In the title complex, [Hg(CH
4N
2S)
4][Mn(NCS)
4], the Hg and Mn atoms sit at special positions with
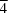
symmetry and are tetrahedrally coordinated to four thiourea (TU) S and four thiocyanate (SCN) N atoms, respectively. The structure consists of discrete cationic and anionic [Hg(TU)
4]
2+ and [Mn(SCN)
4]
2- complexes, and weak N
TU-H

S
SCN hydrogen-bond bridges exist between these complexes.
Supporting information
CCDC reference: 159976
To an aqueous solution (20 ml) containing MnHg(SCN)4 (5.2 g, 10.7 mmol)
thiourea (4.2 g, 13.8 mmol) was added; the pH of the solution was adjusted to
3 by adding hydrochloric acid. This mixture was heated and stirred until the
MnHg(SCN)4 had dissolved. The aqueous solution was then allowed to stand at
room temperature. After a few hours, colourless crystals of (I) suitable for
X-ray structure analysis were obtained.
All H atoms were placed in calculated positions and refined riding on their
parent atoms. N—H = ?
Data collection: XSCANS (Siemens, 1996); cell refinement: XSCANS; data reduction: XSCANS; program(s) used to solve structure: SHELXTL (Bruker, 1997); program(s) used to refine structure: SHELXTL; molecular graphics: SHELXTL.
Tetrathioureamercury(II) tetrathiocyanatomanganate(II)
top
Crystal data top
| [Hg(CH4N2S)4][Mn(NCS)4] | Dx = 2.074 Mg m−3 |
| Mr = 792.32 | Mo Kα radiation, λ = 0.71073 Å |
| Tetragonal, I4 | Cell parameters from 36 reflections |
| a = 17.4297 (8) Å | θ = 4.7–12.6° |
| c = 4.1754 (3) Å | µ = 7.22 mm−1 |
| V = 1268.46 (12) Å3 | T = 293 K |
| Z = 2 | Needle, colourless |
| F(000) = 762 | 0.26 × 0.22 × 0.20 mm |
Data collection top
Bruker P4
diffractometer | 1337 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.025 |
| Graphite monochromator | θmax = 30.0°, θmin = 2.3° |
| ω/2θ scans | h = −24→1 |
Absorption correction: ψ-scan
XSCANS (Siemens, 1996) | k = −24→1 |
| Tmin = 0.181, Tmax = 0.236 | l = −5→1 |
| 1492 measured reflections | 3 standard reflections every 97 reflections |
| 1374 independent reflections | intensity decay: none |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.033 | H-atom parameters constrained |
| wR(F2) = 0.080 | w = 1/[σ2(Fo2) + (0.0451P)2 + 0.118P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.05 | (Δ/σ)max < 0.001 |
| 1374 reflections | Δρmax = 0.70 e Å−3 |
| 68 parameters | Δρmin = −1.02 e Å−3 |
| 0 restraints | Absolute structure: Flack (1983) |
| Primary atom site location: structure-invariant direct methods | Absolute structure parameter: 0.008 (11) |
Crystal data top
| [Hg(CH4N2S)4][Mn(NCS)4] | Z = 2 |
| Mr = 792.32 | Mo Kα radiation |
| Tetragonal, I4 | µ = 7.22 mm−1 |
