A—H6A | 0.9300 | O11C—H1C | 0.8200 |
| C11A—C12A | 1.316 (5) | C1D—C2D | 1.3865 |
| C11A—H11A | 0.9300 | C1D—C6D | 1.3961 |
| C12A—C13A | 1.454 (6) | C1D—C11D | 1.4979 |
| C12A—H12A | 0.9300 | C2D—O21D | 1.3716 |
| C13A—O11A | 1.251 (4) | C2D—C3D | 1.4116 |
| C13A—O12A | 1.264 (5) | C3D—C4D | 1.3885 |
| C21A—O21A | 1.437 (4) | C3D—H3D | 0.9300 |
| C21A—C22A | 1.488 (6) | C4D—C5D | 1.3674 |
| C21A—H21C | 0.9700 | C4D—H4D | 0.9300 |
| C21A—H21D | 0.9700 | C5D—C6D | 1.3890 |
| C22A—H22D | 0.9600 | C5D—H5D | 0.9300 |
| C22A—H22E | 0.9600 | C6D—H6D | 0.9300 |
| C22A—H22F | 0.9600 | C11D—C12D | 1.5394 |
| O12A—H1A | 0.8200 | C11D—C12E | 1.6166 |
| C1B—C6B | 1.384 (5) | C11D—H11D | 0.9800 |
| C1B—C2B | 1.403 (5) | C12D—C13D | 1.5337 |
| C1B—C11B | 1.458 (5) | C12D—C11E | 1.6084 |
| C2B—O21B | 1.367 (4) | C12D—H12D | 0.9800 |
| C2B—C3B | 1.390 (5) | C13D—O12D | 1.2148 |
| C3B—C4B | 1.374 (5) | C13D—O11D | 1.3234 |
| C3B—H3B | 0.9300 | C21D—O21D | 1.4229 |
| C4B—C5B | 1.373 (6) | C21D—C22D | 1.5151 |
| C4B—H4B | 0.9300 | C21D—H21G | 0.9700 |
| C5B—C6B | 1.385 (6) | C21D—H21H | 0.9700 |
| C5B—H5B | 0.9300 | C22D—H22J | 0.9600 |
| C6B—H6B | 0.9300 | C22D—H22K | 0.9600 |
| C11B—C12B | 1.321 (5) | C22D—H22L | 0.9600 |
| C11B—H11B | 0.9300 | O11D—H1D | 0.8200 |
| C12B—C13B | 1.454 (5) | C1E—C2E | 1.3829 |
| C12B—H12B | 0.9300 | C1E—C6E | 1.3919 |
| C13B—O12B | 1.254 (4) | C1E—C11E | 1.5118 |
| C13B—O11B | 1.279 (4) | C2E—O21E | 1.3878 |
| C21B—O21B | 1.429 (4) | C2E—C3E | 1.3985 |
| C21B—C22B | 1.491 (5) | C3E—C4E | 1.3933 |
| C21B—H21E | 0.9700 | C3E—H3E | 0.9300 |
| C21B—H21F | 0.9700 | C4E—C5E | 1.3663 |
| C22B—H22G | 0.9600 | C4E—H4E | 0.9300 |
| C22B—H22H | 0.9600 | C5E—C6E | 1.3865 |
| C22B—H22I | 0.9600 | C5E—H5E | 0.9300 |
| O11B—H1B | 0.8200 | C6E—H6E | 0.9300 |
| C1C—C2C | 1.382 (4) | C11E—C12E | 1.5393 |
| C1C—C6C | 1.391 (4) | C11E—H11E | 0.9800 |
| C1C—C11C | 1.466 (4) | C12E—C13E | 1.5245 |
| C2C—O21C | 1.368 (4) | C12E—H12E | 0.9800 |
| C2C—C3C | 1.393 (5) | C13E—O12E | 1.2245 |
| C3C—C4C | 1.383 (5) | C13E—O11E | 1.3047 |
| C3C—H3C | 0.9300 | C21E—O21E | 1.4391 |
| C4C—C5C | 1.376 (5) | C21E—C22E | 1.5213 |
| C4C—H4C | 0.9300 | C21E—H21I | 0.9700 |
| C5C—C6C | 1.369 (5) | C21E—H21J | 0.9700 |
| C5C—H5C | 0.9300 | C22E—H22M | 0.9600 |
| C6C—H6C | 0.9300 | C22E—H22N | 0.9600 |
| C11C—C12C | 1.326 (4) | C22E—H22O | 0.9600 |
| C11C—H11C | 0.9300 | O11E—H1E | 0.8200 |
| | | |
| C6A—C1A—C2A | 117.5 (5) | O21C—C21C—C22C | 106.9 (3) |
| C6A—C1A—C11A | 122.2 (4) | O21C—C21C—H21A | 110.3 |
| C2A—C1A—C11A | 120.3 (4) | C22C—C21C—H21A | 110.3 |
| O21A—C2A—C3A | 124.0 (4) | O21C—C21C—H21B | 110.3 |
| O21A—C2A—C1A | 115.2 (4) | C22C—C21C—H21B | 110.3 |
| C3A—C2A—C1A | 120.9 (4) | H21A—C21C—H21B | 108.6 |
| C4A—C3A—C2A | 120.1 (4) | C21C—C22C—H22A | 109.5 |
| C4A—C3A—H3A | 120.0 | C21C—C22C—H22B | 109.5 |
| C2A—C3A—H3A | 120.0 | H22A—C22C—H22B | 109.5 |
| C3A—C4A—C5A | 120.2 (5) | C21C—C22C—H22C | 109.5 |
| C3A—C4A—H4A | 119.9 | H22A—C22C—H22C | 109.5 |
| C5A—C4A—H4A | 119.9 | H22B—C22C—H22C | 109.5 |
| C6A—C5A—C4A | 119.2 (5) | C13C—O11C—H1C | 109.5 |
| C6A—C5A—H5A | 120.4 | C2C—O21C—C21C | 119.0 (3) |
| C4A—C5A—H5A | 120.4 | C2D—C1D—C6D | 117.6 |
| C5A—C6A—C1A | 122.0 (5) | C2D—C1D—C11D | 116.5 |
| C5A—C6A—H6A | 119.0 | C6D—C1D—C11D | 125.3 |
| C1A—C6A—H6A | 119.0 | O21D—C2D—C1D | 115.0 |
| C12A—C11A—C1A | 127.3 (4) | O21D—C2D—C3D | 124.0 |
| C12A—C11A—H11A | 116.3 | C1D—C2D—C3D | 121.0 |
| C1A—C11A—H11A | 116.3 | C4D—C3D—C2D | 118.8 |
| C11A—C12A—C13A | 123.6 (4) | C4D—C3D—H3D | 120.6 |
| C11A—C12A—H12A | 118.2 | C2D—C3D—H3D | 120.6 |
| C13A—C12A—H12A | 118.2 | C5D—C4D—C3D | 121.6 |
| O11A—C13A—O12A | 124.0 (5) | C5D—C4D—H4D | 119.2 |
| O11A—C13A—C12A | 118.8 (4) | C3D—C4D—H4D | 119.2 |
| O12A—C13A—C12A | 117.2 (4) | C4D—C5D—C6D | 118.6 |
| O21A—C21A—C22A | 106.8 (4) | C4D—C5D—H5D | 120.7 |
| O21A—C21A—H21C | 110.4 | C6D—C5D—H5D | 120.7 |
| C22A—C21A—H21C | 110.4 | C5D—C6D—C1D | 122.5 |
| O21A—C21A—H21D | 110.4 | C5D—C6D—H6D | 118.7 |
| C22A—C21A—H21D | 110.4 | C1D—C6D—H6D | 118.7 |
| H21C—C21A—H21D | 108.6 | C1D—C11D—C12D | 119.2 |
| C21A—C22A—H22D | 109.5 | C1D—C11D—C12E | 116.5 |
| C21A—C22A—H22E | 109.5 | C12D—C11D—C12E | 89.6 |
| H22D—C22A—H22E | 109.5 | C1D—C11D—H11D | 110.0 |
| C21A—C22A—H22F | 109.5 | C12D—C11D—H11D | 110.0 |
| H22D—C22A—H22F | 109.5 | C12E—C11D—H11D | 110.0 |
| H22E—C22A—H22F | 109.5 | C13D—C12D—C11D | 114.6 |
| C13A—O12A—H1A | 109.5 | C13D—C12D—C11E | 112.4 |
| C2A—O21A—C21A | 118.8 (3) | C11D—C12D—C11E | 90.4 |
| C6B—C1B—C2B | 117.7 (4) | C13D—C12D—H12D | 112.6 |
| C6B—C1B—C11B | 121.8 (4) | C11D—C12D—H12D | 112.6 |
| C2B—C1B—C11B | 120.5 (3) | C11E—C12D—H12D | 112.6 |
| O21B—C2B—C3B | 123.8 (3) | O12D—C13D—O11D | 122.4 |
| O21B—C2B—C1B | 115.8 (3) | O12D—C13D—C12D | 124.0 |
| C3B—C2B—C1B | 120.4 (4) | O11D—C13D—C12D | 113.0 |
| C4B—C3B—C2B | 120.1 (4) | O21D—C21D—C22D | 107.7 |
| C4B—C3B—H3B | 119.9 | O21D—C21D—H21G | 110.2 |
| C2B—C3B—H3B | 119.9 | C22D—C21D—H21G | 110.2 |
| C5B—C4B—C3B | 120.4 (5) | O21D—C21D—H21H | 110.2 |
| C5B—C4B—H4B | 119.8 | C22D—C21D—H21H | 110.2 |
| C3B—C4B—H4B | 119.8 | H21G—C21D—H21H | 108.5 |
| C4B—C5B—C6B | 119.5 (4) | C21D—C22D—H22J | 109.5 |
| C4B—C5B—H5B | 120.2 | C21D—C22D—H22K | 109.5 |
| C6B—C5B—H5B | 120.2 | H22J—C22D—H22K | 109.5 |
| C1B—C6B—C5B | 121.8 (4) | C21D—C22D—H22L | 109.5 |
| C1B—C6B—H6B | 119.1 | H22J—C22D—H22L | 109.5 |
| C5B—C6B—H6B | 119.1 | H22K—C22D—H22L | 109.5 |
| C12B—C11B—C1B | 127.1 (4) | C13D—O11D—H1D | 109.5 |
| C12B—C11B—H11B | 116.4 | C2D—O21D—C21D | 118.0 |
| C1B—C11B—H11B | 116.4 | C2E—C1E—C6E | 118.5 |
| C11B—C12B—C13B | 122.9 (4) | C2E—C1E—C11E | 115.6 |
| C11B—C12B—H12B | 118.6 | C6E—C1E—C11E | 124.9 |
