A novel two-dimensional (2D) Zn
II coordination framework, poly[[μ-1,3-bis(2-methyl-1
H-imidazol-1-yl)benzene](μ-5-nitrobenzene-1,3-dicarboxylato)zinc(II)], [Zn(C
8H
3NO
6)(C
14H
14N
4)]
n or [Zn(NO
2-BDC)(1,3-BMIB)]
n [1,3-BMIB is 1,3-bis(2-methyl-1
H-imidazol-1-yl)benzene and NO
2-H
2BDC is 5-nitrobenzene-1,3-dicarboxylic acid], has been prepared and characterized by IR, elemental analysis, thermal analysis and single-crystal X-ray diffraction. Single-crystal X-ray diffraction analysis revealed that the compound is a new 2D polymer with a 6
3 topology parallel to the (10
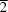
) crystal planes based on left-handed helices, right-handed helical NO
2-BDC–Zn chains and [Zn
2(1,3-BMIB)
2]
n clusters. In the crystal, adjacent layers are further connected by C—H

O hydrogen bonds, C—H

π interactions, C—O

π interactions and N—O

π interactions to form a three-dimensional structure in the solid state. In addition, the compound exhibits strong fluorescence emissions in the solid state at room temperature.
Supporting information
CCDC reference: 1892014
Data collection: SMART (Bruker, 2001); cell refinement: SMART (Bruker, 2001); data reduction: SAINT-Plus (Bruker, 2009); program(s) used to solve structure: SHELXT (Sheldrick, 2015a); program(s) used to refine structure: SHELXL2014 (Sheldrick, 2015b); molecular graphics: DIAMOND (Brandenburg, 2010); software used to prepare material for publication: publCIF (Westrip, 2010).
Poly[[µ-1,3-bis(2-methyl-1
H-imidazol-1-yl)benzene](µ-5-nitrobenzene-1,3-dicarboxylato)zinc(II)]
top
Crystal data top
| [Zn(C8H3NO6)(C14H14N4)] | F(000) = 1048 |
| Mr = 512.80 | Dx = 1.533 Mg m−3 |
| Monoclinic, P21/c | Mo Kα radiation, λ = 0.71073 Å |
| a = 12.568 (6) Å | Cell parameters from 9121 reflections |
| b = 13.324 (7) Å | θ = 2.7–27.6° |
| c = 14.207 (7) Å | µ = 1.16 mm−1 |
| β = 110.912 (5)° | T = 296 K |
| V = 2222.3 (19) Å3 | Block, colorless |
| Z = 4 | 0.21 × 0.19 × 0.17 mm |
Data collection top
Bruker SMART CCD area detector
diffractometer | 4022 reflections with I > 2σ(I) |
| Radiation source: x-ray tube | Rint = 0.019 |
| phi and ω scans | θmax = 26.0°, θmin = 2.2° |
Absorption correction: multi-scan
(SADABS; Bruker, 2008) | h = −15→15 |
