1,3-Ninhydrin dihydrazone featuring an R44(12) motif with 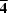 symmetry and comprising only N and H atoms
symmetry and comprising only N and H atoms
 O hydrogen bonds. Each molecule is asymmetrically involved in an extensive three-dimensional network of N-H
O hydrogen bonds. Each molecule is asymmetrically involved in an extensive three-dimensional network of N-H O and N-H
O and N-H N hydrogen bonds, and the structure also exhibits weaker
N hydrogen bonds, and the structure also exhibits weaker ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) and C=O
and C=O C interactions. The structure features an R44(12) motif consisting solely of N and H atoms and possessing crystallographic
C interactions. The structure features an R44(12) motif consisting solely of N and H atoms and possessing crystallographic Supporting information
Crystallographic Information File (CIF) https://doi.org/10.1107/S0108270112034749/uk3049sup1.cif | |
Structure factor file (CIF format) https://doi.org/10.1107/S0108270112034749/uk3049Isup2.hkl | |
Chemdraw file https://doi.org/10.1107/S0108270112034749/uk3049Isup3.cdx | |
Chemical Markup Language (CML) file https://doi.org/10.1107/S0108270112034749/uk3049Isup4.cml |
CCDC reference: 908139
For related literature, see: Allen (2002); Derebe et al. (2002, 2003); Etter et al. (1990); Everse & Menzel (1986); Fregeau et al. (2000); Glidewell et al. (2004); Holme & Peck (1998); Koteswara & Ram Reddy (1990); Mehabaw (2001); Menzel & Mitchell (1990); Negash (2003); Urban et al. (2003); Vogel (1989).
All reagents used were of analytical grade and solvents were dried and distilled prior to use (Vogel, 1989). Ninhydrin (2.00 g, 11.23 mmol) was dissolved in hot ethanol (100 ml). The solution was cooled to room temperature and hydrazine monohydrate (20 ml, 41.2 mmol) was added. The reaction mixture was stirred continuously for 1 h. Water (100 ml) was then added and the mixture left to stand overnight. The yellow precipitate which formed was filtered off and washed with aqueous ethanol. The product, (I), was recrystallized from ethanol (yield 1.40 g, 60%; m.p. 517–518 K).
The H atoms of the NH2 groups were located in ΔF syntheses and refined freely, with restraints of 0.88 (1) Å on the N—H distances and with Uiso(H) = 1.5Ueq(N). The remaining H atoms were placed geometrically and treated as riding, with C—H = 0.96 Å and Uiso(H) = 1.2Ueq(C). Six reflections flagged as outliers were omitted from the refinement.
Data collection: SMART (Bruker, 2001); cell refinement: SAINT (Bruker, 2002); data reduction: SAINT (Bruker, 2002) and SHELXTL (Sheldrick, 2008); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008); molecular graphics: Mercury (Macrae et al., 2008); software used to prepare material for publication: enCIFer (Allen et al., 2004), PLATON (Spek, 2009) and publCIF (Westrip, 2010).
| C9H8N4O | Dx = 1.422 Mg m−3 |
| Mr = 188.19 | Melting point = 517–518 K |
| Tetragonal, P42/n | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 4bc | Cell parameters from 2364 reflections |
| a = 13.808 (6) Å | θ = 2.7–27.5° |