| C13B—C12B—H12B | 118.6 | C1E—C2E—O21E | 115.2 |
| O12B—C13B—O11B | 123.2 (4) | C1E—C2E—C3E | 120.7 |
| O12B—C13B—C12B | 120.9 (3) | O21E—C2E—C3E | 124.1 |
| O11B—C13B—C12B | 115.9 (4) | C4E—C3E—C2E | 119.0 |
| O21B—C21B—C22B | 107.1 (3) | C4E—C3E—H3E | 120.5 |
| O21B—C21B—H21E | 110.3 | C2E—C3E—H3E | 120.5 |
| C22B—C21B—H21E | 110.3 | C5E—C4E—C3E | 121.3 |
| O21B—C21B—H21F | 110.3 | C5E—C4E—H4E | 119.4 |
| C22B—C21B—H21F | 110.3 | C3E—C4E—H4E | 119.4 |
| H21E—C21B—H21F | 108.5 | C4E—C5E—C6E | 119.0 |
| C21B—C22B—H22G | 109.5 | C4E—C5E—H5E | 120.5 |
| C21B—C22B—H22H | 109.5 | C6E—C5E—H5E | 120.5 |
| H22G—C22B—H22H | 109.5 | C5E—C6E—C1E | 121.6 |
| C21B—C22B—H22I | 109.5 | C5E—C6E—H6E | 119.2 |
| H22G—C22B—H22I | 109.5 | C1E—C6E—H6E | 119.2 |
| H22H—C22B—H22I | 109.5 | C1E—C11E—C12E | 119.6 |
| C13B—O11B—H1B | 109.5 | C1E—C11E—C12D | 116.7 |
| C2B—O21B—C21B | 118.6 (3) | C12E—C11E—C12D | 89.9 |
| C2C—C1C—C6C | 118.7 (3) | C1E—C11E—H11E | 109.7 |
| C2C—C1C—C11C | 119.7 (3) | C12E—C11E—H11E | 109.7 |
| C6C—C1C—C11C | 121.5 (3) | C12D—C11E—H11E | 109.7 |
| O21C—C2C—C1C | 116.5 (3) | C13E—C12E—C11E | 115.4 |
| O21C—C2C—C3C | 122.9 (3) | C13E—C12E—C11D | 112.1 |
| C1C—C2C—C3C | 120.6 (3) | C11E—C12E—C11D | 90.1 |
| C4C—C3C—C2C | 119.3 (4) | C13E—C12E—H12E | 112.5 |
| C4C—C3C—H3C | 120.4 | C11E—C12E—H12E | 112.5 |
| C2C—C3C—H3C | 120.4 | C11D—C12E—H12E | 112.5 |
| C5C—C4C—C3C | 120.4 (4) | O12E—C13E—O11E | 123.8 |
| C5C—C4C—H4C | 119.8 | O12E—C13E—C12E | 123.0 |
| C3C—C4C—H4C | 119.8 | O11E—C13E—C12E | 112.5 |
| C6C—C5C—C4C | 120.0 (4) | O21E—C21E—C22E | 107.3 |
| C6C—C5C—H5C | 120.0 | O21E—C21E—H21I | 110.3 |
| C4C—C5C—H5C | 120.0 | C22E—C21E—H21I | 110.3 |
| C5C—C6C—C1C | 120.9 (4) | O21E—C21E—H21J | 110.3 |
| C5C—C6C—H6C | 119.5 | C22E—C21E—H21J | 110.3 |
| C1C—C6C—H6C | 119.5 | H21I—C21E—H21J | 108.5 |
| C12C—C11C—C1C | 127.6 (3) | C21E—C22E—H22M | 109.5 |
| C12C—C11C—H11C | 116.2 | C21E—C22E—H22N | 109.5 |
| C1C—C11C—H11C | 116.2 | H22M—C22E—H22N | 109.5 |
| C11C—C12C—C13C | 122.7 (3) | C21E—C22E—H22O | 109.5 |
| C11C—C12C—H12C | 118.7 | H22M—C22E—H22O | 109.5 |
| C13C—C12C—H12C | 118.7 | H22N—C22E—H22O | 109.5 |
| O12C—C13C—O11C | 123.0 (4) | C13E—O11E—H1E | 109.5 |
| O12C—C13C—C12C | 121.4 (3) | C2E—O21E—C21E | 117.6 |
| O11C—C13C—C12C | 115.6 (4) | | |
| | | |
| C6A—C1A—C2A—O21A | −179.4 (3) | C6D—C1D—C2D—C3D | −1.6 |
| C11A—C1A—C2A—O21A | −0.8 (5) | C11D—C1D—C2D—C3D | −172.7 |
| C6A—C1A—C2A—C3A | 1.1 (6) | O21D—C2D—C3D—C4D | −179.8 |
| C11A—C1A—C2A—C3A | 179.7 (4) | C1D—C2D—C3D—C4D | 1.7 |
| O21A—C2A—C3A—C4A | 179.5 (4) | C2D—C3D—C4D—C5D | −1.5 |
| C1A—C2A—C3A—C4A | −1.1 (6) | C3D—C4D—C5D—C6D | 1.1 |
| C2A—C3A—C4A—C5A | 1.5 (7) | C4D—C5D—C6D—C1D | −1.0 |
| C3A—C4A—C5A—C6A | −1.9 (7) | C2D—C1D—C6D—C5D | 1.3 |
| C4A—C5A—C6A—C1A | 2.0 (7) | C11D—C1D—C6D—C5D | 171.5 |
| C2A—C1A—C6A—C5A | −1.6 (6) | C2D—C1D—C11D—C12D | −176.6 |
| C11A—C1A—C6A—C5A | 179.9 (4) | C6D—C1D—C11D—C12D | 13.1 |
| C6A—C1A—C11A—C12A | 0.4 (6) | C2D—C1D—C11D—C12E | 77.8 |
| C2A—C1A—C11A—C12A | −178.1 (4) | C6D—C1D—C11D—C12E | −92.6 |
| C1A—C11A—C12A—C13A | 179.8 (4) | C1D—C11D—C12D—C13D | 125.2 |
| C11A—C12A—C13A—O11A | 180.0 (4) | C12E—C11D—C12D—C13D | −114.3 |
| C11A—C12A—C13A—O12A | −0.3 (6) | C1D—C11D—C12D—C11E | −119.8 |
| C3A—C2A—O21A—C21A | 1.5 (6) | C12E—C11D—C12D—C11E | 0.7 |
| C1A—C2A—O21A—C21A | −177.9 (3) | C11D—C12D—C13D—O12D | 31.8 |
| C22A—C21A—O21A—C2A | 175.8 (3) | C11E—C12D—C13D—O12D | −69.5 |
| C6B—C1B—C2B—O21B | 179.9 (3) | C11D—C12D—C13D—O11D | −157.0 |
| C11B—C1B—C2B—O21B | 1.9 (5) | C11E—C12D—C13D—O11D | 101.6 |
| C6B—C1B—C2B—C3B | −1.9 (5) | C1D—C2D—O21D—C21D | −169.1 |
| C11B—C1B—C2B—C3B | −180.0 (3) | C3D—C2D—O21D—C21D | 12.3 |
| O21B—C2B—C3B—C4B | 179.5 (4) | C22D—C21D—O21D—C2D | 174.3 |
| C1B—C2B—C3B—C4B | 1.5 (6) | C6E—C1E—C2E—O21E | 179.2 |
| C2B—C3B—C4B—C5B | −1.0 (7) | C11E—C1E—C2E—O21E | −11.3 |
| C3B—C4B—C5B—C6B | 0.9 (7) | C6E—C1E—C2E—C3E | 0.2 |
| C2B—C1B—C6B—C5B | 1.8 (6) | C11E—C1E—C2E—C3E | 169.8 |
| C11B—C1B—C6B—C5B | 179.8 (4) | C1E—C2E—C3E—C4E | 0.1 |
| C4B—C5B—C6B—C1B | −1.3 (7) | O21E—C2E—C3E—C4E | −178.7 |
| C6B—C1B—C11B—C12B | 8.8 (6) | C2E—C3E—C4E—C5E | 0.0 |
| C2B—C1B—C11B—C12B | −173.2 (4) | C3E—C4E—C5E—C6E | −0.5 |
| C1B—C11B—C12B—C13B | −179.7 (3) | C4E—C5E—C6E—C1E | 0.9 |
| C11B—C12B—C13B—O12B | 6.4 (6) | C2E—C1E—C6E—C5E | −0.7 |
| C11B—C12B—C13B—O11B | −173.8 (3) | C11E—C1E—C6E—C5E | −169.2 |
| C3B—C2B—O21B—C21B | −0.1 (5) | C2E—C1E—C11E—C12E | −176.1 |
| C1B—C2B—O21B—C21B | 178.0 (3) | C6E—C1E—C11E—C12E | −7.4 |
| C22B—C21B—O21B—C2B | −177.7 (3) | C2E—C1E—C11E—C12D | −69.7 |
| C6C—C1C—C2C—O21C | −180.0 (3) | C6E—C1E—C11E—C12D | 99.1 |
| C11C—C1C—C2C—O21C | −1.0 (4) | C13D—C12D—C11E—C1E | −7.3 |
| C6C—C1C—C2C—C3C | −0.4 (5) | C11D—C12D—C11E—C1E | −124.2 |
| C11C—C1C—C2C—C3C | 178.6 (3) | C13D—C12D—C11E—C12E | 116.2 |
| O21C—C2C—C3C—C4C | 179.5 (3) | C11D—C12D—C11E—C12E | −0.7 |
| C1C—C2C—C3C—C4C | −0.1 (6) | C1E—C11E—C12E—C13E | −123.6 |
| C2C—C3C—C4C—C5C | 0.7 (6) | C12D—C11E—C12E—C13E | 115.4 |
| C3C—C4C—C5C—C6C | −0.9 (6) | C1E—C11E—C12E—C11D | 121.7 |
| C4C—C5C—C6C—C1C | 0.5 (6) | C12D—C11E—C12E—C11D | 0.7 |
| C2C—C1C—C6C—C5C | 0.2 (5) | C1D—C11D—C12E—C13E | 4.5 |
| C11C—C1C—C6C—C5C | −178.8 (3) | C12D—C11D—C12E—C13E | −118.3 |
| C2C—C1C—C11C—C12C | −177.8 (3) | C1D—C11D—C12E—C11E | 122.1 |
| C6C—C1C—C11C—C12C | 1.1 (5) | C12D—C11D—C12E—C11E | −0.7 |
| C1C—C11C—C12C—C13C | 178.9 (3) | C11E—C12E—C13E—O12E | −23.8 |
| C11C—C12C—C13C—O12C | 5.2 (5) | C11D—C12E—C13E—O12E | 77.5 |
| C11C—C12C—C13C—O11C | −176.2 (3) | C11E—C12E—C13E—O11E | 165.0 |
| C1C—C2C—O21C—C21C | 175.8 (3) | C11D—C12E—C13E—O11E | −93.7 |
| C3C—C2C—O21C—C21C | −3.8 (5) | C1E—C2E—O21E—C21E | 165.9 |
