Buy article online - an online subscription or single-article purchase is required to access this article.
The title compound, C
16H
17NO
3, crystallizes in the space group
P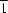
, with one molecule in the asymmetric unit. The amide H atom is engaged in an intramolecular N—H

O contact to the O atom of one of the two carbonyl groups. The molecules form centrosymmetric dimers through self-complementary C—H

O hydrogen-bonding interactions defining the
R22(14)[
R21(6)] motif. The supramolecular structure is achieved by aryl C—H

π interactions, as well as face-to-face π–π stacking interactions between lactone and benzenoid rings.
Supporting information
CCDC reference: 259572
Key indicators
- Single-crystal X-ray study
- T = 100 K
- Mean
![[sigma]](//journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.003 Å
(C-C) = 0.003 Å
- R factor = 0.056
- wR factor = 0.142
- Data-to-parameter ratio = 12.0
checkCIF/PLATON results
No syntax errors found
No errors found in this datablock
Data collection: SMART (Bruker, 2000); cell refinement: SAINT (Bruker, 2000); data reduction: SAINT; program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: SHELXTL (Bruker, 2000); software used to prepare material for publication: SHELXL97 and WinGX2003 (Farrugia, 1999).
N-Cyclohexyl-2-oxo-2
H-1-benzopyran-3-carboxamide
top
Crystal data top
| C16H17NO3 | Z = 2 |
| Mr = 271.31 | F(000) = 288 |
| Triclinic, P1 | Dx = 1.345 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 6.0870 (14) Å | Cell parameters from 600 reflections |
| b = 9.529 (2) Å | θ = 20–25° |
| c = 12.021 (3) Å | µ = 0.09 mm−1 |
| α = 93.682 (4)° | T = 100 K |
| β = 96.977 (4)° | Block, colorless |
| γ = 103.543 (4)° | 0.43 × 0.41 × 0.03 mm |
| V = 669.7 (3) Å3 | |
Data collection top
Bruker SMART area-detector
diffractometer | Rint = 0.035 |
| Graphite monochromator | θmax = 27.5°, θmin = 1.7° |
| φ and ω scans | h = −7→7 |
| 7666 measured reflections | k = −12→12 |
| 2997 independent reflections | l = −15→15 |
| 2550 reflections with I > 2σ(I) | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.056 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.142 | All H-atom parameters refined |
| S = 1.10 | w = 1/[σ2(Fo2) + (0.0662P)2 + 0.1763P]
where P = (Fo2 + 2Fc2)/3 |
| 2997 reflections | (Δ/σ)max < 0.001 |
| 249 parameters | Δρmax = 0.36 e Å−3 |
| 0 restraints | Δρmin = −0.26 e Å−3 |
Special details top
Geometry. Bond distances, angles etc. have been calculated using the rounded
fractional coordinates. All e.s.d.'s are estimated from the variances of the
(full) variance-covariance matrix. The cell e.s.d.'s are taken into account in
the estimation of distances, angles and torsion angles |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor
wR and goodness of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The threshold expression of F2 >
σ(F2) is used only for calculating R-factors(gt) etc.
and is not relevant to the choice of reflections for refinement.