| c = 9.219 (4) Å | µ = 0.10 mm−1 |
| V = 1757.7 (13) Å3 | T = 150 K |
| Z = 8 | Block, pale yellow |
| F(000) = 784 | 0.22 × 0.19 × 0.09 mm |
| Bruker SMART1000 CCD area-detector diffractometer | 1275 reflections with I > 2σ(I) |
| Radiation source: normal-focus sealed tube | Rint = 0.071 |
| Graphite monochromator | θmax = 27.6°, θmin = 2.1° |
| Detector resolution: 8.336 pixels mm-1 | h = −12→17 |
| ω scans | k = −16→17 |
| 11027 measured reflections | l = −11→11 |
| 2006 independent reflections |
| Refinement on F2 | 0 constraints |
| Least-squares matrix: full | Primary atom site location: structure-invariant direct methods |
| R[F2 > 2σ(F2)] = 0.039 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.098 | H atoms treated by a mixture of independent and constrained refinement |
| S = 0.92 | w = 1/[σ2(Fo2) + (0.052P)2] where P = (Fo2 + 2Fc2)/3 |
| 2006 reflections | (Δ/σ)max < 0.001 |
| 139 parameters | Δρmax = 0.18 e Å−3 |
| 4 restraints | Δρmin = −0.15 e Å−3 |
| C9H8N4O | Z = 8 |
| Mr = 188.19 | Mo Kα radiation |
| Tetragonal, P42/n | µ = 0.10 mm−1 |
| a = 13.808 (6) Å | T = 150 K |
| c = 9.219 (4) Å | 0.22 × 0.19 × 0.09 mm |
| V = 1757.7 (13) Å3 |
| Bruker SMART1000 CCD area-detector diffractometer | 1275 reflections with I > 2σ(I) |
| 11027 measured reflections | Rint = 0.071 |
| 2006 independent reflections |
| R[F2 > 2σ(F2)] = 0.039 | 4 restraints |
| wR(F2) = 0.098 | H atoms treated by a mixture of independent and constrained refinement |
| S = 0.92 | Δρmax = 0.18 e Å−3 |
| 2006 reflections | Δρmin = −0.15 e Å−3 |
| 139 parameters |
Refinement. The N—H H atoms were refined with restraints of 0.88 (1) Å on the N—H distances. |
| x | y | z | Uiso*/Ueq | ||
| C1 | 0.12468 (10) | 0.38955 (11) | −0.00914 (16) | 0.0286 (4) | |
| C2 | 0.09595 (11) | 0.36494 (11) | 0.14018 (17) | 0.0299 (4) | |
| O2 | 0.08356 (9) | 0.28287 (8) | 0.19053 (12) | 0.0385 (3) | |
| C3 | 0.08281 (11) | 0.45781 (11) | 0.21813 (17) | 0.0304 (4) | |
| C4 | 0.10809 (11) | 0.53580 (11) | 0.11763 (17) | 0.0292 (4) | |
| C5 | 0.13211 (11) | 0.49525 (11) | −0.01863 (16) | 0.0288 (4) | |
| C6 | 0.10800 (12) | 0.63560 (12) | 0.13719 (19) | 0.0369 (4) | |
| H6 | 0.0921 | 0.6629 | 0.2287 | 0.044* | |
| C7 | 0.13161 (12) | 0.69448 (13) | 0.0207 (2) | 0.0419 (4) | |
| H7 | 0.1325 | 0.7628 | 0.0329 | 0.050* | |
| C8 | 0.15400 (13) | 0.65477 (12) | −0.1140 (2) | 0.0418 (4) | |
| H8 | 0.1692 | 0.6964 | −0.1928 | 0.050* | |
| C9 | 0.15444 (12) | 0.55537 (12) | −0.13482 (18) | 0.0350 (4) | |
| H9 | 0.1698 | 0.5288 | −0.2270 | 0.042* | |
| N10 | 0.05134 (10) | 0.47118 (10) | 0.34980 (14) | 0.0353 (4) | |
| N11 | 0.03183 (12) | 0.39269 (12) | 0.43159 (16) | 0.0445 (4) | |
| H11A | 0.0300 (15) | 0.3365 (10) | 0.384 (2) | 0.067* | |
| H11B | −0.0002 (14) | 0.4046 (15) | 0.5128 (15) | 0.067* | |
| N12 | 0.13888 (9) | 0.33026 (9) | −0.11777 (14) | 0.0301 (3) | |
| N13 | 0.12666 (11) | 0.23621 (10) | −0.09558 (15) | 0.0359 (3) | |
| H13A | 0.1120 (13) | 0.2143 (14) | −0.0066 (13) | 0.054* | |
| H13B | 0.1391 (13) | 0.1970 (12) | −0.1695 (15) | 0.054* |
| U11 | U22 | U33 | U12 | U13 | U23 | |