| Tmin = 0.785, Tmax = 0.822 | k = −16→16 |
| 22595 measured reflections | l = −17→17 |
| 4359 independent reflections | |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.027 | H-atom parameters constrained |
| wR(F2) = 0.075 | w = 1/[σ2(Fo2) + (0.0402P)2 + 1.0538P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.07 | (Δ/σ)max = 0.001 |
| 4359 reflections | Δρmax = 0.77 e Å−3 |
| 309 parameters | Δρmin = −0.25 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| C22 | 1.13159 (19) | −0.11355 (16) | 0.7143 (2) | 0.0572 (6) | |
| H22A | 1.2090 | −0.1085 | 0.7175 | 0.086* | |
| H22B | 1.1292 | −0.1060 | 0.7807 | 0.086* | |
| H22C | 1.0868 | −0.0617 | 0.6713 | 0.086* | |
| C21 | 1.08495 (15) | −0.21308 (12) | 0.67325 (14) | 0.0346 (4) | |
| N4 | 1.14047 (12) | −0.28589 (10) | 0.64756 (12) | 0.0354 (3) | |
| C20 | 1.06470 (17) | −0.36404 (15) | 0.6134 (2) | 0.0540 (6) | |
| H20 | 1.0811 | −0.4254 | 0.5904 | 0.065* | |
| C19 | 0.96355 (19) | −0.33822 (16) | 0.6184 (2) | 0.0613 (7) | |
| H19 | 0.8979 | −0.3772 | 0.5996 | 0.074* | |
| N3 | 0.97671 (13) | −0.24200 (12) | 0.65710 (14) | 0.0404 (4) | |
| C17 | 0.89046 (15) | −0.17656 (13) | 0.66832 (15) | 0.0383 (4) | |
| C16 | 0.87098 (18) | −0.17327 (15) | 0.75749 (16) | 0.0466 (5) | |
| H16 | 0.9100 | −0.2160 | 0.8103 | 0.056* | |
| C15 | 0.79188 (19) | −0.10500 (16) | 0.76769 (16) | 0.0495 (5) | |
| H15 | 0.7768 | −0.1033 | 0.8272 | 0.059* | |
| Zn1 | 0.69472 (2) | 0.27715 (2) | 0.34318 (2) | 0.02960 (8) | |
| O1 | 0.63764 (11) | 0.40797 (9) | 0.36525 (10) | 0.0387 (3) | |
| O2 | 0.76641 (15) | 0.39284 (13) | 0.51902 (12) | 0.0633 (5) | |
| O3 | 0.39155 (12) | 0.70809 (9) | 0.28248 (9) | 0.0397 (3) | |
| O4 | 0.42046 (13) | 0.83278 (9) | 0.39240 (11) | 0.0471 (3) | |
| O6 | 0.74405 (17) | 0.61513 (14) | 0.78460 (11) | 0.0703 (5) | |
| O5 | 0.61084 (16) | 0.72622 (13) | 0.75137 (12) | 0.0616 (4) | |
| N1 | 0.67650 (12) | 0.15549 (11) | 0.42241 (11) | 0.0334 (3) | |
| N2 | 0.70400 (13) | 0.02398 (11) | 0.52039 (11) | 0.0360 (3) | |
| N5 | 0.66318 (15) | 0.66121 (13) | 0.72578 (12) | 0.0442 (4) | |
| C1 | 0.68582 (15) | 0.43617 (13) | 0.45671 (14) | 0.0355 (4) | |
| C18 | 0.83326 (16) | −0.11390 (14) | 0.58925 (15) | 0.0388 (4) | |
