Buy article online - an online subscription or single-article purchase is required to access this article.
The title compound, (C
4H
12N)
4[Ta
6Cl
18]Cl, crystallizes in the cubic space group
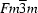
. The crystal structure contains two different types of coordination polyhedra,
i.e. four tetrahedral [(CH
3)
4N]
+ cations and one octahedral [(Ta
6Cl
12)Cl
6]
3- cluster anion, and one Cl
- ion. The presence of three different kinds of Cl atoms [bridging (
![[mu]](/logos/entities/mu_rmgif.gif) 2
2), terminal and counter-anion] in one molecule makes this substance unique in the chemistry of hexanuclear halide clusters of niobium and tantalum. The Ta
6 octahedron has an ideal
Oh symmetry, with a Ta-Ta interatomic distance of 2.9215 (7) Å.
Supporting information
CCDC reference: 231041
Ethanol solutions of freshly prepared [Ta6Cl12]Cl2·6C2H5OH (10 ml, 0.376 g, 0.202 mmol) and (CH3)4NCl (5 ml, 0.133 g, 1.212 mmol) were mixed in air, and then the reaction flask was degassed with a stream of dry nitrogen and the reaction mixture was stirred magnetically for half an hour. After stirring, the contents of the flask were transferred into the ampoule, which was filled with dry nitrogen, sealed and left at room temperature. Dark green, nearly black, octahedral single crystals appeared on the walls of the ampoule over a period of one month.
All H atoms were included in the structure-factor calculation in idealized positions and were allowed to ride on neighbouring atoms, with relative isotropic displacement coefficients.
Data collection: CAD-4 Software (Enraf–Nonius, 1998); cell refinement: CAD-4 Software; data reduction: HELENA (Spek, 1997); program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: WinGX (Farrugia, 1999); software used to prepare material for publication: ORTEP-3 for Windows (Farrugia, 1997).
Tetrakis(tetramethylammonium)dodeca-µ-chloro-hexachloro-
octahedro- hexatantalate chloride
top
Crystal data top
| (C4H12N)4[Ta6Cl18]Cl | Dx = 2.273 Mg m−3 |
| Mr = 2055.83 | Mo Kα radiation, λ = 0.71073 Å |
| Cubic, Fm3m | Cell parameters from 25 reflections |
| Hall symbol: -F 4 2 3 | θ = 20.4–23.2° |
| a = 18.1781 (4) Å | µ = 11.75 mm−1 |
| V = 6006.8 (2) Å3 | T = 100 K |
| Z = 4 | Octahedra-like, dark green |
| F(000) = 3732 | 0.15 × 0.12 × 0.12 mm |
Data collection top
Enraf–Nonius CAD4
diffractometer | 412 independent reflections |
| Radiation source: fine-focus sealed tube | 351 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.063 |
| Detector resolution: ? # Not in version 2.0.1 pixels mm-1 | θmax = 27.9°, θmin = 2.2° |
| ω/2θ scans | h = −23→0 |
Absorption correction: empirical (using intensity measurements)
(North et al., 1968) | k = −23→0 |
| Tmin = 0.201, Tmax = 0.244 | l = −23→0 |
| 1958 measured reflections | |
Refinement top
| Refinement on F2 | 8 restraints |
| Least-squares matrix: full | H-atom parameters constrained |
| R[F2 > 2σ(F2)] = 0.027 | w = 1/[σ2(Fo2) + (0.0331P)2 + 58.2675P]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.068 | (Δ/σ)max = 0.001 |