| C22C—C21C—O21C—C2C | −174.0 (3) | C3E—C2E—O21E—C21E | −15.2 |
| C6D—C1D—C2D—O21D | 179.8 | C22E—C21E—O21E—C2E | −171.1 |
| C11D—C1D—C2D—O21D | 8.7 | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11C—H1C···O12Bi | 0.82 | 1.81 | 2.618 (4) | 166 |
| O11B—H1B···O12Ci | 0.82 | 1.79 | 2.593 (4) | 167 |
| O12A—H1A···O11Aii | 0.82 | 1.77 | 2.578 (6) | 170 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
(d6hlt_s) (
E)-3-(2-Ethoxyphenyl)propenoic acid and
trans-2,4-bis(2-ethoxyphenyl)-
trans-1,3-cyclobutane-dicarboxylic acid
top
Crystal data top
| 0.124(C22H24O6)·2.752(C11H12O3) | Z = 2 |
| Mr = 576.62 | F(000) = 612 |
| Triclinic, P1 | Dx = 1.283 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71069 Å |
| a = 8.6452 (13) Å | Cell parameters from 728 reflections |
| b = 10.8577 (17) Å | θ = 2.4–26.3° |
| c = 17.262 (3) Å | µ = 0.09 mm−1 |
| α = 92.470 (3)° | T = 173 K |
| β = 92.102 (3)° | Irregular, white |
| γ = 112.563 (3)° | 0.38 × 0.33 × 0.24 mm |
| V = 1492.4 (4) Å3 | |
Data collection top
Bruker SMART 1K
diffractometer | 3167 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.022 |
| Graphite monochromator | θmax = 25.5°, θmin = 1.2° |
| phi and ω scans | h = −10→8 |
| 8177 measured reflections | k = −13→13 |
| 5500 independent reflections | l = −20→20 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.053 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.136 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.03 | w = 1/[σ2(Fo2) + (0.0572P)2 + 0.0749P]
where P = (Fo2 + 2Fc2)/3 |
| 5500 reflections | (Δ/σ)max < 0.001 |
| 390 parameters | Δρmax = 0.14 e Å−3 |
| 0 restraints | Δρmin = −0.21 e Å−3 |
Crystal data top
| 0.124(C22H24O6)·2.752(C11H12O3) | γ = 112.563 (3)° |
| Mr = 576.62 | V = 1492.4 (4) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 8.6452 (13) Å | Mo Kα radiation |
| b = 10.8577 (17) Å | µ = 0.09 mm−1 |
| c = 17.262 (3) Å | T = 173 K |
| α = 92.470 (3)° | 0.38 × 0.33 × 0.24 mm |
| β = 92.102 (3)° | |
Data collection top
Bruker SMART 1K
diffractometer | 3167 reflections with I > 2σ(I) |
| 8177 measured reflections | Rint = 0.022 |
| 5500 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.053 | 0 restraints |
| wR(F2) = 0.136 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.03 | Δρmax = 0.14 e Å−3 |
| 5500 reflections | Δρmin = −0.21 e Å−3 |
| 390 parameters | |
Special details top
Experimental. The following alerts are triggered when using the ACTA-C CIF validation software: PLAT301_ALERT_3_B Main Residue Disorder ························. 40.00 Perc. PLAT432_ALERT_2_B Short Inter X···Y Contact C3C.. C4D.. 3.09 A ng. PLAT432_ALERT_2_B Short Inter X···Y Contact C4C.. C3D.. 3.07 A ng. PLAT165_ALERT_3_C Nr. of Status R Flagged Non-Hydrogen Atoms ···.. 28 PLAT432_ALERT_2_C Short Inter X···Y Contact C3C.. C3D.. 3.18 A ng. —— Authors comment: The assymetric unit consists of a mixture of the monomer and dimer. The short contacts are most probably due to the dimer not being properly optimized for the crystal structure at this point. Coordinates for the dimer molecule at a higher conversion stage was used as a starting point for the refinement. The dimer was also refined isotropically as a rigid body. This is necessary as the dimer is only present in about 4–5% occupancy (4% dimer occupancy = 8% of monomer consumed). PLAT042_ALERT_1_C Calc. and Rep. MoietyFormula Strings Differ ···. ? —— Authors commment: Our moiety formula is correct as it takes the occupancy of the dimer and monomer molecules into account. |
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| C1A | 1.0162 (3) | 0.0296 (2) | 0.22157 (15) | 0.0409 (7) | 0.876 (2) |
| C2A | 1.0094 (4) | 0.0241 (3) | 0.30238 (15) | 0.0419 (7) | 0.876 (2) |
| C3A | 1.1499 (4) | 0.0338 (3) | 0.34793 (17) | 0.0495 (8) | 0.876 (2) |
| H3A | 1.1454 | 0.0318 | 0.4028 | 0.059* | 0.876 (2) |
| C4A | 1.2960 (4) | 0.0462 (3) | 0.31332 (19) | 0.0562 (8) | 0.876 (2) |
| H4A | 1.3916 | 0.0519 | 0.3445 | 0.067* | 0.876 (2) |
| C5A | 1.3042 (4) | 0.0504 (3) | 0.2336 (2) | 0.0578 (8) | 0.876 (2) |
| H5A | 1.4053 | 0.0595 | 0.2100 | 0.069* | 0.876 (2) |
| C6A | 1.1662 (4) | 0.0414 (3) | 0.18871 (17) | 0.0511 (8) | 0.876 (2) |
| H6A | 1.1725 | 0.0432 | 0.1339 | 0.061* | 0.876 (2) |
| C11A | 0.8669 (3) | 0.0204 (2) | 0.17418 (14) | 0.0431 (7) | 0.876 (2) |
| H11A | 0.7711 | 0.0140 | 0.2016 | 0.052* | 0.876 (2) |
| C12A | 0.8511 (4) | 0.0198 (2) | 0.09700 (14) | 0.0458 (7) | 0.876 (2) |
| H12A | 0.9449 | 0.0270 | 0.0678 | 0.055* | 0.876 (2) |
| C13A | 0.6963 (4) | 0.0087 (2) | 0.05566 (15) | 0.0453 (7) | 0.876 (2) |
| C21A | 0.8459 (4) | 0.0094 (3) | 0.41466 (14) | 0.0461 (7) | 0.876 (2) |
| H21C | 0.9338 | 0.0898 | 0.4412 | 0.055* | 0.876 (2) |
| H21D | 0.8592 | −0.0708 | 0.4338 | 0.055* | 0.876 (2) |
| C22A | 0.6753 (4) | 0.0064 (3) | 0.43047 (16) | 0.0522 (8) | 0.876 (2) |
| H22D | 0.6661 | 0.0886 | 0.4139 | 0.078* | 0.876 (2) |
| H22E | 0.6586 | −0.0005 | 0.4862 | 0.078* | 0.876 (2) |
| H22F | 0.5897 | −0.0709 | 0.4016 | 0.078* | 0.876 (2) |
| O11A | 0.6924 (2) | 0.01017 (19) | −0.01746 (11) | 0.0515 (6) | 0.876 (2) |
| O12A | 0.5693 (3) | −0.0032 (2) | 0.09562 (12) | 0.0584 (6) | 0.876 (2) |
| H1A | 0.4880 | −0.0083 | 0.0657 | 0.088* | 0.876 (2) |
| O21A | 0.8599 (3) | 0.0121 (2) | 0.33185 (10) | 0.0461 (5) | 0.876 (2) |
| C1B | 0.7885 (3) | 0.3467 (2) | 0.12841 (14) | 0.0335 (6) | 0.876 (2) |
| C2B | 0.7885 (3) | 0.3357 (2) | 0.04688 (14) | 0.0335 (6) | 0.876 (2) |
| C3B | 0.6507 (3) | 0.3319 (3) | 0.00137 (16) | 0.0429 (7) | 0.876 (2) |
| H3B | 0.6512 | 0.3246 | −0.0537 | 0.051* | 0.876 (2) |
| C4B | 0.5132 (4) | 0.3388 (3) | 0.03635 (18) | 0.0509 (8) | 0.876 (2) |
| H4B | 0.4188 | 0.3355 | 0.0051 | 0.061* | 0.876 (2) |