R-factors based on F2 are statistically about twice as large
as those based on F, and R- factors based on ALL data will be
even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O1 | −0.26356 (19) | 0.16633 (12) | 0.00991 (10) | 0.0181 (3) | |
| O2 | −0.2852 (2) | 0.31126 (13) | 0.15537 (10) | 0.0227 (4) | |
| O11 | 0.36029 (19) | 0.58065 (13) | 0.12896 (10) | 0.0198 (4) | |
| N12 | 0.0854 (2) | 0.53211 (15) | 0.24168 (11) | 0.0160 (4) | |
| C2 | −0.1688 (3) | 0.28584 (17) | 0.08511 (14) | 0.0156 (5) | |
| C3 | 0.0613 (3) | 0.36935 (17) | 0.07359 (13) | 0.0146 (5) | |
| C4 | 0.1752 (3) | 0.32328 (18) | −0.00581 (14) | 0.0148 (5) | |
| C5 | 0.1924 (3) | 0.13624 (19) | −0.15886 (14) | 0.0183 (5) | |
| C6 | 0.0868 (3) | 0.00897 (19) | −0.22438 (15) | 0.0204 (5) | |
| C7 | −0.1365 (3) | −0.06339 (19) | −0.21298 (14) | 0.0194 (5) | |
| C8 | −0.2550 (3) | −0.00988 (19) | −0.13586 (14) | 0.0192 (5) | |
| C9 | −0.1458 (3) | 0.11797 (18) | −0.06955 (14) | 0.0157 (5) | |
| C10 | 0.0763 (3) | 0.19394 (17) | −0.07922 (13) | 0.0157 (5) | |
| C11 | 0.1810 (3) | 0.50328 (17) | 0.14994 (13) | 0.0146 (5) | |
| C13 | 0.1973 (3) | 0.65462 (17) | 0.32436 (14) | 0.0149 (5) | |
| C14 | 0.0268 (3) | 0.69626 (19) | 0.39493 (14) | 0.0176 (5) | |
| C15 | 0.1454 (3) | 0.8274 (2) | 0.47928 (16) | 0.0215 (5) | |
| C16 | 0.3476 (3) | 0.7979 (2) | 0.55463 (15) | 0.0221 (5) | |
| C17 | 0.5153 (3) | 0.7500 (2) | 0.48492 (15) | 0.0211 (5) | |
| C18 | 0.3940 (3) | 0.61972 (19) | 0.40022 (15) | 0.0189 (5) | |
| H4 | 0.318 (3) | 0.373 (2) | −0.0132 (15) | 0.016 (5)* | |
| H5 | 0.345 (3) | 0.188 (2) | −0.1631 (15) | 0.017 (5)* | |
| H6 | 0.168 (3) | −0.032 (2) | −0.2772 (17) | 0.028 (5)* | |
| H7 | −0.203 (3) | −0.150 (2) | −0.2558 (17) | 0.023 (5)* | |
| H8 | −0.406 (4) | −0.056 (2) | −0.1257 (16) | 0.026 (5)* | |
| H12 | −0.056 (4) | 0.474 (2) | 0.2462 (17) | 0.033 (6)* | |
| H13 | 0.255 (3) | 0.734 (2) | 0.2851 (15) | 0.012 (4)* | |
| H14A | −0.101 (3) | 0.720 (2) | 0.3446 (16) | 0.024 (5)* | |
| H14B | −0.043 (3) | 0.611 (2) | 0.4364 (16) | 0.023 (5)* | |
| H15A | 0.198 (3) | 0.907 (2) | 0.4370 (16) | 0.023 (5)* | |
| H15B | 0.035 (4) | 0.854 (2) | 0.5235 (17) | 0.029 (5)* | |
| H16A | 0.430 (3) | 0.885 (2) | 0.6037 (17) | 0.026 (5)* | |
| H16B | 0.288 (3) | 0.724 (2) | 0.6037 (16) | 0.022 (5)* | |
| H17A | 0.588 (3) | 0.831 (2) | 0.4416 (16) | 0.025 (5)* | |
| H17B | 0.640 (4) | 0.723 (2) | 0.5350 (17) | 0.031 (5)* | |