| S = 1.14 | Δρmax = 0.91 e Å−3 |
| 412 reflections | Δρmin = −1.10 e Å−3 |
| 24 parameters | |
Crystal data top
| (C4H12N)4[Ta6Cl18]Cl | Z = 4 |
| Mr = 2055.83 | Mo Kα radiation |
| Cubic, Fm3m | µ = 11.75 mm−1 |
| a = 18.1781 (4) Å | T = 100 K |
| V = 6006.8 (2) Å3 | 0.15 × 0.12 × 0.12 mm |
Data collection top
Enraf–Nonius CAD4
diffractometer | 412 independent reflections |
Absorption correction: empirical (using intensity measurements)
(North et al., 1968) | 351 reflections with I > 2σ(I) |
| Tmin = 0.201, Tmax = 0.244 | Rint = 0.063 |
| 1958 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.027 | 8 restraints |
| wR(F2) = 0.068 | H-atom parameters constrained |
| S = 1.14 | w = 1/[σ2(Fo2) + (0.0331P)2 + 58.2675P]
where P = (Fo2 + 2Fc2)/3 |
| 412 reflections | Δρmax = 0.91 e Å−3 |
| 24 parameters | Δρmin = −1.10 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. A successful solution by the direct methods provided all Ta and Cl atoms from the E-map. The remaining non-hydrogen atoms were located in an alternating series of least-squares cycles and difference Fourier maps. All non-hydrogen atoms were refined with anisotropic displacement coefficients. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Ta | 0.61364 (3) | 0 | 0.5 | 0.0172 (2) | |
| Cl1 | 0.63330 (9) | 0 | 0.63330 (9) | 0.0232 (4) | |
| Cl2 | 0.75337 (17) | 0 | 0.5 | 0.0269 (6) | |
| Cl3 | 0.75 | 0.25 | 0.75 | 0.072 (5) | 0.5 |
| N | 0.6330 (5) | 0.1330 (5) | 0.8670 (5) | 0.027 (4) | 0.5 |
| C1 | 0.6772 (3) | 0.1772 (3) | 0.9126 (5) | 0.065 (6) | 0.5 |
| H1A | 0.6612 | 0.1718 | 0.9638 | 0.077* | 0.25 |
| H1B | 0.6725 | 0.2288 | 0.8977 | 0.077* | 0.25 |
| H1C | 0.7287 | 0.1618 | 0.9081 | 0.077* | 0.25 |
| C2 | 0.5884 (4) | 0.0884 (4) | 0.9116 (4) | 0.075 (14) | 0.5 |
| H2A | 0.5695 | 0.0472 | 0.8825 | 0.09* | 0.08334 |
| H2B | 0.5472 | 0.1175 | 0.9305 | 0.09* | 0.08334 |
| H2C | 0.6175 | 0.0695 | 0.9528 | 0.09* | 0.08334 |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ta | 0.0157 (3) | 0.0180 (2) | 0.0180 (2) | 0 | 0 | 0 |
| Cl1 | 0.0208 (6) | 0.0282 (10) | 0.0208 (6) | 0 | −0.0028 (8) | 0 |
| Cl2 | 0.0180 (14) | 0.0313 (10) | 0.0313 (10) | 0 | 0 | 0 |
| Cl3 | 0.072 (5) | 0.072 (5) | 0.072 (5) | 0 | 0 | 0 |
| N | 0.027 (4) | 0.027 (4) | 0.027 (4) | 0.005 (5) | −0.005 (5) | −0.005 (5) |
| C1 | 0.045 (6) | 0.045 (6) | 0.103 (17) | 0.005 (9) | −0.025 (8) | −0.025 (8) |
| C2 | 0.075 (14) | 0.075 (14) | 0.075 (14) | 0.046 (18) | −0.046 (18) | −0.046 (18) |
Geometric parameters (Å, º) top
| Ta—Tai | 2.9215 (7) | N—C2 | 1.41 (1) |
| Ta—Taii | 2.9215 (7) | N—C1 | 1.407 (8) |
| Ta—Taiii | 2.9215 (7) | N—C1vii | 1.407 (8) |
| Ta—Taiv | 2.9215 (7) | N—C1viii | 1.407 (8) |
| Ta—Cl1iv | 2.449 (2) | C1—H1A | 0.9801 |
| Ta—Cl1v | 2.449 (2) | C1—H1B | 0.9801 |
| Ta—Cl1vi | 2.449 (2) | C1—H1C | 0.9801 |
| Ta—Cl1 | 2.449 (2) | C2—H2A | 0.9801 |
| Ta—Cl2 | 2.540 (3) | C2—H2B | 0.9801 |
| Cl1—Taiii | 2.449 (2) | C2—H2C | 0.9801 |
| | | |
