The title compound,
catena-poly[[[dioxouranium(VI)]-bis(μ-2-hydroxybenzohydroxamato)] dihydrate], {[U(C
7H
6NO
3)
2O
2]·2H
2O}
n, is a uranyl coordination polymer based on the salicylhydroxamate ligand. The ligand acts both as a chelate, forming five-membered rings, and as a bridge between two U atoms. The coordination around each U atom is a distorted hexagonal bipyramid. Infinite chains running along [001] are formed
via bridging salicylhydroxamate O atoms. The chains have
C2 symmetry, with U atoms in special positions on twofold axes. The water molecules are not coordinated to the metal but crosslink, through hydrogen bonding, adjacent coordination polymer chains in the [110] and [1
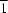
0] directions.
Supporting information
CCDC reference: 652492
Compound (I) was prepared from uranium(VI) perchlorate, 2-hydroxybenzohydroxamic
acid and sodium perchlorate. Stock solutions of uranium(VI) perchlorate,
perchloric acid and sodium perchlorate were obtained as already described
(Ciavatta et al., 1979). Commercial 2-hydroxybenzohydroxamic acid
(Sigma–Aldrich) was recrystallized twice from methanol–water mixtures
(Ciavatta et al., 2004). A solution of 0.005 mol dm-3
2-hydroxybenzohydroxamic acid, 0.002 mol dm-3 uranium(VI) perchlorate and 1 mol dm-3 sodium perchlorate was neutralized quantitatively by adding 0.05 mol dm-3 NaOH. Subsequently, the acidity of the solution was increased
slowly by addition of 0.02 mol dm-3 perchloric acid, until incipient
precipitation of an orange–red solid. All operations were performed in a
thermostatted vessel at 298.00 (5) K. The solutions were purged by a
continuous stream of pure nitrogen. The resulting solid, which developed as
aggregates of elongated prismatic crystals, was separated from the solution by
filtration, washed with 1 mol dm-3 sodium perchlorate then with water, and
finally dried under vacuum at room temperature. Compound (I) was identified by
IR spectroscopy and chemical and thermal analysis. An intense IR band at
914–930 cm-1 is observed, which is typical of the asymmetric O═U═O
uranyl stretching; typical amide C═O stretching at 1600 cm-1 is also
observed. The composition of the solid was determined by thermogravimetric
analysis in a nitrogen gas atmosphere, within the temperature range 323–1073 K, after which the transformation to U3O8 is complete. The total weight
loss (52.6%) and the water weight loss (5.5%) correspond to the formula
[UO2(C7H6NO3)2]·2H2O.
H atoms of the phenyl ring were generated stereochemically. All other H atoms
were located in a difference map. All H atoms were refined using a riding
model, with Uiso(H) = Ueq(carrier atom). The following
feature of the diffraction pattern of (I) has not escaped our attention. Since
the U atoms lie in special positions on twofold axes, their contribution to
hkl reflections with odd l is zero. However, uranium alone
accounts for a large fraction of the overall electronic density of the
compound (32%), so it follows that reflections of hkl layers with odd
l have intensities systematically lower than layers with even l.
This fraction of low-intensity reflections may account for the relatively high
Rint value.
Data collection: COLLECT (Nonius, 1999); cell refinement: DIRAX/LSQ (Duisenberg et al., 2000); data reduction: EVALCCD (Duisenberg et al., 2003); program(s) used to solve structure: SIR97 (Altomare et al., 1999); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: ORTEP-3 for Windows (Farrugia, 1997); software used to prepare material for publication: WinGX (Farrugia, 1999).
ii in Table 1.