| H18A | 0.500 (3) | 0.595 (2) | 0.3535 (15) | 0.019 (5)* | |
| H18B | 0.326 (3) | 0.536 (2) | 0.4427 (17) | 0.025 (5)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O1 | 0.0161 (6) | 0.0171 (6) | 0.0184 (6) | −0.0007 (5) | 0.0032 (5) | −0.0027 (5) |
| O2 | 0.0181 (6) | 0.0243 (7) | 0.0226 (7) | −0.0015 (5) | 0.0076 (5) | −0.0052 (5) |
| O11 | 0.0166 (6) | 0.0176 (6) | 0.0214 (7) | −0.0043 (5) | 0.0075 (5) | −0.0040 (5) |
| N12 | 0.0140 (7) | 0.0165 (7) | 0.0152 (7) | −0.0002 (5) | 0.0020 (5) | −0.0017 (5) |
| C2 | 0.0159 (8) | 0.0140 (8) | 0.0155 (8) | 0.0017 (6) | 0.0015 (6) | −0.0002 (6) |
| C3 | 0.0138 (8) | 0.0139 (8) | 0.0151 (8) | 0.0018 (6) | 0.0016 (6) | 0.0014 (6) |
| C4 | 0.0119 (8) | 0.0144 (8) | 0.0171 (8) | 0.0009 (6) | 0.0019 (6) | 0.0022 (6) |
| C5 | 0.0170 (8) | 0.0196 (9) | 0.0176 (9) | 0.0039 (7) | 0.0020 (7) | −0.0010 (7) |
| C6 | 0.0236 (9) | 0.0216 (9) | 0.0168 (8) | 0.0082 (7) | 0.0026 (7) | −0.0017 (7) |
| C7 | 0.0244 (9) | 0.0143 (8) | 0.0163 (8) | 0.0028 (7) | −0.0034 (7) | −0.0025 (6) |
| C8 | 0.0178 (9) | 0.0185 (9) | 0.0191 (9) | 0.0010 (7) | −0.0002 (7) | 0.0027 (7) |
| C9 | 0.0176 (8) | 0.0166 (8) | 0.0133 (8) | 0.0045 (6) | 0.0019 (6) | 0.0027 (6) |
| C10 | 0.0167 (8) | 0.0143 (8) | 0.0155 (8) | 0.0029 (6) | 0.0010 (6) | 0.0023 (6) |
| C11 | 0.0154 (8) | 0.0137 (8) | 0.0149 (8) | 0.0040 (6) | 0.0018 (6) | 0.0017 (6) |
| C13 | 0.0156 (8) | 0.0136 (8) | 0.0148 (8) | 0.0018 (6) | 0.0037 (6) | −0.0004 (6) |
| C14 | 0.0130 (8) | 0.0213 (9) | 0.0179 (8) | 0.0043 (7) | 0.0017 (7) | −0.0016 (7) |
| C15 | 0.0187 (9) | 0.0243 (10) | 0.0212 (9) | 0.0069 (7) | 0.0031 (7) | −0.0073 (8) |
| C16 | 0.0200 (9) | 0.0260 (10) | 0.0176 (9) | 0.0038 (7) | 0.0006 (7) | −0.0063 (8) |
| C17 | 0.0148 (8) | 0.0241 (9) | 0.0228 (9) | 0.0051 (7) | −0.0006 (7) | −0.0052 (7) |
| C18 | 0.0164 (8) | 0.0205 (9) | 0.0203 (9) | 0.0061 (7) | 0.0038 (7) | −0.0030 (7) |
Geometric parameters (Å, º) top
| O1—C2 | 1.371 (2) | C15—C16 | 1.529 (3) |
| O1—C9 | 1.380 (2) | C16—C17 | 1.526 (3) |
| O2—C2 | 1.214 (2) | C17—C18 | 1.533 (3) |
| O11—C11 | 1.230 (2) | C4—H4 | 0.905 (19) |
| N12—C11 | 1.350 (2) | C5—H5 | 0.953 (19) |
| N12—C13 | 1.459 (2) | C6—H6 | 0.967 (19) |
| N12—H12 | 0.92 (2) | C7—H7 | 0.921 (19) |
| C2—C3 | 1.467 (3) | C8—H8 | 0.95 (2) |
| C3—C11 | 1.501 (2) | C13—H13 | 0.938 (19) |
| C3—C4 | 1.354 (2) | C14—H14A | 1.004 (19) |
| C4—C10 | 1.430 (2) | C14—H14B | 1.015 (19) |