| a = 17.4297 (8) Å | T = 293 K |
| c = 4.1754 (3) Å | 0.26 × 0.22 × 0.20 mm |
| V = 1268.46 (12) Å3 | |
Data collection top
Bruker P4
diffractometer | 1337 reflections with I > 2σ(I) |
Absorption correction: ψ-scan
XSCANS (Siemens, 1996) | Rint = 0.025 |
| Tmin = 0.181, Tmax = 0.236 | 3 standard reflections every 97 reflections |
| 1492 measured reflections | intensity decay: none |
| 1374 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.033 | H-atom parameters constrained |
| wR(F2) = 0.080 | Δρmax = 0.70 e Å−3 |
| S = 1.05 | Δρmin = −1.02 e Å−3 |
| 1374 reflections | Absolute structure: Flack (1983) |
| 68 parameters | Absolute structure parameter: 0.008 (11) |
| 0 restraints | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Hg1 | 0 | 1/2 | 1/4 | 0.03778 (13) | |
| Mn1 | 0 | 0 | 0 | 0.0526 (7) | |
| C1 | 0.1269 (3) | −0.0982 (3) | −0.382 (2) | 0.0416 (14) | |
| C2 | 0.1938 (3) | 0.4517 (4) | 0.4411 (19) | 0.0411 (15) | |
| N1 | 0.0793 (3) | −0.0646 (3) | −0.258 (5) | 0.0561 (15) | |
| N2 | 0.2657 (3) | 0.4714 (4) | 0.501 (2) | 0.061 (2) | |
| H2A | 0.3025 | 0.4411 | 0.4484 | 0.073* | |
| H2B | 0.2755 | 0.5144 | 0.5936 | 0.073* | |
| N3 | 0.1800 (3) | 0.3870 (3) | 0.303 (3) | 0.060 (3) | |
| H3A | 0.2174 | 0.3572 | 0.2507 | 0.072* | |
| H3B | 0.1335 | 0.3736 | 0.2622 | 0.072* | |
| S1 | 0.19485 (12) | −0.14458 (13) | −0.5666 (7) | 0.0599 (5) | |
| S2 | 0.12388 (8) | 0.51356 (9) | 0.5818 (5) | 0.0397 (4) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Hg1 | 0.03218 (14) | 0.03218 (14) | 0.0490 (2) | 0 | 0 | 0 |
| Mn1 | 0.0330 (5) | 0.0330 (5) | 0.092 (2) | 0 | 0 | 0 |
| C1 | 0.035 (3) | 0.035 (3) | 0.054 (4) | −0.006 (2) | −0.007 (3) | 0.005 (3) |
| C2 | 0.029 (2) | 0.038 (3) | 0.056 (4) | 0.002 (2) | −0.001 (3) | 0.016 (3) |
| N1 | 0.045 (2) | 0.046 (2) | 0.077 (4) | −0.001 (2) | 0.007 (7) | 0.008 (7) |
| N2 | 0.025 (2) | 0.056 (4) | 0.102 (6) | 0.004 (2) | −0.004 (3) | −0.003 (4) |
| N3 | 0.035 (2) | 0.047 (2) | 0.098 (8) | 0.0108 (19) | −0.008 (4) | −0.019 (5) |
| S1 | 0.0491 (9) | 0.0570 (10) | 0.0736 (14) | −0.0035 (8) | 0.0090 (10) | −0.0125 (11) |
| S2 | 0.0284 (6) | 0.0365 (7) | 0.0542 (10) | 0.0003 (5) | −0.0028 (7) | −0.0025 (7) |
Geometric parameters (Å, º) top
| Hg1—S2i | 2.5762 (16) | Mn1—N1vi | 2.083 (12) |
| Hg1—S2 | 2.5762 (16) | C1—N1 | 1.141 (13) |
| Hg1—S2ii | 2.5762 (16) | C1—S1 | 1.627 (7) |
| Hg1—S2iii | 2.5762 (16) | C2—N3 | 1.291 (10) |
| Mn1—N1iv | 2.083 (12) | C2—N2 | 1.324 (8) |
| Mn1—N1v | 2.083 (12) | C2—S2 | 1.729 (7) |
| Mn1—N1 | 2.083 (12) | | |
| | | |
| S2i—Hg1—S2 | 106.81 (4) | N1iv—Mn1—N1vi | 105.5 (4) |
| S2i—Hg1—S2ii | 106.81 (4) | N1v—Mn1—N1vi | 105.5 (4) |