| H18 | 0.8454 | −0.1182 | 0.5285 | 0.047* | |
| C2 | 0.43737 (14) | 0.74757 (13) | 0.36904 (12) | 0.0297 (3) | |
| C5 | 0.51988 (13) | 0.67903 (12) | 0.44649 (12) | 0.0284 (3) | |
| C3 | 0.63867 (14) | 0.53008 (12) | 0.48752 (13) | 0.0299 (3) | |
| C4 | 0.56069 (14) | 0.59160 (12) | 0.41710 (12) | 0.0292 (3) | |
| H4 | 0.5353 | 0.5742 | 0.3493 | 0.035* | |
| C8 | 0.67298 (14) | 0.55369 (13) | 0.58900 (13) | 0.0324 (3) | |
| H8 | 0.7243 | 0.5129 | 0.6372 | 0.039* | |
| C6 | 0.55464 (15) | 0.70326 (12) | 0.54799 (13) | 0.0318 (3) | |
| H6 | 0.5284 | 0.7612 | 0.5691 | 0.038* | |
| C11 | 0.74495 (15) | 0.11483 (13) | 0.50702 (14) | 0.0353 (4) | |
| C13 | 0.75728 (15) | −0.04436 (13) | 0.60172 (14) | 0.0359 (4) | |
| C9 | 0.58795 (15) | 0.08804 (14) | 0.38118 (14) | 0.0378 (4) | |
| H9 | 0.5269 | 0.0978 | 0.3212 | 0.045* | |
| C10 | 0.60323 (16) | 0.00658 (14) | 0.44041 (14) | 0.0396 (4) | |
| H10 | 0.5563 | −0.0494 | 0.4300 | 0.047* | |
| C14 | 0.73531 (17) | −0.03944 (14) | 0.68993 (15) | 0.0426 (4) | |
| H14 | 0.6835 | 0.0070 | 0.6973 | 0.051* | |
| C7 | 0.62931 (14) | 0.63912 (13) | 0.61724 (12) | 0.0325 (3) | |
| C12 | 0.85115 (18) | 0.16005 (16) | 0.57886 (17) | 0.0535 (5) | |
| H12A | 0.9161 | 0.1284 | 0.5709 | 0.080* | |
| H12B | 0.8537 | 0.1503 | 0.6466 | 0.080* | |
| H12C | 0.8522 | 0.2306 | 0.5655 | 0.080* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C22 | 0.0445 (11) | 0.0418 (11) | 0.0854 (17) | −0.0048 (9) | 0.0233 (11) | −0.0264 (11) |
| C21 | 0.0314 (9) | 0.0307 (8) | 0.0412 (9) | 0.0021 (7) | 0.0124 (7) | −0.0016 (7) |
| N4 | 0.0298 (7) | 0.0294 (7) | 0.0467 (9) | 0.0011 (6) | 0.0133 (6) | −0.0050 (6) |
| C20 | 0.0398 (10) | 0.0302 (9) | 0.0921 (17) | −0.0024 (8) | 0.0237 (11) | −0.0163 (10) |
| C19 | 0.0385 (11) | 0.0332 (10) | 0.114 (2) | −0.0060 (8) | 0.0288 (12) | −0.0119 (11) |
| N3 | 0.0312 (8) | 0.0298 (7) | 0.0618 (10) | 0.0046 (6) | 0.0184 (7) | 0.0020 (7) |
| C17 | 0.0311 (9) | 0.0309 (9) | 0.0559 (11) | 0.0052 (7) | 0.0191 (8) | 0.0049 (8) |
| C16 | 0.0467 (11) | 0.0423 (10) | 0.0508 (11) | 0.0085 (8) | 0.0173 (9) | 0.0137 (9) |
| C15 | 0.0589 (12) | 0.0517 (12) | 0.0466 (11) | 0.0100 (10) | 0.0296 (10) | 0.0080 (9) |
