The title molecule, C
21H
19NO
3, consists of a 2-ethoxyquinolyl group linked to an aroylvinyl group. The quinolyl ring forms a dihedral angle of 2.88 (6)° with the benzene ring. The crystal structure can be described by two types of crossed layers which are parallel to (110) and (1
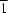
0). The packing is stabilized by C—H

O and O—H

O intra- and intermolecular hydrogen bonds, resulting in the formation of a two-dimensional network.
Supporting information
CCDC reference: 647269
Key indicators
- Single-crystal X-ray study
- T = 296 K
- Mean
![[sigma]](//journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.004 Å
(C-C) = 0.004 Å
- R factor = 0.033
- wR factor = 0.085
- Data-to-parameter ratio = 7.9
checkCIF/PLATON results
No syntax errors found
 Alert level C
PLAT089_ALERT_3_C Poor Data / Parameter Ratio (Zmax .LT. 18) ..... 7.92
PLAT128_ALERT_4_C Non-standard setting of Space group Pna21 .... Pn21a
Alert level C
PLAT089_ALERT_3_C Poor Data / Parameter Ratio (Zmax .LT. 18) ..... 7.92
PLAT128_ALERT_4_C Non-standard setting of Space group Pna21 .... Pn21a
 Alert level G
REFLT03_ALERT_4_G Please check that the estimate of the number of Friedel pairs is
correct. If it is not, please give the correct count in the
_publ_section_exptl_refinement section of the submitted CIF.
From the CIF: _diffrn_reflns_theta_max 26.32
From the CIF: _reflns_number_total 1869
Count of symmetry unique reflns 1872
Completeness (_total/calc) 99.84%
TEST3: Check Friedels for noncentro structure
Estimate of Friedel pairs measured 0
Fraction of Friedel pairs measured 0.000
Are heavy atom types Z>Si present no
Alert level G
REFLT03_ALERT_4_G Please check that the estimate of the number of Friedel pairs is
correct. If it is not, please give the correct count in the
_publ_section_exptl_refinement section of the submitted CIF.
From the CIF: _diffrn_reflns_theta_max 26.32
From the CIF: _reflns_number_total 1869
Count of symmetry unique reflns 1872
Completeness (_total/calc) 99.84%
TEST3: Check Friedels for noncentro structure
Estimate of Friedel pairs measured 0
Fraction of Friedel pairs measured 0.000
Are heavy atom types Z>Si present no
0 ALERT level A = In general: serious problem
0 ALERT level B = Potentially serious problem
2 ALERT level C = Check and explain
1 ALERT level G = General alerts; check
0 ALERT type 1 CIF construction/syntax error, inconsistent or missing data
0 ALERT type 2 Indicator that the structure model may be wrong or deficient
1 ALERT type 3 Indicator that the structure quality may be low
2 ALERT type 4 Improvement, methodology, query or suggestion
0 ALERT type 5 Informative message, check
Data collection: COLLECT (Nonius, 1998); cell refinement: SCALEPACK (Otwinowski & Minor, 1997); data reduction: SCALEPACK and DENZO (Otwinowski & Minor, 1997); program(s) used to solve structure: SIR2002 (Burla et al., 2003); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: ORTEP-3 (Farrugia,1997)and DIAMOND (Brandenburg
& Berndt, 2001); software used to prepare material for publication: WinGX (Farrugia, 1999).
(
E)-3-(2-Ethoxyquinolin-3-yl)-1-(2-hydroxy-6-methylphenyl)prop-2-en-1-one
top
Crystal data top
| C21H19NO3 | F(000) = 704 |
| Mr = 333.37 | Dx = 1.259 Mg m−3 |
| Orthorhombic, Pn21a | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P -2ac -2n | Cell parameters from 24253 reflections |
| a = 7.6026 (2) Å | θ = 2.8–26.3° |
| b = 14.7425 (4) Å | µ = 0.08 mm−1 |
| c = 15.6862 (4) Å | T = 296 K |
| V = 1758.13 (8) Å3 | Needle, yellow |
| Z = 4 | 0.15 × 0.11 × 0.06 mm |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 1348 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.055 |
| Graphite monochromator | θmax = 26.3°, θmin = 2.8° |
| φ scans, and ω scans with κ offsets | h = −9→9 |
| 24253 measured reflections | k = −18→18 |
| 1869 independent reflections | l = −19→18 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.033 | H-atom parameters constrained |
| wR(F2) = 0.085 | w = 1/[σ2(Fo2) + (0.0293P)2 + 1.3984P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.03 | (Δ/σ)max = 0.006 |
| 1869 reflections | Δρmax = 0.10 e Å−3 |
| 236 parameters | Δρmin = −0.12 e Å−3 |
| 1 restraint | Extinction correction: SHELXL97 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: none |
Special details top
Geometry. Bond distances, angles etc. have been calculated using the rounded
fractional coordinates. All su's are estimated from the variances of the
(full) variance-covariance matrix. The cell e.s.d.'s are taken into account in
the estimation of distances, angles and torsion angles |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor
wR and goodness of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The threshold expression of F2 >
σ(F2) is used only for calculating R-factors(gt) etc.
and is not relevant to the choice of reflections for refinement.