| S2—Hg1—S2ii | 114.94 (8) | N1—Mn1—N1vi | 117.8 (10) |
| S2i—Hg1—S2iii | 114.94 (8) | N1—C1—S1 | 178.6 (10) |
| S2—Hg1—S2iii | 106.81 (4) | N3—C2—N2 | 119.2 (6) |
| S2ii—Hg1—S2iii | 106.81 (4) | N3—C2—S2 | 124.5 (5) |
| N1iv—Mn1—N1v | 117.8 (10) | N2—C2—S2 | 116.2 (6) |
| N1iv—Mn1—N1 | 105.5 (4) | C1—N1—Mn1 | 174.7 (12) |
| N1v—Mn1—N1 | 105.5 (4) | C2—S2—Hg1 | 110.5 (3) |
| Symmetry codes: (i) y−1/2, −x+1/2, −z+1/2; (ii) −x, −y+1, z; (iii) −y+1/2, x+1/2, −z+1/2; (iv) −y, x, −z; (v) y, −x, −z; (vi) −x, −y, z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H2B···S1vii | 0.86 | 2.73 | 3.584 (8) | 175 |
| N3—H3A···S1viii | 0.86 | 2.68 | 3.515 (7) | 163 |
| N3—H3B···S2i | 0.86 | 2.54 | 3.322 (8) | 152 |
| Symmetry codes: (i) y−1/2, −x+1/2, −z+1/2; (vii) −x+1/2, −y+1/2, z+3/2; (viii) y+1/2, −x+1/2, −z−1/2. |
Experimental details
| Crystal data |
| Chemical formula | [Hg(CH4N2S)4][Mn(NCS)4] |
| Mr | 792.32 |
| Crystal system, space group | Tetragonal, I4 |
| Temperature (K) | 293 |
| a, c (Å) | 17.4297 (8), 4.1754 (3) |
| V (Å3) | 1268.46 (12) |
| Z | 2 |
| Radiation type | Mo Kα |
| µ (mm−1) | 7.22 |
| Crystal size (mm) | 0.26 × 0.22 × 0.20 |
| |
| Data collection |
| Diffractometer | Bruker P4
diffractometer |
| Absorption correction | ψ-scan
XSCANS (Siemens, 1996) |
| Tmin, Tmax | 0.181, 0.236 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 1492, 1374, 1337 |
| Rint | 0.025 |
| (sin θ/λ)max (Å−1) | 0.702 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.033, 0.080, 1.05 |
| No. of reflections | 1374 |
| No. of parameters | 68 |
| H-atom treatment | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.70, −1.02 |
| Absolute structure | Flack (1983) |
| Absolute structure parameter | 0.008 (11) |
Selected geometric parameters (Å, º) top| Hg1—S2 | 2.5762 (16) | C2—N3 | 1.291 (10) |
| Mn1—N1 | 2.083 (12) | C2—N2 | 1.324 (8) |
| C1—N1 | 1.141 (13) | C2—S2 | 1.729 (7) |
| C1—S1 | 1.627 (7) | | |
| | | |
| S2i—Hg1—S2 | 106.81 (4) | N3—C2—N2 | 119.2 (6) |
| S2—Hg1—S2ii | 114.94 (8) | N3—C2—S2 | 124.5 (5) |
| N1iii—Mn1—N1iv | 117.8 (10) | N2—C2—S2 | 116.2 (6) |
| N1iii—Mn1—N1 | 105.5 (4) | C1—N1—Mn1 | 174.7 (12) |
| N1—C1—S1 | 178.6 (10) | C2—S2—Hg1 | 110.5 (3) |
| Symmetry codes: (i) y−1/2, −x+1/2, −z+1/2; (ii) −x, −y+1, z; (iii) −y, x, −z; (iv) y, −x, −z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H2B···S1v | 0.86 | 2.726 | 3.584 (8) | 175.33 |
| N3—H3A···S1vi | 0.86 | 2.684 | 3.515 (7) | 162.67 |
| N3—H3B···S2i | 0.86 | 2.537 | 3.322 (8) | 152.08 |
| Symmetry codes: (i) y−1/2, −x+1/2, −z+1/2; (v) −x+1/2, −y+1/2, z+3/2; (vi) y+1/2, −x+1/2, −z−1/2. |
 SSCN hydrogen-bond bridges exist between these complexes.