| C5B | 0.5107 (4) | 0.3504 (3) | 0.11661 (19) | 0.0505 (8) | 0.876 (2) |
| H5B | 0.4157 | 0.3555 | 0.1403 | 0.061* | 0.876 (2) |
| C6B | 0.6481 (3) | 0.3544 (3) | 0.16136 (15) | 0.0417 (7) | 0.876 (2) |
| H6B | 0.6467 | 0.3626 | 0.2163 | 0.050* | 0.876 (2) |
| C11B | 0.9351 (3) | 0.3525 (2) | 0.17578 (13) | 0.0362 (6) | 0.876 (2) |
| H11B | 1.0318 | 0.3591 | 0.1489 | 0.043* | 0.876 (2) |
| C12B | 0.9469 (3) | 0.3493 (2) | 0.25290 (13) | 0.0408 (7) | 0.876 (2) |
| H12B | 0.8521 | 0.3422 | 0.2814 | 0.049* | 0.876 (2) |
| C13B | 1.1002 (4) | 0.3566 (2) | 0.29525 (14) | 0.0403 (6) | 0.876 (2) |
| C21B | 0.9331 (3) | 0.3141 (3) | −0.06611 (13) | 0.0354 (6) | 0.876 (2) |
| H21E | 0.9263 | 0.3928 | −0.0906 | 0.043* | 0.876 (2) |
| H21F | 0.8372 | 0.2333 | −0.0871 | 0.043* | 0.876 (2) |
| C22B | 1.0954 (4) | 0.3014 (3) | −0.08263 (15) | 0.0448 (7) | 0.876 (2) |
| H22G | 1.1891 | 0.3847 | −0.0654 | 0.067* | 0.876 (2) |
| H22H | 1.0987 | 0.2837 | −0.1386 | 0.067* | 0.876 (2) |
| H22I | 1.1045 | 0.2275 | −0.0548 | 0.067* | 0.876 (2) |
| O11B | 1.0898 (2) | 0.34100 (19) | 0.36854 (10) | 0.0481 (5) | 0.876 (2) |
| H1B | 1.1827 | 0.3460 | 0.3880 | 0.072* | 0.876 (2) |
| O12B | 1.2329 (3) | 0.3776 (2) | 0.26032 (11) | 0.0467 (5) | 0.876 (2) |
| O21B | 0.9293 (2) | 0.32927 (19) | 0.01674 (10) | 0.0383 (5) | 0.876 (2) |
| C1C | 0.2212 (3) | 0.6717 (2) | 0.42538 (12) | 0.0383 (5) | |
| C2C | 0.2336 (3) | 0.6865 (2) | 0.34524 (12) | 0.0387 (5) | |
| C3C | 0.1000 (3) | 0.6899 (2) | 0.29955 (14) | 0.0519 (6) | |
| H3C | 0.1096 | 0.7004 | 0.2454 | 0.062* | |
| C4C | −0.0476 (3) | 0.6779 (3) | 0.33352 (15) | 0.0573 (7) | |
| H4C | −0.1396 | 0.6791 | 0.3022 | 0.069* | |
| C5C | −0.0623 (3) | 0.6642 (2) | 0.41232 (15) | 0.0536 (7) | |
| H5C | −0.1636 | 0.6567 | 0.4352 | 0.064* | |
| C6C | 0.0706 (3) | 0.6614 (2) | 0.45758 (13) | 0.0467 (6) | |
| H6C | 0.0600 | 0.6522 | 0.5118 | 0.056* | |
| C11C | 0.3617 (3) | 0.66488 (19) | 0.47177 (12) | 0.0386 (5) | |
| H11C | 0.4601 | 0.6754 | 0.4454 | 0.046* | |
| C12C | 0.3664 (3) | 0.6453 (2) | 0.54770 (12) | 0.0444 (6) | |
| H12C | 0.2711 | 0.6359 | 0.5762 | 0.053* | |
| C13C | 0.5136 (3) | 0.6378 (2) | 0.58787 (12) | 0.0430 (6) | |
| C21C | 0.3994 (3) | 0.7040 (2) | 0.23407 (11) | 0.0406 (6) | |
| H21A | 0.3936 | 0.7882 | 0.2171 | 0.049* | |
| H21B | 0.3084 | 0.6279 | 0.2057 | 0.049* | |
| C22C | 0.5668 (3) | 0.6999 (2) | 0.21845 (12) | 0.0456 (6) | |
| H22A | 0.6551 | 0.7730 | 0.2491 | 0.068* | |
| H22B | 0.5862 | 0.7099 | 0.1631 | 0.068* | |
| H22C | 0.5686 | 0.6142 | 0.2330 | 0.068* | |
| O11C | 0.49716 (19) | 0.61046 (16) | 0.65985 (8) | 0.0557 (5) | |
| H1C | 0.5869 | 0.6081 | 0.6787 | 0.084* | |
| O12C | 0.6474 (2) | 0.65866 (16) | 0.55414 (8) | 0.0515 (4) | |
| O21C | 0.38280 (17) | 0.69618 (15) | 0.31622 (8) | 0.0435 (4) | |
| C1D | 1.0226 (10) | 0.0482 (7) | 0.2390 (4) | 0.0386 (17)* | 0.124 (2) |
| C2D | 1.0064 (12) | 0.0102 (8) | 0.3149 (4) | 0.0386 (17)* | 0.124 (2) |
| C3D | 1.1339 (14) | −0.0175 (10) | 0.3546 (5) | 0.0386 (17)* | 0.124 (2) |
| H3D | 1.1208 | −0.0458 | 0.4062 | 0.046* | 0.124 (2) |
| C4D | 1.2788 (14) | −0.0024 (12) | 0.3171 (6) | 0.0386 (17)* | 0.124 (2) |
| H4D | 1.3662 | −0.0188 | 0.3439 | 0.046* | 0.124 (2) |
| C5D | 1.2986 (13) | 0.0354 (12) | 0.2424 (6) | 0.0386 (17)* | 0.124 (2) |
| H5D | 1.3976 | 0.0438 | 0.2170 | 0.046* | 0.124 (2) |
| C6D | 1.1710 (11) | 0.0613 (9) | 0.2043 (5) | 0.0386 (17)* | 0.124 (2) |
| H6D | 1.1853 | 0.0890 | 0.1526 | 0.046* | 0.124 (2) |
| C11D | 0.8902 (8) | 0.0913 (5) | 0.2057 (4) | 0.0386 (17)* | 0.124 (2) |
| H11D | 0.7763 | 0.0248 | 0.2159 | 0.046* | 0.124 (2) |
| C12D | 0.8950 (8) | 0.1272 (6) | 0.1203 (3) | 0.0386 (17)* | 0.124 (2) |
| H12D | 0.9967 | 0.1256 | 0.0952 | 0.046* | 0.124 (2) |
| C13D | 0.7324 (10) | 0.0497 (7) | 0.0709 (4) | 0.0386 (17)* | 0.124 (2) |
| C21D | 0.8448 (16) | −0.0161 (12) | 0.4273 (5) | 0.0386 (17)* | 0.124 (2) |
| H21G | 0.9383 | 0.0568 | 0.4566 | 0.046* | 0.124 (2) |
| H21H | 0.8496 | −0.1024 | 0.4404 | 0.046* | 0.124 (2) |
| C22D | 0.6783 (16) | −0.0116 (15) | 0.4483 (5) | 0.0386 (17)* | 0.124 (2) |
| H22J | 0.6655 | −0.0219 | 0.5041 | 0.058* | 0.124 (2) |
| H22K | 0.5867 | −0.0843 | 0.4191 | 0.058* | 0.124 (2) |
| H22L | 0.6750 | 0.0744 | 0.4352 | 0.058* | 0.124 (2) |
| O11D | 0.7562 (13) | 0.0444 (9) | −0.0043 (4) | 0.0386 (17)* | 0.124 (2) |
| H11E | 0.6641 | 0.0012 | −0.0289 | 0.058* | 0.124 (2) |
| O12D | 0.5934 (9) | 0.0114 (8) | 0.0960 (5) | 0.0386 (17)* | 0.124 (2) |
| O21D | 0.8584 (12) | −0.0007 (9) | 0.3461 (4) | 0.0386 (17)* | 0.124 (2) |
| C1E | 0.7828 (11) | 0.3225 (7) | 0.1120 (5) | 0.0386 (17)* | 0.124 (2) |
| C2E | 0.7926 (13) | 0.3444 (8) | 0.0336 (5) | 0.0386 (17)* | 0.124 (2) |
| C3E | 0.6643 (15) | 0.3668 (11) | −0.0075 (6) | 0.0386 (17)* | 0.124 (2) |
| H3E | 0.6712 | 0.3817 | −0.0613 | 0.046* | 0.124 (2) |
| C4E | 0.5264 (15) | 0.3669 (13) | 0.0317 (7) | 0.0386 (17)* | 0.124 (2) |
| H4E | 0.4388 | 0.3820 | 0.0041 | 0.046* | 0.124 (2) |
| C5E | 0.5146 (14) | 0.3458 (13) | 0.1091 (7) | 0.0386 (17)* | 0.124 (2) |
| H5E | 0.4203 | 0.3468 | 0.1353 | 0.046* | 0.124 (2) |
| C6E | 0.6424 (12) | 0.3228 (10) | 0.1488 (5) | 0.0386 (17)* | 0.124 (2) |
| H6E | 0.6338 | 0.3068 | 0.2025 | 0.046* | 0.124 (2) |
| C11E | 0.9126 (9) | 0.2761 (6) | 0.1467 (4) | 0.0386 (17)* | 0.124 (2) |
| H11F | 1.0277 | 0.3412 | 0.1370 | 0.046* | 0.124 (2) |
| C12E | 0.9055 (9) | 0.2403 (6) | 0.2322 (4) | 0.0386 (17)* | 0.124 (2) |
| H12E | 0.8023 | 0.2408 | 0.2562 | 0.046* | 0.124 (2) |
| C13E | 1.0645 (11) | 0.3168 (7) | 0.2831 (4) | 0.0386 (17)* | 0.124 (2) |
| C21E | 0.9380 (16) | 0.3355 (12) | −0.0822 (4) | 0.0386 (17)* | 0.124 (2) |
| H21I | 0.9374 | 0.4185 | −0.1035 | 0.046* | 0.124 (2) |