| Zn1 | 0.02918 (12) | 0.02538 (11) | 0.03324 (12) | 0.00164 (7) | 0.00989 (8) | −0.00228 (7) |
| O1 | 0.0460 (7) | 0.0300 (6) | 0.0417 (7) | 0.0074 (5) | 0.0175 (6) | −0.0040 (5) |
| O2 | 0.0710 (10) | 0.0612 (10) | 0.0491 (8) | 0.0392 (8) | 0.0110 (8) | −0.0005 (7) |
| O3 | 0.0449 (7) | 0.0341 (6) | 0.0337 (7) | 0.0067 (5) | 0.0063 (6) | 0.0020 (5) |
| O4 | 0.0597 (9) | 0.0300 (6) | 0.0475 (8) | 0.0134 (6) | 0.0143 (7) | −0.0002 (6) |
| O6 | 0.0894 (13) | 0.0677 (11) | 0.0355 (8) | 0.0249 (10) | 0.0000 (8) | −0.0005 (7) |
| O5 | 0.0766 (11) | 0.0679 (11) | 0.0426 (8) | 0.0131 (8) | 0.0240 (8) | −0.0136 (7) |
| N1 | 0.0344 (7) | 0.0315 (7) | 0.0337 (7) | 0.0001 (6) | 0.0113 (6) | 0.0014 (6) |
| N2 | 0.0353 (8) | 0.0308 (7) | 0.0419 (8) | 0.0045 (6) | 0.0136 (6) | 0.0030 (6) |
| N5 | 0.0541 (10) | 0.0433 (9) | 0.0332 (8) | 0.0005 (8) | 0.0132 (7) | −0.0043 (7) |
| C1 | 0.0395 (9) | 0.0306 (8) | 0.0409 (10) | 0.0073 (7) | 0.0196 (8) | 0.0030 (7) |
| C18 | 0.0399 (9) | 0.0354 (9) | 0.0459 (10) | 0.0055 (8) | 0.0211 (8) | 0.0037 (8) |
| C2 | 0.0289 (8) | 0.0282 (8) | 0.0336 (8) | 0.0013 (6) | 0.0130 (7) | 0.0024 (7) |
| C5 | 0.0272 (8) | 0.0255 (7) | 0.0340 (8) | 0.0004 (6) | 0.0126 (7) | 0.0005 (6) |
| C3 | 0.0296 (8) | 0.0272 (8) | 0.0351 (8) | 0.0023 (6) | 0.0141 (7) | 0.0004 (6) |
| C4 | 0.0318 (8) | 0.0278 (8) | 0.0292 (8) | 0.0010 (6) | 0.0122 (6) | −0.0009 (6) |
| C8 | 0.0312 (8) | 0.0315 (8) | 0.0335 (9) | 0.0045 (7) | 0.0102 (7) | 0.0035 (7) |
| C6 | 0.0341 (9) | 0.0268 (8) | 0.0364 (9) | 0.0019 (7) | 0.0147 (7) | −0.0035 (7) |
| C11 | 0.0341 (9) | 0.0312 (8) | 0.0395 (9) | 0.0033 (7) | 0.0120 (7) | 0.0009 (7) |
| C13 | 0.0352 (9) | 0.0299 (8) | 0.0427 (10) | 0.0051 (7) | 0.0140 (7) | 0.0046 (7) |
| C9 | 0.0355 (9) | 0.0407 (10) | 0.0345 (9) | −0.0032 (7) | 0.0093 (7) | −0.0013 (7) |
| C10 | 0.0379 (9) | 0.0355 (9) | 0.0452 (10) | −0.0039 (7) | 0.0146 (8) | −0.0028 (8) |
| C14 | 0.0429 (10) | 0.0383 (10) | 0.0519 (11) | 0.0096 (8) | 0.0235 (9) | 0.0026 (8) |
| C7 | 0.0353 (9) | 0.0331 (8) | 0.0296 (8) | −0.0024 (7) | 0.0121 (7) | −0.0031 (7) |
| C12 | 0.0476 (12) | 0.0448 (11) | 0.0528 (12) | −0.0063 (9) | −0.0007 (9) | 0.0068 (9) |
Geometric parameters (Å, º) top
| C22—C21 | 1.483 (3) | O5—N5 | 1.219 (2) |
| C22—H22A | 0.9600 | N1—C11 | 1.319 (2) |