-->
catena-poly[[[dioxouranium(VI)]-bis(µ-2-hydroxybenzohydroxamato)]
dihydrate]
top
Crystal data top
| [U(C7H6NO3)2O2]·2H2O | F(000) = 1144 |
| Mr = 610.32 | Dx = 2.251 Mg m−3 |
| Orthorhombic, Pccn | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 2ab 2ac | Cell parameters from 82 reflections |
| a = 12.649 (3) Å | θ = 3.2–21.1° |
| b = 16.258 (5) Å | µ = 9.07 mm−1 |
| c = 8.756 (3) Å | T = 295 K |
| V = 1800.6 (9) Å3 | Prism, orange |
| Z = 4 | 0.08 × 0.05 × 0.03 mm |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 2062 independent reflections |
| Radiation source: fine-focus sealed tube | 1122 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.139 |
| Detector resolution: 9 pixels mm-1 | θmax = 27.5°, θmin = 3.1° |
| CCD rotation images, thick slices scans | h = −15→16 |
Absorption correction: multi-scan
(SADABS; Bruker–Nonius, 2002) | k = −21→20 |
| Tmin = 0.520, Tmax = 0.762 | l = −11→9 |
| 11824 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.036 | Hydrogen site location: difference Fourier map |
| wR(F2) = 0.074 | H-atom parameters constrained |
| S = 1.00 | w = 1/[σ2(Fo2) + (0.0207P)2 + 0.355P]
where P = (Fo2 + 2Fc2)/3 |
| 2062 reflections | (Δ/σ)max < 0.001 |
| 123 parameters | Δρmax = 0.84 e Å−3 |
| 0 restraints | Δρmin = −1.24 e Å−3 |
Crystal data top
| [U(C7H6NO3)2O2]·2H2O | V = 1800.6 (9) Å3 |
| Mr = 610.32 | Z = 4 |
| Orthorhombic, Pccn | Mo Kα radiation |
| a = 12.649 (3) Å | µ = 9.07 mm−1 |
| b = 16.258 (5) Å | T = 295 K |
| c = 8.756 (3) Å | 0.08 × 0.05 × 0.03 mm |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 2062 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker–Nonius, 2002) | 1122 reflections with I > 2σ(I) |
| Tmin = 0.520, Tmax = 0.762 | Rint = 0.139 |
| 11824 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.036 | 0 restraints |
| wR(F2) = 0.074 | H-atom parameters constrained |
| S = 1.00 | Δρmax = 0.84 e Å−3 |
| 2062 reflections | Δρmin = −1.24 e Å−3 |
| 123 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell e.s.d.'s are taken
into account individually in the estimation of e.s.d.'s in distances, angles
and torsion angles; correlations between e.s.d.'s in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s.
planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor
wR and goodness of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The threshold expression of F2 >
σ(F2) is used only for calculating R-factors(gt) etc.
and is not relevant to the choice of reflections for refinement.
R-factors based on F2 are statistically about twice as large
as those based on F, and R- factors based on ALL data will be
even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| U1 | 0.2500 | 0.2500 | 0.34840 (4) | 0.01839 (11) | |
| O1 | 0.2298 (5) | 0.1790 (3) | 0.5953 (5) | 0.0252 (18) | |
| O2 | 0.1942 (5) | 0.1071 (4) | 0.3428 (6) | 0.0329 (15) | |
| O3 | 0.0999 (5) | −0.0259 (4) | 0.7240 (7) | 0.0371 (16) | |