| H21J | 0.8374 | 0.2594 | −0.1042 | 0.046* | 0.124 (2) |
| C22E | 1.0965 (16) | 0.3150 (15) | −0.1027 (5) | 0.0386 (17)* | 0.124 (2) |
| H22M | 1.1011 | 0.3084 | −0.1593 | 0.058* | 0.124 (2) |
| H22N | 1.0954 | 0.2325 | −0.0813 | 0.058* | 0.124 (2) |
| H22O | 1.1950 | 0.3910 | −0.0806 | 0.058* | 0.124 (2) |
| O11E | 1.0396 (14) | 0.3063 (9) | 0.3571 (4) | 0.0386 (17)* | 0.124 (2) |
| H11G | 1.1299 | 0.3497 | 0.3830 | 0.058* | 0.124 (2) |
| O12E | 1.2033 (10) | 0.3675 (7) | 0.2571 (5) | 0.0386 (17)* | 0.124 (2) |
| O21E | 0.9371 (13) | 0.3445 (9) | 0.0012 (4) | 0.0386 (17)* | 0.124 (2) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C1A | 0.0517 (17) | 0.0311 (14) | 0.0388 (16) | 0.0136 (13) | 0.0141 (13) | 0.0042 (11) |
| C2A | 0.0439 (17) | 0.0392 (15) | 0.0404 (17) | 0.0132 (13) | 0.0096 (13) | 0.0004 (12) |
| C3A | 0.0512 (19) | 0.0520 (19) | 0.0458 (17) | 0.0199 (16) | 0.0086 (14) | 0.0015 (14) |
| C4A | 0.0473 (19) | 0.054 (2) | 0.068 (2) | 0.0208 (16) | 0.0072 (15) | 0.0002 (16) |
| C5A | 0.053 (2) | 0.0555 (19) | 0.069 (2) | 0.0239 (16) | 0.0231 (16) | 0.0051 (16) |
| C6A | 0.065 (2) | 0.0455 (17) | 0.0446 (18) | 0.0222 (15) | 0.0219 (15) | 0.0031 (13) |
| C11A | 0.0577 (18) | 0.0315 (14) | 0.0387 (16) | 0.0146 (13) | 0.0153 (13) | 0.0028 (11) |
| C12A | 0.0614 (19) | 0.0382 (15) | 0.0389 (16) | 0.0193 (14) | 0.0142 (13) | 0.0051 (12) |
| C13A | 0.065 (2) | 0.0314 (15) | 0.0370 (17) | 0.0148 (14) | 0.0128 (14) | 0.0042 (12) |
| C21A | 0.0558 (18) | 0.0586 (19) | 0.0285 (16) | 0.0268 (15) | 0.0062 (12) | 0.0016 (12) |
| C22A | 0.0528 (18) | 0.069 (2) | 0.0363 (17) | 0.0246 (16) | 0.0078 (13) | 0.0028 (14) |
| O11A | 0.0632 (14) | 0.0536 (13) | 0.0315 (11) | 0.0145 (11) | 0.0119 (9) | 0.0058 (8) |
| O12A | 0.0741 (15) | 0.0621 (14) | 0.0405 (12) | 0.0258 (11) | 0.0200 (11) | 0.0091 (9) |
| O21A | 0.0435 (11) | 0.0631 (12) | 0.0301 (11) | 0.0187 (9) | 0.0065 (8) | 0.0013 (8) |
| C1B | 0.0376 (15) | 0.0333 (14) | 0.0308 (14) | 0.0146 (12) | 0.0040 (11) | 0.0036 (10) |
| C2B | 0.0347 (15) | 0.0327 (14) | 0.0339 (15) | 0.0134 (11) | 0.0047 (11) | 0.0035 (10) |
| C3B | 0.0403 (16) | 0.0552 (19) | 0.0349 (15) | 0.0209 (15) | −0.0031 (12) | 0.0022 (12) |
| C4B | 0.0364 (17) | 0.067 (2) | 0.0522 (19) | 0.0226 (16) | 0.0010 (13) | 0.0041 (15) |
| C5B | 0.0437 (17) | 0.0582 (18) | 0.0563 (19) | 0.0261 (14) | 0.0139 (14) | 0.0036 (14) |
| C6B | 0.0456 (17) | 0.0462 (17) | 0.0349 (15) | 0.0188 (14) | 0.0090 (12) | 0.0033 (12) |
| C11B | 0.0464 (16) | 0.0321 (14) | 0.0291 (14) | 0.0136 (12) | 0.0059 (11) | 0.0044 (10) |
| C12B | 0.0489 (17) | 0.0446 (16) | 0.0298 (15) | 0.0188 (13) | 0.0041 (12) | 0.0022 (11) |
| C13B | 0.0595 (19) | 0.0349 (15) | 0.0288 (14) | 0.0202 (14) | 0.0059 (13) | 0.0043 (11) |
| C21B | 0.0369 (15) | 0.0435 (16) | 0.0246 (14) | 0.0140 (12) | 0.0024 (10) | 0.0017 (11) |
| C22B | 0.0479 (17) | 0.0572 (18) | 0.0341 (16) | 0.0249 (14) | 0.0089 (13) | 0.0044 (13) |
| O11B | 0.0549 (14) | 0.0664 (13) | 0.0278 (10) | 0.0282 (11) | 0.0041 (8) | 0.0055 (9) |
| O12B | 0.0518 (13) | 0.0585 (12) | 0.0290 (10) | 0.0190 (10) | 0.0096 (9) | 0.0092 (8) |
| O21B | 0.0377 (10) | 0.0567 (12) | 0.0246 (10) | 0.0226 (9) | 0.0037 (8) | 0.0023 (8) |
| C1C | 0.0407 (14) | 0.0386 (13) | 0.0344 (13) | 0.0136 (11) | 0.0082 (10) | −0.0003 (10) |
| C2C | 0.0339 (13) | 0.0437 (14) | 0.0367 (13) | 0.0131 (11) | 0.0045 (10) | −0.0010 (10) |
| C3C | 0.0440 (15) | 0.0720 (17) | 0.0411 (14) | 0.0241 (13) | 0.0036 (11) | 0.0029 (12) |
| C4C | 0.0409 (15) | 0.0758 (19) | 0.0599 (18) | 0.0278 (14) | 0.0044 (12) | 0.0022 (14) |
| C5C | 0.0447 (16) | 0.0603 (17) | 0.0607 (17) | 0.0243 (13) | 0.0187 (13) | 0.0053 (13) |
| C6C | 0.0515 (16) | 0.0478 (14) | 0.0418 (14) | 0.0189 (12) | 0.0153 (12) | 0.0049 (11) |
| C11C | 0.0436 (14) | 0.0365 (13) | 0.0343 (13) | 0.0136 (11) | 0.0079 (10) | 0.0021 (9) |
| C12C | 0.0514 (15) | 0.0490 (14) | 0.0334 (13) | 0.0189 (12) | 0.0123 (11) | 0.0060 (10) |
| C13C | 0.0612 (17) | 0.0415 (13) | 0.0255 (12) | 0.0190 (12) | 0.0049 (11) | 0.0016 (10) |
| C21C | 0.0405 (14) | 0.0543 (14) | 0.0280 (12) | 0.0190 (11) | 0.0028 (9) | 0.0048 (10) |
| C22C | 0.0437 (14) | 0.0591 (15) | 0.0355 (13) | 0.0207 (12) | 0.0070 (10) | 0.0048 (11) |
| O11C | 0.0534 (11) | 0.0795 (12) | 0.0327 (9) | 0.0229 (9) | 0.0045 (7) | 0.0121 (8) |
| O12C | 0.0570 (11) | 0.0668 (11) | 0.0339 (9) | 0.0264 (9) | 0.0128 (8) | 0.0037 (7) |
| O21C | 0.0369 (9) | 0.0689 (11) | 0.0267 (8) | 0.0227 (8) | 0.0026 (6) | 0.0021 (7) |
Geometric parameters (Å, º) top
| C1A—C6A | 1.397 (4) | C12C—C13C | 1.458 (3) |
| C1A—C2A | 1.401 (3) | C12C—H12C | 0.9500 |
| C1A—C11A | 1.470 (4) | C13C—O12C | 1.260 (3) |
| C2A—O21A | 1.368 (3) | C13C—O11C | 1.290 (2) |
| C2A—C3A | 1.389 (4) | C21C—O21C | 1.433 (2) |
| C3A—C4A | 1.379 (4) | C21C—C22C | 1.499 (3) |
| C3A—H3A | 0.9500 | C21C—H21A | 0.9900 |
| C4A—C5A | 1.383 (4) | C21C—H21B | 0.9900 |
| C4A—H4A | 0.9500 | C22C—H22A | 0.9800 |
| C5A—C6A | 1.366 (4) | C22C—H22B | 0.9800 |
| C5A—H5A | 0.9500 | C22C—H22C | 0.9800 |
| C6A—H6A | 0.9500 | O11C—H1C | 0.8400 |
| C11A—C12A | 1.334 (3) | C1D—C2D | 1.3866 |
| C11A—H11A | 0.9500 | C1D—C6D | 1.3961 |
| C12A—C13A | 1.454 (4) | C1D—C11D | 1.4979 |
| C12A—H12A | 0.9500 | C2D—O21D | 1.3716 |
| C13A—O11A | 1.262 (3) | C2D—C3D | 1.4116 |
| C13A—O12A | 1.285 (3) | C3D—C4D | 1.3884 |
| C21A—O21A | 1.440 (3) | C3D—H3D | 0.9500 |
| C21A—C22A | 1.497 (3) | C4D—C5D | 1.3674 |
| C21A—H21C | 0.9900 | C4D—H4D | 0.9500 |
| C21A—H21D | 0.9900 | C5D—C6D | 1.3890 |
| C22A—H22D | 0.9800 | C5D—H5D | 0.9500 |
| C22A—H22E | 0.9800 | C6D—H6D | 0.9500 |
| C22A—H22F | 0.9800 | C11D—C12D | 1.5394 |
| O12A—H1A | 0.8400 | C11D—C12E | 1.6166 |