| C5—C6 | 1.376 (3) | C15—H15A | 0.955 (19) |
| C5—C10 | 1.413 (2) | C15—H15B | 0.97 (2) |
| C6—C7 | 1.400 (3) | C16—H16A | 0.979 (19) |
| C7—C8 | 1.383 (3) | C16—H16B | 0.980 (19) |
| C8—C9 | 1.389 (2) | C17—H17A | 1.005 (19) |
| C9—C10 | 1.399 (3) | C17—H17B | 1.00 (2) |
| C13—C18 | 1.528 (3) | C18—H18A | 0.964 (19) |
| C13—C14 | 1.522 (3) | C18—H18B | 1.009 (19) |
| C14—C15 | 1.532 (3) | | |
| | | |
| O1···O11i | 3.1448 (18) | C11···H18A | 2.881 (18) |
| O1···C6ii | 3.364 (2) | C14···H18Av | 3.092 (19) |
| O2···N12 | 2.7426 (19) | C16···H6vii | 2.98 (2) |
| O11···C4iii | 3.317 (2) | C17···H14Avi | 3.097 (19) |
| O11···C2i | 3.160 (2) | C17···H15Bvi | 3.05 (2) |
| O11···C18 | 3.234 (2) | C18···H18Bviii | 3.072 (19) |
| O11···O1i | 3.1448 (18) | H4···O11 | 2.474 (18) |
| O11···C5iii | 3.319 (2) | H4···H5 | 2.49 (3) |
| O1···H8iv | 2.64 (2) | H4···O11iii | 2.510 (19) |
| O2···H12 | 1.99 (2) | H5···H4 | 2.49 (3) |
| O2···H8iv | 2.68 (2) | H5···O11iii | 2.470 (19) |
| O11···H13 | 2.539 (18) | H6···C16ix | 2.98 (2) |
| O11···H18A | 2.714 (18) | H6···H15Bix | 2.52 (3) |
| O11···H4 | 2.474 (18) | H6···H16Aix | 2.49 (3) |
| O11···H4iii | 2.510 (19) | H8···O1iv | 2.64 (2) |
| O11···H5iii | 2.470 (19) | H8···O2iv | 2.68 (2) |
| N12···O2 | 2.7426 (19) | H8···C2iv | 3.09 (2) |
| C2···C7ii | 3.447 (3) | H12···O2 | 1.99 (2) |
| C2···O11i | 3.160 (2) | H12···C2 | 2.47 (2) |
| C2···C6ii | 3.464 (3) | H12···H14B | 2.54 (3) |
| C2···C11i | 3.574 (2) | H13···O11 | 2.539 (18) |
| C3···C7ii | 3.547 (3) | H13···H15A | 2.49 (3) |
| C3···C11i | 3.341 (2) | H13···H17A | 2.54 (3) |
| C3···C3i | 3.317 (2) | H14A···C17v | 3.097 (19) |
| C4···C11i | 3.401 (3) | H14A···H18Av | 2.46 (3) |
| C4···O11iii | 3.317 (2) | H14A···C5i | 2.760 (19) |
| C5···O11iii | 3.319 (2) | H14A···C6i | 3.028 (19) |
| C6···C2ii | 3.464 (3) | H14B···H12 | 2.54 (3) |
| C6···O1ii | 3.364 (2) | H14B···H18B | 2.51 (3) |
| C7···C3ii | 3.547 (3) | H15A···H13 | 2.49 (3) |
| C7···C2ii | 3.447 (3) | H15B···C17v | 3.05 (2) |
| C8···C10ii | 3.426 (3) | H15B···H6vii | 2.52 (3) |
| C9···C9ii | 3.554 (3) | H15B···H17Bv | 2.46 (3) |
| C10···C8ii | 3.426 (3) | H16A···H6vii | 2.49 (3) |
| C11···C3i | 3.341 (2) | H17A···H13 | 2.54 (3) |
| C11···C2i | 3.574 (2) | H17B···H15Bvi | 2.46 (3) |
| C11···C4i | 3.401 (3) | H17B···H18Bviii | 2.55 (3) |
| C14···C17v | 3.552 (3) | H18A···O11 | 2.714 (18) |
| C17···C14vi | 3.552 (3) | H18A···C11 | 2.881 (18) |