| H3 | 0.0771 | −0.0477 | 0.8018 | 0.037* | |
| O4 | 0.1148 (4) | 0.2778 (4) | 0.3522 (6) | 0.0306 (15) | |
| O5 | 0.0340 (7) | 0.4054 (6) | 0.5740 (9) | 0.082 (3) | |
| H5A | 0.0442 | 0.3734 | 0.5057 | 0.082* | |
| H5B | 0.0706 | 0.3822 | 0.6421 | 0.082* | |
| N1 | 0.1881 (6) | 0.1030 (5) | 0.5942 (8) | 0.0335 (18) | |
| H1 | 0.1709 | 0.0793 | 0.6784 | 0.034* | |
| C1 | 0.1737 (6) | 0.0654 (5) | 0.4631 (9) | 0.0209 (18) | |
| C2 | 0.1413 (6) | −0.0197 (5) | 0.4609 (10) | 0.0238 (19) | |
| C3 | 0.1064 (7) | −0.0660 (5) | 0.5882 (10) | 0.029 (2) | |
| C4 | 0.0792 (8) | −0.1470 (6) | 0.5736 (11) | 0.044 (3) | |
| H4 | 0.0563 | −0.1761 | 0.6589 | 0.044* | |
| C5 | 0.0853 (9) | −0.1868 (7) | 0.4324 (13) | 0.053 (3) | |
| H5 | 0.0659 | −0.2418 | 0.4242 | 0.053* | |
| C6 | 0.1200 (9) | −0.1447 (7) | 0.3048 (13) | 0.057 (3) | |
| H6 | 0.1246 | −0.1711 | 0.2109 | 0.057* | |
| C7 | 0.1478 (8) | −0.0628 (6) | 0.3186 (10) | 0.037 (2) | |
| H7 | 0.1714 | −0.0348 | 0.2326 | 0.037* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| U1 | 0.02941 (19) | 0.01142 (17) | 0.01434 (16) | −0.0012 (5) | 0.000 | 0.000 |
| O1 | 0.044 (6) | 0.015 (3) | 0.017 (2) | −0.005 (3) | 0.002 (3) | 0.002 (2) |
| O2 | 0.065 (4) | 0.021 (4) | 0.013 (3) | −0.010 (3) | 0.005 (3) | 0.002 (3) |
| O3 | 0.057 (4) | 0.029 (4) | 0.025 (4) | −0.012 (3) | 0.008 (3) | 0.003 (3) |
| O4 | 0.035 (3) | 0.027 (4) | 0.030 (3) | 0.005 (2) | −0.004 (3) | 0.002 (2) |
| O5 | 0.102 (7) | 0.078 (8) | 0.067 (6) | 0.024 (6) | 0.030 (5) | −0.025 (5) |
| N1 | 0.045 (5) | 0.026 (5) | 0.029 (4) | −0.014 (4) | 0.006 (4) | 0.008 (4) |
| C1 | 0.030 (5) | 0.015 (5) | 0.018 (4) | 0.000 (4) | −0.006 (3) | −0.006 (4) |
| C2 | 0.025 (5) | 0.020 (5) | 0.026 (4) | 0.000 (4) | −0.006 (4) | −0.003 (4) |
| C3 | 0.035 (5) | 0.016 (5) | 0.036 (5) | −0.007 (4) | 0.005 (4) | 0.006 (4) |
| C4 | 0.066 (7) | 0.020 (6) | 0.045 (7) | −0.017 (5) | −0.009 (5) | 0.014 (4) |
| C5 | 0.071 (8) | 0.017 (6) | 0.069 (9) | −0.011 (6) | 0.000 (6) | −0.004 (5) |
| C6 | 0.085 (9) | 0.023 (7) | 0.064 (8) | −0.013 (6) | 0.005 (6) | −0.017 (5) |
| C7 | 0.058 (6) | 0.026 (6) | 0.028 (6) | −0.015 (5) | −0.002 (5) | 0.000 (4) |
Geometric parameters (Å, º) top
| U1—O4i | 1.769 (6) | O5—H5B | 0.8439 |
| U1—O4 | 1.769 (6) | N1—C1 | 1.313 (10) |
| U1—O2 | 2.428 (6) | N1—H1 | 0.8600 |
| U1—O2i | 2.428 (6) | C1—C2 | 1.444 (11) |
| U1—O1i | 2.464 (5) | C2—C3 | 1.415 (12) |
| U1—O1 | 2.464 (5) | C2—C7 | 1.431 (12) |
| U1—O1ii | 2.512 (5) | C3—C4 | 1.368 (12) |
| U1—O1iii | 2.512 (5) | C4—C5 | 1.397 (14) |
| O1—N1 | 1.343 (9) | C4—H4 | 0.9300 |
| O1—U1iv | 2.512 (5) | C5—C6 | 1.382 (15) |
| O2—C1 | 1.279 (10) | C5—H5 | 0.9300 |
| O3—C3 | 1.359 (11) | C6—C7 | 1.381 (14) |
| O3—H3 | 0.8197 | C6—H6 | 0.9300 |
| O5—H5A | 0.8038 | C7—H7 | 0.9300 |
| | | |
| O4i—U1—O4 | 177.8 (3) | N1—O1—U1iv | 118.0 (4) |
| O4i—U1—O2 | 92.1 (2) | U1—O1—U1iv | 123.2 (2) |
| O4—U1—O2 | 87.9 (2) | C1—O2—U1 | 123.4 (5) |