| Tai—Ta—Taiii | 90 | Cl1iv—Ta—Cl1vi | 88.781 (9) |
| Taii—Ta—Taiii | 60 | Cl1v—Ta—Cl1vi | 88.781 (9) |
| Tai—Ta—Taii | 60 | Cl1iv—Ta—Cl1 | 88.781 (9) |
| Tai—Ta—Taiv | 60 | Cl1v—Ta—Cl1 | 88.781 (9) |
| Taii—Ta—Taiv | 90 | Cl1vi—Ta—Cl1 | 163.23 (6) |
| Taiii—Ta—Taiv | 60 | Cl1iv—Ta—Cl2 | 81.61 (3) |
| Taiii—Cl1—Ta | 73.23 (6) | Cl1v—Ta—Cl2 | 81.61 (3) |
| Cl1iv—Ta—Tai | 95.92 (2) | Cl1vi—Ta—Cl2 | 81.61 (3) |
| Cl1v—Ta—Tai | 95.92 (2) | Cl1—Ta—Cl2 | 81.61 (3) |
| Cl1vi—Ta—Tai | 53.39 (3) | C1—N—C1viii | 110.4 (6) |
| Cl1—Ta—Tai | 143.39 (3) | C1vii—N—C1viii | 110.4 (6) |
| Cl1iv—Ta—Taii | 143.39 (3) | C1—N—C1vii | 110.4 (6) |
| Cl1v—Ta—Taii | 53.39 (3) | C2—N—C1 | 108.6 (7) |
| Cl1vi—Ta—Taii | 95.92 (2) | C2—N—C1vii | 108.6 (7) |
| Cl1—Ta—Taii | 95.92 (2) | C2—N—C1viii | 108.6 (7) |
| Cl1iv—Ta—Taiii | 95.92 (2) | N—C1—H1A | 109.5 |
| Cl1v—Ta—Taiii | 95.92 (2) | N—C1—H1B | 109.5 |
| Cl1vi—Ta—Taiii | 143.39 (3) | H1A—C1—H1B | 109.5 |
| Cl1—Ta—Taiii | 53.39 (3) | N—C1—H1C | 109.5 |
| Cl1iv—Ta—Taiv | 53.39 (3) | H1A—C1—H1C | 109.5 |
| Cl1v—Ta—Taiv | 143.39 (3) | H1B—C1—H1C | 109.5 |
| Cl1vi—Ta—Taiv | 95.92 (2) | N—C2—H2A | 109.5 |
| Cl1—Ta—Taiv | 95.92 (2) | N—C2—H2B | 109.5 |
| Cl2—Ta—Taiv | 135 | H2A—C2—H2B | 109.5 |
| Cl2—Ta—Taiii | 135 | N—C2—H2C | 109.5 |
| Cl2—Ta—Taii | 135 | H2A—C2—H2C | 109.5 |
| Cl2—Ta—Tai | 135 | H2B—C2—H2C | 109.5 |
| Cl1iv—Ta—Cl1v | 163.23 (6) | | |
| Symmetry codes: (i) −y+1/2, −z+1/2, −x+1; (ii) −z+1, −x+1/2, −y+1/2; (iii) y+1/2, z−1/2, x; (iv) z, x−1/2, y+1/2; (v) z, −x+1/2, −y+1/2; (vi) x, −y, −z+1; (vii) y+1/2, −z+1, −x+3/2; (viii) −z+3/2, x−1/2, −y+1. |
Experimental details
| Crystal data |
| Chemical formula | (C4H12N)4[Ta6Cl18]Cl |
| Mr | 2055.83 |
| Crystal system, space group | Cubic, Fm3m |
| Temperature (K) | 100 |
| a (Å) | 18.1781 (4) |
| V (Å3) | 6006.8 (2) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 11.75 |
| Crystal size (mm) | 0.15 × 0.12 × 0.12 |
| |
| Data collection |
| Diffractometer | Enraf–Nonius CAD4
diffractometer |
| Absorption correction | Empirical (using intensity measurements)
(North et al., 1968) |
| Tmin, Tmax | 0.201, 0.244 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 1958, 412, 351 |
| Rint | 0.063 |
| (sin θ/λ)max (Å−1) | 0.657 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.027, 0.068, 1.14 |
| No. of reflections | 412 |
| No. of parameters | 24 |
| No. of restraints | 8 |
| H-atom treatment | H-atom parameters constrained |
| w = 1/[σ2(Fo2) + (0.0331P)2 + 58.2675P]
where P = (Fo2 + 2Fc2)/3 |
| Δρmax, Δρmin (e Å−3) | 0.91, −1.10 |
Selected geometric parameters (Å, º) top| Ta—Tai | 2.9215 (7) | N—C2 | 1.41 (1) |
| Ta—Cl1ii | 2.449 (2) | N—C1 | 1.407 (8) |
| Ta—Cl2 | 2.540 (3) | | |
| | | |
| Tai—Ta—Taiii | 90 | Cl2—Ta—Tai | 135 |
| Taiv—Ta—Taiii | 60 | Cl1ii—Ta—Cl1vi | 163.23 (6) |
| Taiii—Cl1—Ta | 73.23 (6) | Cl1ii—Ta—Cl1v | 88.781 (9) |
| Cl1ii—Ta—Tai | 95.92 (2) | Cl1ii—Ta—Cl2 | 81.61 (3) |
| Cl1v—Ta—Tai | 53.39 (3) | C1—N—C1vii | 110.4 (6) |
| Cl1—Ta—Tai | 143.39 (3) | C2—N—C1 | 108.6 (7) |