| C18···O11 | 3.234 (2) | H18A···C14vi | 3.092 (19) |
| C2···H12 | 2.47 (2) | H18A···H14Avi | 2.46 (3) |
| C2···H8iv | 3.09 (2) | H18B···H14B | 2.51 (3) |
| C5···H14Ai | 2.760 (19) | H18B···C18viii | 3.072 (19) |
| C6···H14Ai | 3.028 (19) | H18B···H17Bviii | 2.55 (3) |
| | | |
| C2—O1—C9 | 122.59 (14) | C10—C5—H5 | 117.1 (11) |
| C11—N12—C13 | 121.00 (14) | C5—C6—H6 | 120.0 (11) |
| C13—N12—H12 | 122.7 (13) | C7—C6—H6 | 119.8 (11) |
| C11—N12—H12 | 116.1 (13) | C6—C7—H7 | 119.1 (12) |
| O1—C2—C3 | 117.46 (15) | C8—C7—H7 | 119.7 (12) |
| O2—C2—C3 | 126.54 (15) | C7—C8—H8 | 123.5 (12) |
| O1—C2—O2 | 116.00 (15) | C9—C8—H8 | 118.4 (12) |
| C4—C3—C11 | 117.93 (16) | N12—C13—H13 | 107.9 (11) |
| C2—C3—C4 | 119.80 (15) | C14—C13—H13 | 107.9 (11) |
| C2—C3—C11 | 122.24 (15) | C18—C13—H13 | 109.2 (12) |
| C3—C4—C10 | 121.39 (17) | C13—C14—H14A | 109.8 (11) |
| C6—C5—C10 | 120.09 (17) | C13—C14—H14B | 109.7 (11) |
| C5—C6—C7 | 120.18 (17) | C15—C14—H14A | 109.8 (11) |
| C6—C7—C8 | 121.20 (16) | C15—C14—H14B | 110.0 (11) |
| C7—C8—C9 | 118.05 (17) | H14A—C14—H14B | 107.3 (15) |
| O1—C9—C10 | 120.49 (14) | C14—C15—H15A | 107.4 (11) |
| O1—C9—C8 | 117.11 (16) | C14—C15—H15B | 109.6 (12) |
| C8—C9—C10 | 122.38 (16) | C16—C15—H15A | 109.6 (11) |
| C5—C10—C9 | 118.09 (15) | C16—C15—H15B | 111.5 (13) |
| C4—C10—C9 | 118.14 (16) | H15A—C15—H15B | 107.0 (16) |
| C4—C10—C5 | 123.75 (17) | C15—C16—H16A | 110.8 (11) |
| O11—C11—N12 | 122.51 (15) | C15—C16—H16B | 108.2 (11) |
| O11—C11—C3 | 120.27 (15) | C17—C16—H16A | 108.2 (11) |
| N12—C11—C3 | 117.21 (15) | C17—C16—H16B | 111.3 (11) |
| N12—C13—C14 | 110.73 (14) | H16A—C16—H16B | 107.0 (16) |
| C14—C13—C18 | 110.28 (14) | C16—C17—H17A | 110.4 (11) |
| N12—C13—C18 | 110.81 (13) | C16—C17—H17B | 110.5 (12) |
| C13—C14—C15 | 110.13 (15) | C18—C17—H17A | 108.0 (11) |
| C14—C15—C16 | 111.47 (15) | C18—C17—H17B | 108.9 (11) |
| C15—C16—C17 | 111.33 (15) | H17A—C17—H17B | 107.9 (16) |
| C16—C17—C18 | 111.00 (15) | C13—C18—H18A | 108.5 (11) |
| C13—C18—C17 | 110.71 (14) | C13—C18—H18B | 107.2 (11) |
| C3—C4—H4 | 120.7 (12) | C17—C18—H18A | 109.9 (11) |
| C10—C4—H4 | 117.9 (12) | C17—C18—H18B | 108.9 (11) |
| C6—C5—H5 | 122.8 (11) | H18A—C18—H18B | 111.6 (16) |
| | | |
| C9—O1—C2—O2 | −175.31 (15) | C6—C5—C10—C9 | 0.1 (2) |
| C9—O1—C2—C3 | 4.3 (2) | C10—C5—C6—C7 | 0.4 (3) |
| C2—O1—C9—C8 | 175.75 (15) | C6—C5—C10—C4 | 178.29 (16) |
| C2—O1—C9—C10 | −2.5 (2) | C5—C6—C7—C8 | −0.4 (3) |