| C1B—C6B | 1.389 (3) | C11D—H11D | 1.0000 |
| C1B—C2B | 1.407 (3) | C12D—C13D | 1.5337 |
| C1B—C11B | 1.462 (3) | C12D—C11E | 1.6084 |
| C2B—O21B | 1.366 (3) | C12D—H12D | 1.0000 |
| C2B—C3B | 1.389 (3) | C13D—O12D | 1.2148 |
| C3B—C4B | 1.379 (3) | C13D—O11D | 1.3234 |
| C3B—H3B | 0.9500 | C21D—O21D | 1.4229 |
| C4B—C5B | 1.387 (4) | C21D—C22D | 1.5151 |
| C4B—H4B | 0.9500 | C21D—H21G | 0.9900 |
| C5B—C6B | 1.378 (4) | C21D—H21H | 0.9900 |
| C5B—H5B | 0.9500 | C22D—H22J | 0.9800 |
| C6B—H6B | 0.9500 | C22D—H22K | 0.9800 |
| C11B—C12B | 1.334 (3) | C22D—H22L | 0.9800 |
| C11B—H11B | 0.9500 | O11D—H11E | 0.8400 |
| C12B—C13B | 1.463 (3) | C1E—C2E | 1.3829 |
| C12B—H12B | 0.9500 | C1E—C6E | 1.3919 |
| C13B—O12B | 1.262 (3) | C1E—C11E | 1.5118 |
| C13B—O11B | 1.285 (3) | C2E—O21E | 1.3878 |
| C21B—O21B | 1.436 (3) | C2E—C3E | 1.3985 |
| C21B—C22B | 1.497 (3) | C3E—C4E | 1.3933 |
| C21B—H21E | 0.9900 | C3E—H3E | 0.9500 |
| C21B—H21F | 0.9900 | C4E—C5E | 1.3664 |
| C22B—H22G | 0.9800 | C4E—H4E | 0.9500 |
| C22B—H22H | 0.9800 | C5E—C6E | 1.3865 |
| C22B—H22I | 0.9800 | C5E—H5E | 0.9500 |
| O11B—H1B | 0.8400 | C6E—H6E | 0.9500 |
| C1C—C6C | 1.401 (3) | C11E—C12E | 1.5393 |
| C1C—C2C | 1.403 (3) | C11E—H11F | 1.0000 |
| C1C—C11C | 1.458 (3) | C12E—C13E | 1.5245 |
| C2C—O21C | 1.369 (2) | C12E—H12E | 1.0000 |
| C2C—C3C | 1.388 (3) | C13E—O12E | 1.2245 |
| C3C—C4C | 1.387 (3) | C13E—O11E | 1.3047 |
| C3C—H3C | 0.9500 | C21E—O21E | 1.4391 |
| C4C—C5C | 1.380 (3) | C21E—C22E | 1.5213 |
| C4C—H4C | 0.9500 | C21E—H21I | 0.9900 |
| C5C—C6C | 1.377 (3) | C21E—H21J | 0.9900 |
| C5C—H5C | 0.9500 | C22E—H22M | 0.9800 |
| C6C—H6C | 0.9500 | C22E—H22N | 0.9800 |
| C11C—C12C | 1.339 (3) | C22E—H22O | 0.9800 |
| C11C—H11C | 0.9500 | O11E—H11G | 0.8400 |
| | | |
| C6A—C1A—C2A | 118.0 (3) | O21C—C21C—C22C | 106.68 (16) |
| C6A—C1A—C11A | 122.2 (2) | O21C—C21C—H21A | 110.4 |
| C2A—C1A—C11A | 119.7 (2) | C22C—C21C—H21A | 110.4 |
| O21A—C2A—C3A | 123.7 (2) | O21C—C21C—H21B | 110.4 |
| O21A—C2A—C1A | 115.9 (2) | C22C—C21C—H21B | 110.4 |
| C3A—C2A—C1A | 120.4 (3) | H21A—C21C—H21B | 108.6 |
| C4A—C3A—C2A | 119.8 (3) | C21C—C22C—H22A | 109.5 |
| C4A—C3A—H3A | 120.1 | C21C—C22C—H22B | 109.5 |
| C2A—C3A—H3A | 120.1 | H22A—C22C—H22B | 109.5 |
| C3A—C4A—C5A | 120.5 (3) | C21C—C22C—H22C | 109.5 |
| C3A—C4A—H4A | 119.8 | H22A—C22C—H22C | 109.5 |
| C5A—C4A—H4A | 119.8 | H22B—C22C—H22C | 109.5 |
| C6A—C5A—C4A | 119.8 (3) | C13C—O11C—H1C | 109.5 |
| C6A—C5A—H5A | 120.1 | C2C—O21C—C21C | 118.45 (15) |
| C4A—C5A—H5A | 120.1 | C2D—C1D—C6D | 117.6 |
| C5A—C6A—C1A | 121.5 (3) | C2D—C1D—C11D | 116.5 |
| C5A—C6A—H6A | 119.2 | C6D—C1D—C11D | 125.3 |
| C1A—C6A—H6A | 119.2 | O21D—C2D—C1D | 115.0 |
| C12A—C11A—C1A | 127.2 (3) | O21D—C2D—C3D | 124.0 |
| C12A—C11A—H11A | 116.4 | C1D—C2D—C3D | 121.0 |
| C1A—C11A—H11A | 116.4 | C4D—C3D—C2D | 118.8 |
| C11A—C12A—C13A | 122.7 (3) | C4D—C3D—H3D | 120.6 |
| C11A—C12A—H12A | 118.6 | C2D—C3D—H3D | 120.6 |
| C13A—C12A—H12A | 118.6 | C5D—C4D—C3D | 121.6 |
| O11A—C13A—O12A | 123.2 (3) | C5D—C4D—H4D | 119.2 |
| O11A—C13A—C12A | 118.7 (2) | C3D—C4D—H4D | 119.2 |
| O12A—C13A—C12A | 118.1 (2) | C4D—C5D—C6D | 118.6 |
| O21A—C21A—C22A | 107.1 (2) | C4D—C5D—H5D | 120.7 |
| O21A—C21A—H21C | 110.3 | C6D—C5D—H5D | 120.7 |
| C22A—C21A—H21C | 110.3 | C5D—C6D—C1D | 122.5 |
| O21A—C21A—H21D | 110.3 | C5D—C6D—H6D | 118.7 |
| C22A—C21A—H21D | 110.3 | C1D—C6D—H6D | 118.7 |
| H21C—C21A—H21D | 108.6 | C1D—C11D—C12D | 119.2 |
| C21A—C22A—H22D | 109.5 | C1D—C11D—C12E | 116.5 |
| C21A—C22A—H22E | 109.5 | C12D—C11D—C12E | 89.6 |
| H22D—C22A—H22E | 109.5 | C1D—C11D—H11D | 110.0 |
| C21A—C22A—H22F | 109.5 | C12D—C11D—H11D | 110.0 |
| H22D—C22A—H22F | 109.5 | C12E—C11D—H11D | 110.0 |
| H22E—C22A—H22F | 109.5 | C13D—C12D—C11D | 114.6 |
| C13A—O12A—H1A | 109.5 | C13D—C12D—C11E | 112.4 |
| C2A—O21A—C21A | 118.73 (19) | C11D—C12D—C11E | 90.4 |
| C6B—C1B—C2B | 118.1 (2) | C13D—C12D—H12D | 112.6 |
| C6B—C1B—C11B | 122.0 (2) | C11D—C12D—H12D | 112.6 |
| C2B—C1B—C11B | 119.9 (2) | C11E—C12D—H12D | 112.6 |
| O21B—C2B—C3B | 123.3 (2) | O12D—C13D—O11D | 122.4 |
| O21B—C2B—C1B | 116.3 (2) | O12D—C13D—C12D | 124.0 |
| C3B—C2B—C1B | 120.4 (2) | O11D—C13D—C12D | 113.0 |
| C4B—C3B—C2B | 119.7 (2) | O21D—C21D—C22D | 107.7 |
| C4B—C3B—H3B | 120.1 | O21D—C21D—H21G | 110.2 |
| C2B—C3B—H3B | 120.1 | C22D—C21D—H21G | 110.2 |
| C3B—C4B—C5B | 120.9 (2) | O21D—C21D—H21H | 110.2 |
| C3B—C4B—H4B | 119.5 | C22D—C21D—H21H | 110.2 |
| C5B—C4B—H4B | 119.5 | H21G—C21D—H21H | 108.5 |
| C6B—C5B—C4B | 119.0 (2) | C21D—C22D—H22J | 109.5 |
| C6B—C5B—H5B | 120.5 | C21D—C22D—H22K | 109.5 |
| C4B—C5B—H5B | 120.5 | H22J—C22D—H22K | 109.5 |
| C5B—C6B—C1B | 121.9 (2) | C21D—C22D—H22L | 109.5 |
| C5B—C6B—H6B | 119.1 | H22J—C22D—H22L | 109.5 |
| C1B—C6B—H6B | 119.1 | H22K—C22D—H22L | 109.5 |
| C12B—C11B—C1B | 126.5 (2) | C13D—O11D—H11E | 109.5 |
| C12B—C11B—H11B | 116.7 | C2D—O21D—C21D | 118.0 |
| C1B—C11B—H11B | 116.7 | C2E—C1E—C6E | 118.5 |
| C11B—C12B—C13B | 122.5 (2) | C2E—C1E—C11E | 115.6 |
| C11B—C12B—H12B | 118.8 | C6E—C1E—C11E | 124.9 |
| C13B—C12B—H12B | 118.8 | C1E—C2E—O21E | 115.2 |
| O12B—C13B—O11B | 123.4 (2) | C1E—C2E—C3E | 120.7 |
| O12B—C13B—C12B | 120.6 (2) | O21E—C2E—C3E | 124.1 |
| O11B—C13B—C12B | 115.9 (2) | C4E—C3E—C2E | 119.0 |
| O21B—C21B—C22B | 107.09 (18) | C4E—C3E—H3E | 120.5 |
| O21B—C21B—H21E | 110.3 | C2E—C3E—H3E | 120.5 |
| C22B—C21B—H21E | 110.3 | C5E—C4E—C3E | 121.3 |
| O21B—C21B—H21F | 110.3 | C5E—C4E—H4E | 119.4 |
| C22B—C21B—H21F | 110.3 | C3E—C4E—H4E | 119.4 |
| H21E—C21B—H21F | 108.6 | C4E—C5E—C6E | 119.0 |
| C21B—C22B—H22G | 109.5 | C4E—C5E—H5E | 120.5 |
| C21B—C22B—H22H | 109.5 | C6E—C5E—H5E | 120.5 |
| H22G—C22B—H22H | 109.5 | C5E—C6E—C1E | 121.6 |