| C1 | 0.0257 (8) | 0.0327 (9) | 0.0274 (8) | −0.0007 (7) | 0.0020 (6) | −0.0038 (7) |
| C2 | 0.0287 (9) | 0.0332 (9) | 0.0278 (9) | 0.0011 (7) | 0.0011 (7) | −0.0022 (7) |
| O2 | 0.0515 (8) | 0.0323 (7) | 0.0318 (7) | 0.0001 (5) | 0.0066 (5) | 0.0006 (5) |
| C3 | 0.0271 (8) | 0.0371 (9) | 0.0270 (8) | 0.0016 (7) | 0.0014 (6) | −0.0047 (7) |
| C4 | 0.0221 (8) | 0.0331 (9) | 0.0325 (9) | 0.0008 (6) | −0.0002 (6) | −0.0038 (7) |
| C5 | 0.0229 (8) | 0.0328 (9) | 0.0309 (9) | −0.0003 (7) | 0.0016 (6) | −0.0025 (7) |
| C6 | 0.0324 (9) | 0.0359 (10) | 0.0424 (10) | 0.0010 (7) | 0.0039 (7) | −0.0093 (8) |
| C7 | 0.0387 (10) | 0.0299 (9) | 0.0569 (12) | −0.0030 (8) | 0.0051 (9) | −0.0031 (8) |
| C8 | 0.0387 (10) | 0.0370 (10) | 0.0496 (11) | −0.0044 (8) | 0.0071 (9) | 0.0063 (8) |
| C9 | 0.0324 (9) | 0.0375 (10) | 0.0351 (9) | −0.0028 (7) | 0.0059 (7) | −0.0006 (8) |
| N10 | 0.0366 (8) | 0.0407 (8) | 0.0286 (8) | 0.0043 (6) | 0.0019 (6) | −0.0037 (6) |
| N11 | 0.0632 (11) | 0.0415 (9) | 0.0288 (9) | 0.0065 (8) | 0.0124 (7) | −0.0003 (7) |
| N12 | 0.0289 (7) | 0.0312 (8) | 0.0303 (7) | 0.0001 (6) | 0.0029 (6) | −0.0045 (6) |
| N13 | 0.0447 (9) | 0.0321 (8) | 0.0310 (8) | −0.0007 (7) | 0.0065 (7) | −0.0065 (6) |
| C1—C2 | 1.472 (2) | C7—C8 | 1.392 (2) |
| C1—C5 | 1.466 (2) | C7—H7 | 0.9500 |
| C1—N12 | 1.308 (2) | C8—C9 | 1.386 (2) |
| C2—O2 | 1.2365 (19) | C8—H8 | 0.9500 |
| C2—C3 | 1.481 (2) | C9—H9 | 0.9500 |
| C3—N10 | 1.302 (2) | N10—N11 | 1.347 (2) |
| C3—C4 | 1.463 (2) | N11—H11A | 0.891 (9) |
| C4—C6 | 1.390 (2) | N11—H11B | 0.885 (9) |
| C4—C5 | 1.415 (2) | N12—N13 | 1.325 (2) |
| C5—C9 | 1.390 (2) | N13—H13A | 0.897 (9) |
| C6—C7 | 1.386 (2) | N13—H13B | 0.888 (9) |
| C6—H6 | 0.9500 | ||
| N12—C1—C5 | 124.54 (14) | C6—C7—C8 | 120.82 (16) |
| N12—C1—C2 | 127.71 (14) | C6—C7—H7 | 119.6 |
| C5—C1—C2 | 107.73 (13) | C8—C7—H7 | 119.6 |
| O2—C2—C1 | 126.86 (14) | C9—C8—C7 | 120.98 (16) |
| O2—C2—C3 | 126.47 (15) | C9—C8—H8 | 119.5 |
| C1—C2—C3 | 106.67 (14) | C7—C8—H8 | 119.5 |
| N10—C3—C4 | 124.44 (14) | C8—C9—C5 | 118.93 (15) |
| N10—C3—C2 | 128.01 (15) | C8—C9—H9 | 120.5 |
| C4—C3—C2 | 107.51 (14) | C5—C9—H9 | 120.5 |
| C6—C4—C5 | 120.52 (14) | C3—N10—N11 | 118.31 (14) |
| C6—C4—C3 | 130.36 (15) | N10—N11—H11A | 115.6 (14) |
| C5—C4—C3 | 109.08 (14) | N10—N11—H11B | 115.1 (14) |
| C9—C5—C4 | 120.00 (15) | H11A—N11—H11B | 124 (2) |
| C9—C5—C1 | 131.02 (14) | C1—N12—N13 | 118.42 (13) |
| C4—C5—C1 | 108.95 (13) | N12—N13—H13A | 120.0 (13) |
| C7—C6—C4 | 118.74 (15) | N12—N13—H13B | 117.1 (13) |
| C7—C6—H6 | 120.6 | H13A—N13—H13B | 122.7 (19) |
| C4—C6—H6 | 120.6 |
| D—H···A | D—H | H···A | D···A | D—H···A |
| N11—H11A···O2 | 0.89 (1) | 2.07 (2) | 2.784 (2) | 136 (2) |
| N13—H13A···O2 | 0.90 (1) | 2.09 (2) | 2.780 (2) | 133 (2) |
| N13—H13B···N12i | 0.89 (1) | 2.16 (1) | 3.024 (2) | 164 (2) |
| N11—H11B···N10ii | 0.89 (1) | 2.25 (2) | 2.986 (2) | 141 (2) |
| N13—H13A···N11iii | 0.90 (1) | 2.61 (2) | 3.213 (3) | 125 (2) |
| Symmetry codes: (i) −y+1/2, x, −z−1/2; (ii) −x, −y+1, −z+1; (iii) −y+1/2, x, −z+1/2. |
Experimental details
| Crystal data | |
| Chemical formula | C9H8N4O |