| C13—N12—C11—O11 | −2.9 (2) | C6—C7—C8—C9 | −0.1 (3) |
| C13—N12—C11—C3 | 176.03 (14) | C7—C8—C9—O1 | −177.46 (15) |
| C11—N12—C13—C14 | 160.26 (14) | C7—C8—C9—C10 | 0.7 (3) |
| C11—N12—C13—C18 | −77.04 (19) | C8—C9—C10—C5 | −0.7 (3) |
| O1—C2—C3—C4 | −3.0 (2) | C8—C9—C10—C4 | −178.99 (16) |
| O1—C2—C3—C11 | 179.12 (14) | O1—C9—C10—C4 | −0.9 (2) |
| O2—C2—C3—C4 | 176.61 (17) | O1—C9—C10—C5 | 177.41 (15) |
| O2—C2—C3—C11 | −1.3 (3) | N12—C13—C14—C15 | −178.56 (14) |
| C2—C3—C11—N12 | 10.5 (2) | C14—C13—C18—C17 | −58.56 (18) |
| C4—C3—C11—O11 | 11.5 (2) | C18—C13—C14—C15 | 58.43 (18) |
| C2—C3—C11—O11 | −170.59 (16) | N12—C13—C18—C17 | 178.48 (14) |
| C2—C3—C4—C10 | −0.2 (2) | C13—C14—C15—C16 | −56.69 (19) |
| C11—C3—C4—C10 | 177.80 (15) | C14—C15—C16—C17 | 54.7 (2) |
| C4—C3—C11—N12 | −167.44 (15) | C15—C16—C17—C18 | −54.2 (2) |
| C3—C4—C10—C9 | 2.1 (2) | C16—C17—C18—C13 | 56.29 (19) |
| C3—C4—C10—C5 | −176.06 (16) | H12—N12—C11—O11 | 173.6 (15) |
| Symmetry codes: (i) −x, −y+1, −z; (ii) −x, −y, −z; (iii) −x+1, −y+1, −z; (iv) −x−1, −y, −z; (v) x−1, y, z; (vi) x+1, y, z; (vii) x, y+1, z+1; (viii) −x+1, −y+1, −z+1; (ix) x, y−1, z−1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N12—H12···O2 | 0.92 (2) | 1.99 (2) | 2.7426 (19) | 138.7 (18) |
| C4—H4···O11iii | 0.905 (19) | 2.510 (19) | 3.317 (2) | 148.7 (15) |
| C5—H5···O11iii | 0.953 (19) | 2.470 (19) | 3.319 (2) | 148.4 (15) |
| Symmetry code: (iii) −x+1, −y+1, −z. |

 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
 O contact to the O atom of one of the two carbonyl groups. The molecules form centrosymmetric dimers through self-complementary C—H
O contact to the O atom of one of the two carbonyl groups. The molecules form centrosymmetric dimers through self-complementary C—H O hydrogen-bonding interactions defining the R22(14)[R21(6)] motif. The supramolecular structure is achieved by aryl C—H
O hydrogen-bonding interactions defining the R22(14)[R21(6)] motif. The supramolecular structure is achieved by aryl C—H π interactions, as well as face-to-face π–π stacking interactions between lactone and benzenoid rings.
π interactions, as well as face-to-face π–π stacking interactions between lactone and benzenoid rings. ![[sigma]](//journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.003 Å
(C-C) = 0.003 Å
 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
 journal menu
journal menu