SSCN hydrogen-bond bridges exist between these complexes.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2001/02/00/sk1404/sk1404fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2001/02/00/sk1404/sk1404fig2thm.gif)




During the last few years, organic non-linear optical (NLO) crystals have attracted much interest, due to their superior properties such as higher susceptibility, faster response and the capability to design components on the molecular level. However, unlike inorganic NLO crystals, they have not come into wide use, owing to drawbacks such as the difficulty of growing large perfect single crystals, poor physicochemical stability and difficulties in cutting and polishing of the devices. Under these circumstances, crystals of complexes of organometallic materials with NLO effects have been developed which are expected not only to retain high NLO effects, but also to minimize some of the shortcomings of pure organic crystals; in other words, they have the advantages of both organic and inorganic crystals in terms of their physicochemical properties. This approach has resulted in their practical use in frequency-doubling of laser radiation (Long, 1995; Jiang & Fang, 1999).
Transition-metal thiourea (TU) and thiocyanate (SCN) coordination complexes are potentially useful candidates for such organometallic systems. As ligands with potential S and N donors, the TU molecule and the SCN ion are interesting not only because of the structural chemistry of their multifunctional coordination modes, but also because of the possibility of formation of organometallic coordination complexes with NLO activities (Xing et al., 1993; Bhat & Dharmaprakash, 1998; Sturmer & Deserno, 1970; Yuan et al., 1997). It would be even more interesting if these two ligands were both present in one compound and this compound were to possess good NLO properties. The crystal structure of [Hg(CH4N2S)4][Co(NCS)4], (II), was reported in the 1960 s (Korczynski & Porai-Koshits, 1965). It crystallizes in a noncentrosymmetric space group, but unfortunately the low energy d-d transitions present in (II) due to the Co2+ ion are normally observed in the visible light region and so limit its NLO usefulness. The title compound, (I), is isostructural with (II). However, the new crystal has far better characteristics than those of (II), such as a relatively high nonlinearity and a greater ease of growing into large single crystals. \sch
The concept of hard and soft acids and bases rationalizes that soft cations show a pronounced affinity for coordination with the softer ligands, while hard cations prefer coordination with harder ligands (Pearson, 1966; Balarew & Duhlew, 1984). In the structure of (I) (Fig.1), the soft Hg2+ ion is coordinated by the softer S (TU), while the hard Mn2+ ion is coordinated by the harder N (SCN). Neither of them is coordinated by the medium S (SCN).
Both of the metal cations in (I) are tetracoordinated and in a tetrahedral environment. Although all the Hg—S and Mn—N bond lengths are 2.576 (2) and 2.083 (12) Å, respectively, the bond angles for S—Hg—S and N—Mn—N range from 106.81 (4) to 114.94 (8) and 105.5 (4) to 117.8 (10)°, respectively, which obviously deviate from typical tetrahedral angles. The TU molecules and the SCN ions are linked by N—H····S hydrogen bonds to form a three-dimensional network in space (Fig. 2).
In short, the tetrahedral geometries of the Hg and Mn cores in (I) are both slightly distorted from the regular forms. The macroscopic non-linear susceptibility may be related to microscopic hyperpolarizabilities of the polarizable TU molecules, the dipolar SCN ions and the deformed tetrahedra HgS4 and MnN4, which are connected via N—H····S hydrogen bonds. These factors result in a large polarization, which in turn, we believe, induces a large macroscopic non-linearity (Zyss & Ledoux, 1994). The second harmonic generation (SHG) effect of the crystals was measured by the powder SHG technique (Kurtz & Perry, 1968) and was found to be somewhat superior to that of urea crystals (specific data and refs for comparison?).