| Mr | 188.19 |
| Crystal system, space group | Tetragonal, P42/n |
| Temperature (K) | 150 |
| a, c (Å) | 13.808 (6), 9.219 (4) |
| V (Å3) | 1757.7 (13) |
| Z | 8 |
| Radiation type | Mo Kα |
| µ (mm−1) | 0.10 |
| Crystal size (mm) | 0.22 × 0.19 × 0.09 |
| Data collection | |
| Diffractometer | Bruker SMART1000 CCD area-detector diffractometer |
| Absorption correction | – |
| No. of measured, independent and observed [I > 2σ(I)] reflections | 11027, 2006, 1275 |
| Rint | 0.071 |
| (sin θ/λ)max (Å−1) | 0.651 |
| Refinement | |
| R[F2 > 2σ(F2)], wR(F2), S | 0.039, 0.098, 0.92 |
| No. of reflections | 2006 |
| No. of parameters | 139 |
| No. of restraints | 4 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 0.18, −0.15 |
Computer programs: SMART (Bruker, 2001), SAINT (Bruker, 2002) and SHELXTL (Sheldrick, 2008), SHELXS97 (Sheldrick, 2008), SHELXL97 (Sheldrick, 2008), Mercury (Macrae et al., 2008), enCIFer (Allen et al., 2004), PLATON (Spek, 2009) and publCIF (Westrip, 2010).
| C1—C2 | 1.472 (2) | C4—C5 | 1.415 (2) |
| C1—C5 | 1.466 (2) | C5—C9 | 1.390 (2) |
| C1—N12 | 1.308 (2) | C6—C7 | 1.386 (2) |
| C2—O2 | 1.2365 (19) | C7—C8 | 1.392 (2) |
| C2—C3 | 1.481 (2) | C8—C9 | 1.386 (2) |
| C3—N10 | 1.302 (2) | N10—N11 | 1.347 (2) |
| C3—C4 | 1.463 (2) | N12—N13 | 1.325 (2) |
| C4—C6 | 1.390 (2) |
| D—H···A | D—H | H···A | D···A | D—H···A |
| N11—H11A···O2 | 0.891 (9) | 2.070 (16) | 2.784 (2) | 136 (2) |
| N13—H13A···O2 | 0.897 (9) | 2.087 (16) | 2.780 (2) | 133 (2) |
| N13—H13B···N12i | 0.888 (9) | 2.161 (11) | 3.024 (2) | 164 (2) |
| N11—H11B···N10ii | 0.885 (9) | 2.246 (15) | 2.986 (2) | 141 (2) |
| N13—H13A···N11iii | 0.897 (9) | 2.614 (17) | 3.213 (3) | 125 (2) |
| Symmetry codes: (i) −y+1/2, x, −z−1/2; (ii) −x, −y+1, −z+1; (iii) −y+1/2, x, −z+1/2. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
- Information on subscribing
- Sample issue
- Purchase subscription
- Reduced-price subscriptions
- If you have already subscribed, you may need to register

 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2012/09/00/uk3049/uk3049fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2012/09/00/uk3049/uk3049fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2012/09/00/uk3049/uk3049fig3thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



Ninhydrin is a widely used reagent for qualitative and quantitative analysis of all amino acids possessing a primary amine function (Holme & Peck, 1998); together they form a purple-coloured compound, popularly known as Ruhemann's Purple. Several methods which utilize ninhydrin have been developed for enhancing fingerprints (Everse & Menzel, 1986; Menzel & Mitchell, 1990; Fregeau et al., 2000). It also exhibits strong antibacterial and antiviral properties. Derivatives of ninhydrin are of importance as multidentate ligands in metal complexation. Reactions between ninhydrin and α-amino acids in the presence of transition metal ions have been documented to form a variety of coloured compounds, which are different from Ruhemann's Purple (Koteswara Rao & Ram Reddy, 1990; Mehabaw, 2001; Derebe et al., 2002, 2003; Negash, 2003).