| Symmetry codes: (i) −y+1/2, −z+1/2, −x+1; (ii) z, x−1/2, y+1/2; (iii) y+1/2, z−1/2, x; (iv) −z+1, −x+1/2, −y+1/2; (v) x, −y, −z+1; (vi) z, −x+1/2, −y+1/2; (vii) −z+3/2, x−1/2, −y+1. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
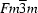 . The crystal structure contains two different types of coordination polyhedra, i.e. four tetrahedral [(CH3)4N]+ cations and one octahedral [(Ta6Cl12)Cl6]3- cluster anion, and one Cl- ion. The presence of three different kinds of Cl atoms [bridging (
. The crystal structure contains two different types of coordination polyhedra, i.e. four tetrahedral [(CH3)4N]+ cations and one octahedral [(Ta6Cl12)Cl6]3- cluster anion, and one Cl- ion. The presence of three different kinds of Cl atoms [bridging (![[mu]](/logos/entities/mu_rmgif.gif) 2), terminal and counter-anion] in one molecule makes this substance unique in the chemistry of hexanuclear halide clusters of niobium and tantalum. The Ta6 octahedron has an ideal Oh symmetry, with a Ta-Ta interatomic distance of 2.9215 (7) Å.
2), terminal and counter-anion] in one molecule makes this substance unique in the chemistry of hexanuclear halide clusters of niobium and tantalum. The Ta6 octahedron has an ideal Oh symmetry, with a Ta-Ta interatomic distance of 2.9215 (7) Å.![[mu]](/logos/entities/mu_rmgif.gif) 2), terminal and counter-anion] in one molecule makes this substance unique in the chemistry of hexanuclear halide clusters of niobium and tantalum. The Ta6 octahedron has an ideal Oh symmetry, with a Ta-Ta interatomic distance of 2.9215 (7) Å.
2), terminal and counter-anion] in one molecule makes this substance unique in the chemistry of hexanuclear halide clusters of niobium and tantalum. The Ta6 octahedron has an ideal Oh symmetry, with a Ta-Ta interatomic distance of 2.9215 (7) Å.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2004/01/00/sq1032/sq1032fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2004/01/00/sq1032/sq1032fig2thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



In the hexanuclear halide clusters [M6X12]n+ (M = Nb, Ta; X = Cl, Br; n = 2, 3, 4), the lowest oxidation potentials for the oxidation of [M6X12]2+ → [M6X12]3+ to [M6X12]4+ have been found for [Ta6Cl12]n+ (Eisenbraun & Schäfer, 1985). Consequently, the largest number of compounds containing [M6X12]n+ (n = 3 or 4) has been isolated for this cluster, by using oxidants that are not common for the oxidation of other members in the [M6X12]n+ series. For example, fast air-oxygen oxidation of [Ta6Cl12]2+ to [Ta6Cl12]4+ in an alkaline medium has been used as a method of preparation of crystalline cluster hydroxides [Ta6Cl12(OH)6]2− (Brničević et al., 1984) or methoxides (Brničević et al., 1988). A direct synthesis of the title compound, which contains [Ta6Cl12]3+ ions, has now been accomplished in a one-step oxidation of the [Ta6Cl12]Cl2·6C2H5OH precursor in ethanol solution in the presence of Me4NCl, by using air-oxygen as an oxidant.