| C21B—C22B—H22I | 109.5 | C5E—C6E—H6E | 119.2 |
| H22G—C22B—H22I | 109.5 | C1E—C6E—H6E | 119.2 |
| H22H—C22B—H22I | 109.5 | C1E—C11E—C12E | 119.6 |
| C13B—O11B—H1B | 109.5 | C1E—C11E—C12D | 116.7 |
| C2B—O21B—C21B | 118.34 (17) | C12E—C11E—C12D | 89.9 |
| C6C—C1C—C2C | 117.9 (2) | C1E—C11E—H11F | 109.7 |
| C6C—C1C—C11C | 122.4 (2) | C12E—C11E—H11F | 109.7 |
| C2C—C1C—C11C | 119.69 (19) | C12D—C11E—H11F | 109.7 |
| O21C—C2C—C3C | 123.3 (2) | C13E—C12E—C11E | 115.4 |
| O21C—C2C—C1C | 115.99 (18) | C13E—C12E—C11D | 112.1 |
| C3C—C2C—C1C | 120.8 (2) | C11E—C12E—C11D | 90.1 |
| C2C—C3C—C4C | 119.6 (2) | C13E—C12E—H12E | 112.5 |
| C2C—C3C—H3C | 120.2 | C11E—C12E—H12E | 112.5 |
| C4C—C3C—H3C | 120.2 | C11D—C12E—H12E | 112.5 |
| C5C—C4C—C3C | 120.7 (2) | O12E—C13E—O11E | 123.8 |
| C5C—C4C—H4C | 119.6 | O12E—C13E—C12E | 123.0 |
| C3C—C4C—H4C | 119.6 | O11E—C13E—C12E | 112.5 |
| C6C—C5C—C4C | 119.6 (2) | O21E—C21E—C22E | 107.3 |
| C6C—C5C—H5C | 120.2 | O21E—C21E—H21I | 110.3 |
| C4C—C5C—H5C | 120.2 | C22E—C21E—H21I | 110.3 |
| C5C—C6C—C1C | 121.4 (2) | O21E—C21E—H21J | 110.3 |
| C5C—C6C—H6C | 119.3 | C22E—C21E—H21J | 110.3 |
| C1C—C6C—H6C | 119.3 | H21I—C21E—H21J | 108.5 |
| C12C—C11C—C1C | 126.7 (2) | C21E—C22E—H22M | 109.5 |
| C12C—C11C—H11C | 116.6 | C21E—C22E—H22N | 109.5 |
| C1C—C11C—H11C | 116.6 | H22M—C22E—H22N | 109.5 |
| C11C—C12C—C13C | 121.9 (2) | C21E—C22E—H22O | 109.5 |
| C11C—C12C—H12C | 119.0 | H22M—C22E—H22O | 109.5 |
| C13C—C12C—H12C | 119.0 | H22N—C22E—H22O | 109.5 |
| O12C—C13C—O11C | 123.0 (2) | C13E—O11E—H11G | 109.5 |
| O12C—C13C—C12C | 121.7 (2) | C2E—O21E—C21E | 117.6 |
| O11C—C13C—C12C | 115.3 (2) | | |
| | | |
| C6A—C1A—C2A—O21A | −179.7 (2) | C6D—C1D—C2D—C3D | −1.6 |
| C11A—C1A—C2A—O21A | −1.0 (3) | C11D—C1D—C2D—C3D | −172.7 |
| C6A—C1A—C2A—C3A | 1.7 (4) | O21D—C2D—C3D—C4D | −179.8 |
| C11A—C1A—C2A—C3A | −179.5 (2) | C1D—C2D—C3D—C4D | 1.7 |
| O21A—C2A—C3A—C4A | −179.7 (2) | C2D—C3D—C4D—C5D | −1.5 |
| C1A—C2A—C3A—C4A | −1.3 (4) | C3D—C4D—C5D—C6D | 1.1 |
| C2A—C3A—C4A—C5A | 0.6 (4) | C4D—C5D—C6D—C1D | −1.0 |
| C3A—C4A—C5A—C6A | −0.4 (4) | C2D—C1D—C6D—C5D | 1.3 |
| C4A—C5A—C6A—C1A | 0.8 (4) | C11D—C1D—C6D—C5D | 171.5 |
| C2A—C1A—C6A—C5A | −1.5 (4) | C2D—C1D—C11D—C12D | −176.6 |
| C11A—C1A—C6A—C5A | 179.8 (2) | C6D—C1D—C11D—C12D | 13.1 |
| C6A—C1A—C11A—C12A | 1.0 (4) | C2D—C1D—C11D—C12E | 77.8 |
| C2A—C1A—C11A—C12A | −177.8 (2) | C6D—C1D—C11D—C12E | −92.6 |
| C1A—C11A—C12A—C13A | 179.3 (2) | C1D—C11D—C12D—C13D | 125.2 |
| C11A—C12A—C13A—O11A | 179.4 (2) | C12E—C11D—C12D—C13D | −114.3 |
| C11A—C12A—C13A—O12A | −1.2 (4) | C1D—C11D—C12D—C11E | −119.8 |
| C3A—C2A—O21A—C21A | 0.1 (4) | C12E—C11D—C12D—C11E | 0.7 |
| C1A—C2A—O21A—C21A | −178.4 (2) | C11D—C12D—C13D—O12D | 31.8 |
| C22A—C21A—O21A—C2A | 175.7 (2) | C11E—C12D—C13D—O12D | −69.5 |
| C6B—C1B—C2B—O21B | 179.6 (2) | C11D—C12D—C13D—O11D | −157.0 |
| C11B—C1B—C2B—O21B | 0.8 (3) | C11E—C12D—C13D—O11D | 101.6 |
| C6B—C1B—C2B—C3B | −0.4 (3) | C1D—C2D—O21D—C21D | −169.1 |
| C11B—C1B—C2B—C3B | −179.2 (2) | C3D—C2D—O21D—C21D | 12.3 |
| O21B—C2B—C3B—C4B | 179.9 (2) | C22D—C21D—O21D—C2D | 174.3 |
| C1B—C2B—C3B—C4B | −0.1 (4) | C6E—C1E—C2E—O21E | 179.2 |
| C2B—C3B—C4B—C5B | 0.5 (4) | C11E—C1E—C2E—O21E | −11.3 |
| C3B—C4B—C5B—C6B | −0.3 (4) | C6E—C1E—C2E—C3E | 0.2 |
| C4B—C5B—C6B—C1B | −0.2 (4) | C11E—C1E—C2E—C3E | 169.8 |
| C2B—C1B—C6B—C5B | 0.6 (4) | C1E—C2E—C3E—C4E | 0.1 |
| C11B—C1B—C6B—C5B | 179.3 (2) | O21E—C2E—C3E—C4E | −178.7 |
| C6B—C1B—C11B—C12B | 8.5 (4) | C2E—C3E—C4E—C5E | 0.0 |
| C2B—C1B—C11B—C12B | −172.8 (2) | C3E—C4E—C5E—C6E | −0.5 |
| C1B—C11B—C12B—C13B | −179.6 (2) | C4E—C5E—C6E—C1E | 0.9 |
| C11B—C12B—C13B—O12B | 6.0 (4) | C2E—C1E—C6E—C5E | −0.7 |
| C11B—C12B—C13B—O11B | −174.2 (2) | C11E—C1E—C6E—C5E | −169.2 |
| C3B—C2B—O21B—C21B | −1.8 (3) | C2E—C1E—C11E—C12E | −176.1 |
| C1B—C2B—O21B—C21B | 178.21 (19) | C6E—C1E—C11E—C12E | −7.4 |
| C22B—C21B—O21B—C2B | −176.56 (19) | C2E—C1E—C11E—C12D | −69.7 |
| C6C—C1C—C2C—O21C | −179.96 (19) | C6E—C1E—C11E—C12D | 99.1 |
| C11C—C1C—C2C—O21C | −1.2 (3) | C13D—C12D—C11E—C1E | −7.3 |
| C6C—C1C—C2C—C3C | −0.4 (3) | C11D—C12D—C11E—C1E | −124.2 |
| C11C—C1C—C2C—C3C | 178.4 (2) | C13D—C12D—C11E—C12E | 116.2 |
| O21C—C2C—C3C—C4C | 179.3 (2) | C11D—C12D—C11E—C12E | −0.7 |
| C1C—C2C—C3C—C4C | −0.3 (3) | C1E—C11E—C12E—C13E | −123.6 |
| C2C—C3C—C4C—C5C | 0.7 (4) | C12D—C11E—C12E—C13E | 115.4 |
| C3C—C4C—C5C—C6C | −0.5 (4) | C1E—C11E—C12E—C11D | 121.7 |
| C4C—C5C—C6C—C1C | −0.2 (4) | C12D—C11E—C12E—C11D | 0.7 |
| C2C—C1C—C6C—C5C | 0.6 (3) | C1D—C11D—C12E—C13E | 4.5 |
| C11C—C1C—C6C—C5C | −178.1 (2) | C12D—C11D—C12E—C13E | −118.3 |
| C6C—C1C—C11C—C12C | 1.7 (3) | C1D—C11D—C12E—C11E | 122.1 |
| C2C—C1C—C11C—C12C | −176.9 (2) | C12D—C11D—C12E—C11E | −0.7 |
| C1C—C11C—C12C—C13C | 178.88 (19) | C11E—C12E—C13E—O12E | −23.8 |
| C11C—C12C—C13C—O12C | 4.9 (3) | C11D—C12E—C13E—O12E | 77.5 |
| C11C—C12C—C13C—O11C | −175.92 (19) | C11E—C12E—C13E—O11E | 165.0 |
| C3C—C2C—O21C—C21C | −3.0 (3) | C11D—C12E—C13E—O11E | −93.7 |
| C1C—C2C—O21C—C21C | 176.63 (17) | C1E—C2E—O21E—C21E | 165.9 |
| C22C—C21C—O21C—C2C | −174.50 (18) | C3E—C2E—O21E—C21E | −15.2 |
| C6D—C1D—C2D—O21D | 179.8 | C22E—C21E—O21E—C2E | −171.1 |
| C11D—C1D—C2D—O21D | 8.7 | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O12A—H1A···O11Ai | 0.84 | 1.73 | 2.566 (3) | 170 |
| O11B—H1B···O12Cii | 0.84 | 1.76 | 2.591 (2) | 168 |
| O11C—H1C···O12Bii | 0.84 | 1.80 | 2.622 (3) | 166 |
| Symmetry codes: (i) −x+1, −y, −z; (ii) −x+2, −y+1, −z+1. |
Experimental details
| | (almer60) | (almer80) | (d6h_s) | (d6hlt_s) |
| Crystal data |
| Chemical formula | C11H12O3 | C11H12O3 | 2.738(C11H12O3)·0.131(C22H24O6) | 0.124(C22H24O6)·2.752(C11H12O3) |