Derivatives of ninhydrin with simple amines carrying suitable substituents, such as hydrazine, hydroxylamine, semicarbazide and thiosemicarbazide, with the potential for metal binding have not been systematically investigated for their structural and metal-complexation characteristics. Literature surveys reveal that no significant attempts have been made to investigate the structural characteristics of ninhydrin dihydrazone. Being a tricarbonyl system, ninhydrin can exhibit variable reactivity towards amine functions. In the case of α-amino acids, only one of the three carbonyl functions is known to be involved in the condensation process, with several subsequent steps leading to Ruhemann's Purple. However, with other amines, a variable number of carbonyls may be involved in the condensation reaction, depending upon steric and electronic factors. It is therefore worthwhile to undertake studies of the reactivity of ninhydrin towards amine systems other than α-amino acids as they are likely to result in interesting network systems. Consequently, these systems could have further applications in fingerprinting, metal targeting and biochemical processes.
Isolated molecules of the title compound, (I), exhibit a good approximation to C2v symmetry, with the atoms lying close to the molecular plane and the NH2 groups both cis to the carbonyl group (Fig. 1). Although the atoms of the two rings are coplanar to within 0.04 Å, they can also be visualized as two separate rings which exhibit greater degrees of planarity, with C1–C5 and C4–C9 showing r.m.s. deviations of only 0.011 and 0.004 Å, respectively: the dihedral angle between these rings is 1.43 (6)°. The inclusion of the NH2 groups in this planar arrangement is assisted by the presence of short intramolecular N11—H11A···O2 and N13—H13A···O2 hydrogen bonds (Fig. 1 and Table 2); these favour the mutually cis arrangement of NH2 groups over a trans arrangement which might be more favourable to the formation of intermolecular hydrogen bonding. The only significant deviation from this local symmetry appears for the N—N distances, where N10—N11 is 0.022 (3) Å longer than N12—N13 (Table 1); we attribute this to the differing contributions made by the NH2 groups of N11 and N13 to the formation of the intermolecular hydrogen-bonding network (see below).
Fig. 2 shows the central molecule in the same orientation as in Fig. 1 to illustrate the local hydrogen-bonding network, which is extensive despite the presence of the short intramolecular hydrogen bonds. A significant feature of the structure is the very different patterns of interaction with neighbouring molecules: whereas the N10/N11 side of the molecule forms pairwise N—H···N hydrogen bonds with a single molecule [graph-set notation R22(6); Etter et al., 1990], the N12/N13 side participates in three hydrogen bonds to three different molecules, with atom N12 acting as a hydrogen-bond acceptor and both H atoms on N13 acting as hydrogen-bond donors. The molecules form undulating chains via N13—H13B···N12i and pairwise N11—H11B···N10ii interactions [symmetry codes: (i) -y + 1/2, x, -z - 1/2; (ii) -x, -y + 1, -z + 1], with the mean planes through alternate pairs of molecules disposed approximately orthogonal to each other. The molecules of each pair are related by inversion [symmetry code (-x, -y + 1, -z + 1)] but are not coplanar, occupying parallel molecular planes 0.547 Å apart.
The undulating chains are crosslinked by N12···H13Biv—N13Biv hydrogen bonds [symmetry code: (iv) y, -x + 1/2, -z - 1/2], resulting in the formation of a three-dimensional network (Fig. 3) which includes an R44(12) motif comprising only N and H atoms and exhibiting crystallographically imposed 4 symmetry. Only about 40 examples of this motif are known, and they are dominated by pyrazoles and similar species containing cyclic N═NH units (Cambridge Structural Database, Version 5.33, May 2012; Allen, 2002). However, two hydrazone examples have been reported, namely anthracene-9-carbaldehyde hydrazone (Urban et al., 2003) and 2-nitrobenzaldehyde hydrazone (Glidewell et al., 2004).
An intermolecular O2···C3iii contact of 2.944 (2) Å [symmetry code: (iii) y + 1/2, x, -z + 1/2] is observed between two molecules which lie approximately perpendicular to each other, as indicated by the dihedral angle of 85.82 (4)° between the molecular planes (Fig. 2). The related C2═O2···C3iii and O2···C3iii—C2iii angles have values of 173.84 (11) and 81.71 (11)°, respectively. Stacking interactions occur between the five-membered ring and the symmetry-related six-membered ring at (-x, -y + 1, -z), with a centroid-to-centroid distance of 3.473 (2) Å and a perpendicular distance of 3.3980 (7) Å.
In summary, the most interesting features of the title molecule are its approximate C2v symmetry and the cis orientation of its NH2 groups, which are assisted by intramolecular N—H···O hydrogen bonding; its asymmetric involvement in a three-dimensional intermolecular hydrogen-bonded network; the presence of an R44(12) motif with 4 symmetry containing only N and H atoms; and the presence of weaker π–π and C═ O···C interactions.