| C22—H22B | 0.9600 | N1—C9 | 1.387 (2) |
| C22—H22C | 0.9600 | N2—C11 | 1.355 (2) |
| C21—N4 | 1.320 (2) | N2—C10 | 1.386 (2) |
| C21—N3 | 1.352 (2) | N2—C13 | 1.435 (2) |
| N4—C20 | 1.377 (2) | N5—C7 | 1.476 (2) |
| N4—Zn1i | 2.0315 (18) | C1—C3 | 1.514 (2) |
| C20—C19 | 1.343 (3) | C18—C13 | 1.387 (2) |
| C20—H20 | 0.9300 | C18—H18 | 0.9300 |
| C19—N3 | 1.381 (3) | C2—C5 | 1.518 (2) |
| C19—H19 | 0.9300 | C5—C6 | 1.387 (2) |
| N3—C17 | 1.444 (2) | C5—C4 | 1.395 (2) |
| C17—C16 | 1.374 (3) | C3—C8 | 1.385 (2) |
| C17—C18 | 1.378 (3) | C3—C4 | 1.390 (2) |
| C16—C15 | 1.393 (3) | C4—H4 | 0.9300 |
| C16—H16 | 0.9300 | C8—C7 | 1.384 (2) |
| C15—C14 | 1.389 (3) | C8—H8 | 0.9300 |
| C15—H15 | 0.9300 | C6—C7 | 1.386 (2) |
| Zn1—O1 | 1.9524 (14) | C6—H6 | 0.9300 |
| Zn1—O3ii | 1.9563 (14) | C11—C12 | 1.488 (3) |
| Zn1—N4i | 2.0315 (18) | C13—C14 | 1.378 (3) |
| Zn1—N1 | 2.0326 (16) | C9—C10 | 1.344 (3) |
| O1—C1 | 1.279 (2) | C9—H9 | 0.9300 |
| O2—C1 | 1.226 (2) | C10—H10 | 0.9300 |
| O3—C2 | 1.271 (2) | C14—H14 | 0.9300 |
| O3—Zn1iii | 1.9563 (14) | C12—H12A | 0.9600 |
| O4—C2 | 1.223 (2) | C12—H12B | 0.9600 |
| O6—N5 | 1.226 (2) | C12—H12C | 0.9600 |
| | | |
| C21—C22—H22A | 109.5 | O2—C1—C3 | 119.22 (17) |
| C21—C22—H22B | 109.5 | O1—C1—C3 | 116.38 (15) |
| H22A—C22—H22B | 109.5 | C17—C18—C13 | 118.91 (17) |
| C21—C22—H22C | 109.5 | C17—C18—H18 | 120.5 |
| H22A—C22—H22C | 109.5 | C13—C18—H18 | 120.5 |
| H22B—C22—H22C | 109.5 | O4—C2—O3 | 125.76 (16) |
| N4—C21—N3 | 110.04 (15) | O4—C2—C5 | 120.11 (15) |
| N4—C21—C22 | 126.32 (17) | O3—C2—C5 | 114.13 (15) |
| N3—C21—C22 | 123.65 (16) | C6—C5—C4 | 119.49 (15) |
| C21—N4—C20 | 106.60 (15) | C6—C5—C2 | 119.58 (14) |
| C21—N4—Zn1i | 124.56 (12) | C4—C5—C2 | 120.94 (15) |
| C20—N4—Zn1i | 128.83 (12) | C8—C3—C4 | 119.51 (15) |
| C19—C20—N4 | 109.65 (18) | C8—C3—C1 | 118.67 (15) |
| C19—C20—H20 | 125.2 | C4—C3—C1 | 121.81 (15) |
| N4—C20—H20 | 125.2 | C3—C4—C5 | 121.11 (15) |
| C20—C19—N3 | 106.12 (18) | C3—C4—H4 | 119.4 |
| C20—C19—H19 | 126.9 | C5—C4—H4 | 119.4 |
| N3—C19—H19 | 126.9 | C7—C8—C3 | 118.69 (15) |
| C21—N3—C19 | 107.59 (16) | C7—C8—H8 | 120.7 |
| C21—N3—C17 | 123.96 (16) | C3—C8—H8 | 120.7 |
| C19—N3—C17 | 128.06 (16) | C7—C6—C5 | 118.47 (15) |