| O4i—U1—O2i | 87.9 (2) | C3—O3—H3 | 122.8 |
| O4—U1—O2i | 92.1 (2) | H5A—O5—H5B | 98.5 |
| O2—U1—O2i | 177.7 (3) | C1—N1—O1 | 119.3 (7) |
| O4i—U1—O1i | 90.2 (2) | C1—N1—H1 | 120.4 |
| O4—U1—O1i | 87.9 (2) | O1—N1—H1 | 120.4 |
| O2—U1—O1i | 119.74 (18) | O2—C1—N1 | 116.4 (8) |
| O2i—U1—O1i | 62.56 (18) | O2—C1—C2 | 123.7 (7) |
| O4i—U1—O1 | 87.9 (2) | N1—C1—C2 | 119.8 (8) |
| O4—U1—O1 | 90.2 (2) | C3—C2—C7 | 116.3 (8) |
| O2—U1—O1 | 62.56 (18) | C3—C2—C1 | 126.0 (8) |
| O2i—U1—O1 | 119.74 (18) | C7—C2—C1 | 117.7 (8) |
| O1i—U1—O1 | 57.4 (2) | O3—C3—C4 | 121.9 (8) |
| O4i—U1—O1ii | 78.5 (2) | O3—C3—C2 | 117.0 (8) |
| O4—U1—O1ii | 103.4 (2) | C4—C3—C2 | 121.1 (9) |
| O2—U1—O1ii | 64.66 (18) | C3—C4—C5 | 121.0 (9) |
| O2i—U1—O1ii | 113.12 (18) | C3—C4—H4 | 119.5 |
| O1i—U1—O1ii | 168.2 (3) | C5—C4—H4 | 119.5 |
| O1—U1—O1ii | 124.7 (2) | C6—C5—C4 | 120.2 (10) |
| O4i—U1—O1iii | 103.4 (2) | C6—C5—H5 | 119.9 |
| O4—U1—O1iii | 78.5 (2) | C4—C5—H5 | 119.9 |
| O2—U1—O1iii | 113.12 (18) | C7—C6—C5 | 119.2 (10) |
| O2i—U1—O1iii | 64.66 (18) | C7—C6—H6 | 120.4 |
| O1i—U1—O1iii | 124.7 (2) | C5—C6—H6 | 120.4 |
| O1—U1—O1iii | 168.2 (3) | C6—C7—C2 | 122.2 (9) |
| O1ii—U1—O1iii | 56.2 (2) | C6—C7—H7 | 118.9 |
| N1—O1—U1 | 117.7 (4) | C2—C7—H7 | 118.9 |
| | | |
| O4i—U1—O1—N1 | −100.6 (6) | U1—O1—N1—C1 | 9.5 (10) |
| O4—U1—O1—N1 | 80.5 (6) | U1iv—O1—N1—C1 | 178.0 (6) |
| O2—U1—O1—N1 | −7.1 (5) | U1—O2—C1—N1 | −2.4 (10) |
| O2i—U1—O1—N1 | 173.0 (5) | U1—O2—C1—C2 | −179.5 (6) |
| O1i—U1—O1—N1 | 167.9 (8) | O1—N1—C1—O2 | −4.8 (12) |
| O1ii—U1—O1—N1 | −26.1 (5) | O1—N1—C1—C2 | 172.4 (7) |
| O1iii—U1—O1—N1 | 64.0 (8) | O2—C1—C2—C3 | −172.4 (8) |
| O4i—U1—O1—U1iv | 91.6 (3) | N1—C1—C2—C3 | 10.6 (13) |
| O4—U1—O1—U1iv | −87.4 (3) | O2—C1—C2—C7 | 9.7 (12) |
| O2—U1—O1—U1iv | −175.0 (4) | N1—C1—C2—C7 | −167.3 (8) |
| O2i—U1—O1—U1iv | 5.1 (4) | C7—C2—C3—O3 | 180.0 (8) |
| O1i—U1—O1—U1iv | 0.0 | C1—C2—C3—O3 | 2.1 (13) |
| O1ii—U1—O1—U1iv | 166.1 (3) | C7—C2—C3—C4 | −0.9 (13) |
| O1iii—U1—O1—U1iv | −103.8 (5) | C1—C2—C3—C4 | −178.8 (9) |
| O4i—U1—O2—C1 | 91.7 (6) | O3—C3—C4—C5 | 179.2 (9) |
| O4—U1—O2—C1 | −86.2 (6) | C2—C3—C4—C5 | 0.2 (16) |
| O2i—U1—O2—C1 | −177.3 (6) | C3—C4—C5—C6 | 0.6 (17) |
| O1i—U1—O2—C1 | 0.2 (7) | C4—C5—C6—C7 | −0.5 (18) |
| O1—U1—O2—C1 | 5.1 (6) | C5—C6—C7—C2 | −0.3 (17) |
| O1ii—U1—O2—C1 | 167.9 (7) | C3—C2—C7—C6 | 1.0 (14) |
| O1iii—U1—O2—C1 | −162.8 (6) | C1—C2—C7—C6 | 179.1 (10) |
| Symmetry codes: (i) −x+1/2, −y+1/2, z; (ii) −x+1/2, y, z−1/2; (iii) x, −y+1/2, z−1/2; (iv) −x+1/2, y, z+1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O3 | 0.86 | 1.97 | 2.633 (10) | 133 |
| O3—H3···O5v | 0.82 | 1.93 | 2.691 (9) | 153 |
| O5—H5A···O4 | 0.80 | 2.24 | 3.020 (9) | 164 |
| O5—H5B···O2vi | 0.84 | 2.36 | 3.112 (10) | 149 |
| Symmetry codes: (v) −x, y−1/2, −z+3/2; (vi) x, −y+1/2, z+1/2. |
Experimental details
| Crystal data |
| Chemical formula | [U(C7H6NO3)2O2]·2H2O |