| Mr | 192.21 | 192.21 | 576.62 | 576.62 |
| Crystal system, space group | Triclinic, P1 | Triclinic, P1 | Triclinic, P1 | Triclinic, P1 |
| Temperature (K) | 345 | 375 | 293 | 173 |
| a, b, c (Å) | 8.7336 (4), 10.9590 (7), 17.2709 (10) | 8.7373 (5), 11.0167 (8), 17.295 (1) | 8.6759 (11), 11.0003 (13), 17.299 (2) | 8.6452 (13), 10.8577 (17), 17.262 (3) |
| α, β, γ (°) | 91.232 (4), 92.703 (4), 109.472 (4) | 91.187 (4), 92.775 (5), 108.938 (4) | 92.552 (3), 92.347 (2), 111.155 (3) | 92.470 (3), 92.102 (3), 112.563 (3) |
| V (Å3) | 1555.53 (15) | 1571.62 (17) | 1535.3 (3) | 1492.4 (4) |
| Z | 6 | 6 | 2 | 2 |
| Radiation type | Mo Kα | Mo Kα | Mo Kα | Mo Kα |
| µ (mm−1) | 0.09 | 0.09 | 0.09 | 0.09 |
| Crystal size (mm) | 1.00 × 0.34 × 0.31 | 1.00 × 0.34 × 0.31 | 0.38 × 0.33 × 0.26 | 0.38 × 0.33 × 0.24 |
| |
| Data collection |
| Diffractometer | Bruker SMART 1K
diffractometer | Bruker SMART 1K
diffractometer | Bruker SMART 1K
diffractometer | Bruker SMART 1K
diffractometer |
| Absorption correction | – | – | – | – |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 4937, 4937, 2689 | 4947, 4947, 1906 | 8251, 5391, 2192 | 8177, 5500, 3167 |
| Rint | 0.000 | 0.000 | 0.028 | 0.022 |
| (sin θ/λ)max (Å−1) | 0.595 | 0.595 | 0.595 | 0.606 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.049, 0.152, 1.02 | 0.049, 0.157, 0.88 | 0.068, 0.220, 0.90 | 0.053, 0.136, 1.03 |
| No. of reflections | 4937 | 4947 | 5391 | 5500 |
| No. of parameters | 386 | 386 | 391 | 390 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement | H atoms treated by a mixture of independent and constrained refinement | H atoms treated by a mixture of independent and constrained refinement | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 0.16, −0.12 | 0.15, −0.11 | 0.19, −0.18 | 0.14, −0.21 |
Hydrogen-bond geometry (Å, º) for (almer60) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11C—H1C···O12Bi | 1.23 | 1.41 | 2.642 (2) | 178.4 |
| O11B—H1B···O12Ci | 1.24 | 1.40 | 2.635 (2) | 175.9 |
| O12A—H1A···O11Aii | 1.17 | 1.49 | 2.626 (3) | 161.2 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
Hydrogen-bond geometry (Å, º) for (almer80) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O12C—H1C···O11Bi | 1.27 | 1.38 | 2.640 (3) | 170.9 |
| O12B—H1B···O11Ci | 1.31 | 1.34 | 2.646 (3) | 169.5 |
| O12A—H1A···O11Aii | 1.26 | 1.38 | 2.630 (3) | 170.1 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
Hydrogen-bond geometry (Å, º) for (d6h_s) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11C—H1C···O12Bi | 0.82 | 1.81 | 2.618 (4) | 166.2 |
| O11B—H1B···O12Ci | 0.82 | 1.79 | 2.593 (4) | 167.2 |
| O12A—H1A···O11Aii | 0.82 | 1.77 | 2.578 (6) | 170.2 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z. |
Hydrogen-bond geometry (Å, º) for (d6hlt_s) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O12A—H1A···O11Ai | 0.84 | 1.73 | 2.566 (3) | 170.4 |
| O11B—H1B···O12Cii | 0.84 | 1.76 | 2.591 (2) | 167.7 |
| O11C—H1C···O12Bii | 0.84 | 1.80 | 2.622 (3) | 166.0 |
| Symmetry codes: (i) −x+1, −y, −z; (ii) −x+2, −y+1, −z+1. |
 O interactions. When exposed to UV radiation the α′-OETCA polymorph can be stabilized below 333 K with ca 8% of the monomer converted into the photodimer. The crystal structures of α′-OETCA are reported at two temperatures above the phase transition point (at 345 and 375 K) as well as the stabilized forms at 173 and 293 K. A mechanism for the phase transition involving a cooperative conformational transformation coupled with a shift of layers of OETCA molecules is proposed. The α′-OETCA polymorph is also an example of a cinnamic acid derivative where two different potentially photoreactive environments exist in one crystal in which each unit cell has two non-centrosymmetric predimer sites and one centrosymmetric predimer site.
O interactions. When exposed to UV radiation the α′-OETCA polymorph can be stabilized below 333 K with ca 8% of the monomer converted into the photodimer. The crystal structures of α′-OETCA are reported at two temperatures above the phase transition point (at 345 and 375 K) as well as the stabilized forms at 173 and 293 K. A mechanism for the phase transition involving a cooperative conformational transformation coupled with a shift of layers of OETCA molecules is proposed. The α′-OETCA polymorph is also an example of a cinnamic acid derivative where two different potentially photoreactive environments exist in one crystal in which each unit cell has two non-centrosymmetric predimer sites and one centrosymmetric predimer site.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig4thm.gif)
![[Figure 5]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig5thm.gif)
![[Figure 6]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig6thm.gif)
![[Figure 7]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig7thm.gif)
![[Figure 8]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig8thm.gif)
![[Figure 9]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig9thm.gif)
![[Figure 10]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig10thm.gif)
![[Figure 11]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig11thm.gif)
![[Figure 12]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig12thm.gif)
![[Figure 13]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig13thm.gif)
![[Figure 14]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig14thm.gif)
![[Figure 15]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig15thm.gif)
![[Figure 16]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig16thm.gif)
![[Figure 17]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig17thm.gif)
![[Figure 18]](http://journals.iucr.org/b/issues/2004/03/00/ws5009/ws5009fig18thm.gif)