| C16—C17—C18 | 121.29 (17) | C7—C6—H6 | 120.8 |
| C16—C17—N3 | 120.60 (17) | C5—C6—H6 | 120.8 |
| C18—C17—N3 | 118.01 (17) | N1—C11—N2 | 109.74 (16) |
| C17—C16—C15 | 119.04 (18) | N1—C11—C12 | 126.32 (17) |
| C17—C16—H16 | 120.5 | N2—C11—C12 | 123.94 (16) |
| C15—C16—H16 | 120.5 | C14—C13—C18 | 121.25 (17) |
| C14—C15—C16 | 120.66 (19) | C14—C13—N2 | 120.98 (16) |
| C14—C15—H15 | 119.7 | C18—C13—N2 | 117.76 (16) |
| C16—C15—H15 | 119.7 | C10—C9—N1 | 110.18 (16) |
| O1—Zn1—O3ii | 116.79 (6) | C10—C9—H9 | 124.9 |
| O1—Zn1—N4i | 111.28 (6) | N1—C9—H9 | 124.9 |
| O3ii—Zn1—N4i | 107.83 (6) | C9—C10—N2 | 105.23 (16) |
| O1—Zn1—N1 | 120.17 (6) | C9—C10—H10 | 127.4 |
| O3ii—Zn1—N1 | 89.71 (7) | N2—C10—H10 | 127.4 |
| N4i—Zn1—N1 | 108.89 (6) | C13—C14—C15 | 118.80 (17) |
| C1—O1—Zn1 | 110.96 (11) | C13—C14—H14 | 120.6 |
| C2—O3—Zn1iii | 126.90 (12) | C15—C14—H14 | 120.6 |
| C11—N1—C9 | 106.42 (15) | C8—C7—C6 | 122.67 (15) |
| C11—N1—Zn1 | 131.79 (12) | C8—C7—N5 | 118.08 (15) |
| C9—N1—Zn1 | 120.99 (12) | C6—C7—N5 | 119.25 (15) |
| C11—N2—C10 | 108.43 (15) | C11—C12—H12A | 109.5 |
| C11—N2—C13 | 126.02 (15) | C11—C12—H12B | 109.5 |
| C10—N2—C13 | 125.52 (15) | H12A—C12—H12B | 109.5 |
| O5—N5—O6 | 123.86 (17) | C11—C12—H12C | 109.5 |
| O5—N5—C7 | 117.93 (16) | H12A—C12—H12C | 109.5 |
| O6—N5—C7 | 118.19 (16) | H12B—C12—H12C | 109.5 |
| O2—C1—O1 | 124.40 (16) | | |
| | | |
| N3—C21—N4—C20 | 0.2 (2) | C6—C5—C4—C3 | −2.2 (2) |
| C22—C21—N4—C20 | 180.0 (2) | C2—C5—C4—C3 | 178.21 (15) |
| N3—C21—N4—Zn1i | 179.29 (13) | C4—C3—C8—C7 | −0.8 (2) |
| C22—C21—N4—Zn1i | −0.9 (3) | C1—C3—C8—C7 | −179.55 (15) |
| C21—N4—C20—C19 | 0.1 (3) | C4—C5—C6—C7 | 0.0 (2) |
| Zn1i—N4—C20—C19 | −178.92 (17) | C2—C5—C6—C7 | 179.52 (15) |
| N4—C20—C19—N3 | −0.4 (3) | C9—N1—C11—N2 | 0.68 (19) |
| N4—C21—N3—C19 | −0.4 (2) | Zn1—N1—C11—N2 | −168.85 (12) |
| C22—C21—N3—C19 | 179.8 (2) | C9—N1—C11—C12 | −178.95 (19) |
| N4—C21—N3—C17 | −173.85 (17) | Zn1—N1—C11—C12 | 11.5 (3) |
| C22—C21—N3—C17 | 6.4 (3) | C10—N2—C11—N1 | −0.8 (2) |
| C20—C19—N3—C21 | 0.5 (3) | C13—N2—C11—N1 | 177.48 (16) |
| C20—C19—N3—C17 | 173.5 (2) | C10—N2—C11—C12 | 178.89 (19) |
| C21—N3—C17—C16 | −94.0 (2) | C13—N2—C11—C12 | −2.9 (3) |