| Mr | 610.32 |
| Crystal system, space group | Orthorhombic, Pccn |
| Temperature (K) | 295 |
| a, b, c (Å) | 12.649 (3), 16.258 (5), 8.756 (3) |
| V (Å3) | 1800.6 (9) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 9.07 |
| Crystal size (mm) | 0.08 × 0.05 × 0.03 |
| |
| Data collection |
| Diffractometer | Bruker–Nonius KappaCCD
diffractometer |
| Absorption correction | Multi-scan
(SADABS; Bruker–Nonius, 2002) |
| Tmin, Tmax | 0.520, 0.762 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 11824, 2062, 1122 |
| Rint | 0.139 |
| (sin θ/λ)max (Å−1) | 0.650 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.036, 0.074, 1.00 |
| No. of reflections | 2062 |
| No. of parameters | 123 |
| H-atom treatment | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.84, −1.24 |
Selected geometric parameters (Å, º) top| U1—O4 | 1.769 (6) | O1—N1 | 1.343 (9) |
| U1—O2 | 2.428 (6) | O2—C1 | 1.279 (10) |
| U1—O1 | 2.464 (5) | N1—C1 | 1.313 (10) |
| U1—O1i | 2.512 (5) | | |
| | | |
| O4ii—U1—O4 | 177.8 (3) | O2—U1—O1i | 64.66 (18) |
| O4—U1—O2 | 87.9 (2) | O1—U1—O1i | 124.7 (2) |
| O2—U1—O2ii | 177.7 (3) | O4—U1—O1iii | 78.5 (2) |
| O4—U1—O1ii | 87.9 (2) | O2—U1—O1iii | 113.12 (18) |
| O2—U1—O1ii | 119.74 (18) | O1—U1—O1iii | 168.2 (3) |
| O4—U1—O1 | 90.2 (2) | O1i—U1—O1iii | 56.2 (2) |
| O2—U1—O1 | 62.56 (18) | N1—O1—U1 | 117.7 (4) |
| O1ii—U1—O1 | 57.4 (2) | N1—O1—U1iv | 118.0 (4) |
| O4—U1—O1i | 103.4 (2) | U1—O1—U1iv | 123.2 (2) |
| | | |
| O2—C1—C2—C3 | −172.4 (8) | | |
| Symmetry codes: (i) −x+1/2, y, z−1/2; (ii) −x+1/2, −y+1/2, z; (iii) x, −y+1/2, z−1/2; (iv) −x+1/2, y, z+1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O3 | 0.86 | 1.97 | 2.633 (10) | 132.6 |
| O3—H3···O5v | 0.82 | 1.93 | 2.691 (9) | 153.2 |
| O5—H5A···O4 | 0.80 | 2.24 | 3.020 (9) | 163.8 |
| O5—H5B···O2vi | 0.84 | 2.36 | 3.112 (10) | 149.0 |
| Symmetry codes: (v) −x, y−1/2, −z+3/2; (vi) x, −y+1/2, z+1/2. |

 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2007/06/00/ln3054/ln3054fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2007/06/00/ln3054/ln3054fig2thm.gif)




Actinide compounds show great structural variety and complexity, owing to the implication of 5f orbitals in the formation of chemical bonds, and in recent years they have been studied in the search for new materials with peculiar optical, magnetic and catalytic properties (Hutchings et al., 1996; Francis et al., 1998; Bean et al., 2001). In this context, a promising area is that of metal-organic coordination polymers built up by U—O units. For these materials, interesting electronic (e.g. semiconducting) and optical properties are foreseen (Chen et al., 2003). Up to now, only a few examples of uranyl coordination polymers have been reported. In some cases, the polymers contain an additional metal besides uranium (Chen et al., 2003; Yu et al., 2004) and, moreover, they are generally based on more than one type of ligand (Thuéry, 2007).