The title compound consists of four tetrahedral [(CH3)4N]+ cations, one octahedral [(Ta6Cl12)Cl6]3− cluster anion and one Cl− counteranion (Fig. 1). The Ta6 octahedron has an ideal Oh symmetry, with a Ta—Ta interatomic distance of 2.9215 (7) Å. This value is in good agreement with the Ta—Ta bond lengths found for other compounds containing the [Ta6Cl12]3+ cluster unit, specifically with the values [2.925 (1) and 2.911 (1) Å] found in CsPb[(Ta6Cl12)Cl6] (Cordier et al., 1999) and [(Ta6Cl12)Cl(H2O)5][HgBr4]·9H2O (Vojnović et al., 1997), respectively. Simultaneously, this value is intermediate between the Ta—Ta bond lengths [2.889 (1) and 2.973 (1) Å] found in In4[(Ta6Cl12)Cl6] (Bájan et al., 1995) and [N(CH3)4]2[Ta6Cl12(OH)6].21H2O (Beck et al., 1997), containing [Ta6Cl12]2+ and [Ta6Cl12]4+ ions, respectively.
The [(CH3)4N]4[(Ta6Cl12)Cl6]Cl cluster is the first in the chemistry of hexanuclear halide clusters of niobium and tantalum to have three different kinds of Cl atoms present simultaneously in the same molecule; 12 Cl atoms are bridging (µ-Cl), six Cl atoms occupy terminal octahedral coordination sites and Cl atom one is a counteranion. The Ta—(µ-Cl) interatomic distance [2.449 (2) Å] is the same as that found for (CH3)4N[Ta6Cl12(H2O)6]Br4 (Brničević et al., 1985), i.e. intermediate between the mean values [2.464 (2) and 2.414 (5) Å] found for [Ta6Cl12(C2H5OH)6][(Mo6Cl8)Cl6] (Bašic et al., 1998) and H2[(Ta6Cl12)Cl6]·6H2O (Thaxton & Jacobson, 1971), respectively, which contain [Ta6Cl12]2+ and [Ta6Cl12]4+ entities. The Ta—Clterminal bond length [2.540 (3) Å] is comparable to the values [2.531 (4) and 2.574 (2) Å] found for trans-[(Ta6Cl12)Cl2(PEt3)4]·CHCl3 (Imoto et al., 1990) and CsPb[(Ta6Cl12)Cl6] (Cordier et al., 1999), respectively.
The interatomic C—N distances in the tetrahedral [(CH3)4N]+ cations are as expected for a single C—N bond [mean 1.408 (9) Å].
The crystal packing of cations and anions in the unit cell of the title compound is shown in Fig. 2. The packing is influenced exclusively by the attractive interionic forces. The [(Ta6Cl12)Cl6]3− cluster anions are situated at the corners and centers of the faces of the unit cell, while the Cl− counteranions lie in the centers of eight octants that make up the unit cell, in a manner similar to that observed in CaF2-type structures. The [(CH3)4N]+ cations, located around the cluster anion, and Cl− counteranions occupy one-half of the possible crystallographic positions, thus satisfying the charge neutrality of the compound.
The title compound is paramagnetic, as found for the [Ta6Cl12]3+ ion. The magnetic susceptibility in the temperature range 5–300 K is nearly constant (µeff = 1.4–1.6 BM), indicating no interacting [Ta6Cl12]3+ cluster entities.