| C19—N3—C17—C16 | 94.0 (3) | C17—C18—C13—C14 | −2.3 (3) |
| C21—N3—C17—C18 | 82.5 (3) | C17—C18—C13—N2 | 176.34 (16) |
| C19—N3—C17—C18 | −89.6 (3) | C11—N2—C13—C14 | 89.3 (2) |
| C18—C17—C16—C15 | −0.1 (3) | C10—N2—C13—C14 | −92.7 (2) |
| N3—C17—C16—C15 | 176.22 (19) | C11—N2—C13—C18 | −89.3 (2) |
| C17—C16—C15—C14 | −1.5 (3) | C10—N2—C13—C18 | 88.6 (2) |
| Zn1—O1—C1—O2 | 8.1 (2) | C11—N1—C9—C10 | −0.4 (2) |
| Zn1—O1—C1—C3 | −172.39 (11) | Zn1—N1—C9—C10 | 170.53 (12) |
| C16—C17—C18—C13 | 2.0 (3) | N1—C9—C10—N2 | −0.1 (2) |
| N3—C17—C18—C13 | −174.43 (17) | C11—N2—C10—C9 | 0.5 (2) |
| Zn1iii—O3—C2—O4 | 12.5 (3) | C13—N2—C10—C9 | −177.75 (16) |
| Zn1iii—O3—C2—C5 | −167.11 (11) | C18—C13—C14—C15 | 0.8 (3) |
| O4—C2—C5—C6 | 15.5 (2) | N2—C13—C14—C15 | −177.86 (18) |
| O3—C2—C5—C6 | −164.86 (15) | C16—C15—C14—C13 | 1.2 (3) |
| O4—C2—C5—C4 | −164.98 (16) | C3—C8—C7—C6 | −1.5 (3) |
| O3—C2—C5—C4 | 14.7 (2) | C3—C8—C7—N5 | 177.69 (16) |
| O2—C1—C3—C8 | −12.1 (3) | C5—C6—C7—C8 | 1.9 (3) |
| O1—C1—C3—C8 | 168.31 (16) | C5—C6—C7—N5 | −177.26 (15) |
| O2—C1—C3—C4 | 169.14 (18) | O5—N5—C7—C8 | −168.03 (18) |
| O1—C1—C3—C4 | −10.4 (2) | O6—N5—C7—C8 | 13.9 (3) |
| C8—C3—C4—C5 | 2.7 (2) | O5—N5—C7—C6 | 11.2 (3) |
| C1—C3—C4—C5 | −178.62 (15) | O6—N5—C7—C6 | −166.90 (19) |
| Symmetry codes: (i) −x+2, −y, −z+1; (ii) −x+1, y−1/2, −z+1/2; (iii) −x+1, y+1/2, −z+1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| C12—H12C···O2 | 0.96 | 2.40 | 3.291 (3) | 154 |
| C9—H9···O3ii | 0.93 | 2.54 | 2.908 (2) | 104 |
| C10—H10···O4iv | 0.93 | 2.24 | 3.160 (3) | 171 |
| C22—H22A···O3v | 0.96 | 2.52 | 3.308 (3) | 139 |
| Symmetry codes: (ii) −x+1, y−1/2, −z+1/2; (iv) x, y−1, z; (v) x+1, −y+1/2, z+1/2. |
 O hydrogen bonds, C—H
O hydrogen bonds, C—H π interactions, C—O
π interactions, C—O π interactions and N—O
π interactions and N—O π interactions to form a three-dimensional structure in the solid state. In addition, the compound exhibits strong fluorescence emissions in the solid state at room temperature.
π interactions to form a three-dimensional structure in the solid state. In addition, the compound exhibits strong fluorescence emissions in the solid state at room temperature.
 journal menu
journal menu