During a study of complex formation equilibria between metal ions and salicylhydroxamic acid, we succeeded in the synthesis of compound (I), [UO2(C7H6NO3)2]·2H2O, which is a rare example of a uranyl coordination polymer containing uranium as the only metal and based on only one type of ligand, 2-hydroxybenzohydroxamate (or salicylhydroxamate), the water molecules being, in fact, not coordinated to the metal.
The hydroxamate group occurs in natural products having biological activity (Kehl, 1982); some of them are inhibitors of enzymes such as peroxidases, metalloproteinases and ureases. The biological effects have been attributed to the affinity of the hydroxamate anion for metal ions. Actually, X-ray crystallographic studies have shown that the salicylhydroxamate group behaves as an O,O'-bidentate ligand, forming a five-membered chelate ring with several transition metal ions. All metal complexes of this ligand reported to date, however, have a mononuclear structure (Comman et al., 1992; Sharipov et al., 1983; Gao et al., 1998) and only one has a dinuclear structure (Stemmler et al., 1995). This report is the first in which the salicylhydroxamate ligand is involved in the formation of a coordination polymer.
The crystallographically independent unit of (I) contains one-half of a uranyl group, where atom U1 sits on a C2 axis, one salicylhydroxamate unit and one water molecule (Fig. 1). The conformation of the ligand shows, as expected, the 2-hydroxy group (C3/O3) and the C1—N1 bond in a syn conformation in order to establish an intramolecular hydrogen bond (Table 2). The carbonyl O atom is coordinated to one U atom only, while the alcoxy O atom acts as a slightly asymmetric bridge between two U atoms. The bond geometry around the alcoxy O atoms is substantially trigonal planar (Table 1).
The coordination environment of uranyl contains two chelate five-atom rings and four µ-O bridge atoms (Fig. 1). Each chelate ring is planar within 0.056 (4) Å, and the two rings and the U atom are coplanar within 0.101 (6) Å. This coordination can be described as a distorted hexagonal bipyramid, with axial O uranyl atoms (Cotton et al., 2005). The distortion is mainly due to the µ-O atoms being 0.588 (7) Å above and 0.588 (7) Å below the average plane of atom U1 and the two chelate rings.
In the crystal structure, the bridging of the U atoms by the salicylhydroxamate ligands leads to infinite chains running parallel to [001] (Figs. 1 and 2), whose direction coincides with the macroscopic elongation axis of the prismatic single crystals. The chains are formed, via µ-bridge alcoxy O atoms, through the two glide c planes and, because those glide planes are mutually perpendicular, the chains have overall C2 symmetry about the U1 atoms. Owing to the distorted coordination geometry around the U atom, adjacent chelate rings along the direction of the chain axis are not coplanar [dihedral angle 37.2 (1)°], thus releasing the overcrowding of phenyl groups.
The water molecules are not coordinated to the metal atoms; they are located between adjacent coordination polymer chains and cross-link them in the [110] and [1–10] directions, thereby contributing to the stability of the crystal packing by completing a three-dimensional framework. The water molecule is involved as an acceptor of a hydrogen bond from the 2-hydroxy group of a neighouring salicylhydroxamate ligand and as a donor in weaker hydrogen bonds with atoms O2 and O4 of symmetry-related units (Table 2).
Finally, we stress the relevant feature of the structure of (I) as compared with the few polymeric structures of uranyl reported to date, i.e. that all O atoms coordinated to the uranyl group belong to the organic ligand. Thus, in principle, it could be possible to influence the metal-to-ligand electronic system (and hence the spectroscopic properties) by introducing suitable substituents onto the aromatic ring (e.g. electron withdrawing groups, such as nitro or cyano, or electron releasing groups, such as dialkylamino).