R-factors based on F2 are statistically about twice as large
as those based on F, and R- factors based on ALL data will be
even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O1 | 0.0711 (2) | 0.63718 (11) | 0.14351 (8) | 0.0521 (5) | |
| O2 | −0.0528 (3) | 0.50410 (13) | 0.44150 (10) | 0.0728 (7) | |
| O3 | −0.1870 (3) | 0.35064 (14) | 0.47616 (11) | 0.0856 (8) | |
| N1 | 0.1983 (3) | 0.77749 (13) | 0.13619 (11) | 0.0489 (6) | |
| C1 | 0.1395 (3) | 0.71106 (16) | 0.18284 (12) | 0.0434 (7) | |
| C2 | 0.1382 (3) | 0.70910 (16) | 0.27460 (11) | 0.0422 (6) | |
| C3 | 0.2078 (3) | 0.78397 (16) | 0.31388 (13) | 0.0468 (7) | |
| C4 | 0.2741 (3) | 0.85789 (15) | 0.26708 (14) | 0.0473 (7) | |
| C5 | 0.3456 (3) | 0.93651 (17) | 0.30510 (17) | 0.0591 (9) | |
| C6 | 0.4052 (3) | 1.00590 (18) | 0.2555 (2) | 0.0677 (10) | |
| C7 | 0.3977 (3) | 1.00029 (18) | 0.16672 (19) | 0.0682 (10) | |
| C8 | 0.3303 (3) | 0.92516 (17) | 0.12784 (18) | 0.0597 (9) | |
| C9 | 0.2666 (3) | 0.85209 (16) | 0.17705 (14) | 0.0479 (7) | |
| C10 | 0.0683 (3) | 0.63482 (17) | 0.32600 (13) | 0.0468 (7) | |
| C11 | −0.0018 (3) | 0.55650 (15) | 0.30215 (14) | 0.0503 (8) | |
| C12 | −0.0656 (3) | 0.48933 (16) | 0.36335 (14) | 0.0520 (8) | |
| C13 | −0.1439 (3) | 0.40414 (16) | 0.33213 (14) | 0.0496 (8) | |
| C14 | −0.1602 (3) | 0.38344 (16) | 0.24530 (15) | 0.0528 (8) | |
| C15 | −0.2252 (3) | 0.30192 (18) | 0.21596 (17) | 0.0631 (10) | |
| C16 | −0.2742 (4) | 0.2383 (2) | 0.2758 (2) | 0.0773 (13) | |
| C17 | −0.2609 (4) | 0.2549 (2) | 0.3612 (2) | 0.0800 (11) | |
| C18 | −0.1983 (3) | 0.33734 (19) | 0.39124 (17) | 0.0619 (9) | |
| C19 | −0.2380 (4) | 0.2816 (3) | 0.1222 (2) | 0.0893 (12) | |
| C20 | 0.0641 (3) | 0.6380 (2) | 0.05150 (12) | 0.0612 (9) | |
| C21 | −0.0297 (4) | 0.5533 (2) | 0.02490 (15) | 0.0750 (11) | |
| H3 | 0.21117 | 0.78601 | 0.37311 | 0.0561* | |
| H5 | 0.35195 | 0.94095 | 0.36416 | 0.0709* | |
| H6 | 0.45141 | 1.05758 | 0.28111 | 0.0812* | |
| H7 | 0.43898 | 1.04822 | 0.13378 | 0.0817* | |
| H8 | 0.32637 | 0.92210 | 0.06865 | 0.0717* | |
| H10 | 0.07380 | 0.64387 | 0.38461 | 0.0561* | |
| H11 | −0.01084 | 0.54379 | 0.24424 | 0.0603* | |
| H13A | 0.18200 | 0.63905 | 0.02788 | 0.0734* | |
| H13B | 0.00118 | 0.69108 | 0.03149 | 0.0734* | |
| H14 | −0.12556 | 0.42674 | 0.20559 | 0.0634* | |
| H16 | −0.31741 | 0.18252 | 0.25748 | 0.0926* | |
| H17 | −0.29436 | 0.21031 | 0.39984 | 0.0960* | |
| H19A | −0.23530 | 0.33734 | 0.09054 | 0.1344* | |
| H19B | −0.34623 | 0.25031 | 0.11074 | 0.1344* | |
| H19C | −0.14067 | 0.24418 | 0.10533 | 0.1344* | |
| H21A | −0.03752 | 0.55129 | −0.03616 | 0.1123* | |
| H21B | −0.14595 | 0.55302 | 0.04883 | 0.1123* | |
| H21C | 0.03406 | 0.50132 | 0.04495 | 0.1123* | |
| H33 | −0.14849 | 0.40163 | 0.48555 | 0.1284* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O1 | 0.0711 (9) | 0.0490 (9) | 0.0361 (7) | −0.0058 (8) | −0.0016 (7) | −0.0024 (7) |
| O2 | 0.1039 (14) | 0.0721 (12) | 0.0424 (9) | −0.0109 (10) | −0.0007 (9) | 0.0080 (8) |
| O3 | 0.1050 (14) | 0.0858 (14) | 0.0661 (12) | −0.0122 (12) | 0.0142 (10) | 0.0260 (11) |
| N1 | 0.0548 (10) | 0.0481 (11) | 0.0439 (9) | 0.0050 (9) | 0.0009 (9) | 0.0049 (9) |
| C1 | 0.0460 (12) | 0.0428 (12) | 0.0414 (10) | 0.0062 (10) | −0.0016 (9) | −0.0015 (11) |
| C2 | 0.0451 (12) | 0.0407 (11) | 0.0407 (10) | 0.0083 (9) | 0.0001 (9) | 0.0007 (10) |
| C3 | 0.0502 (12) | 0.0484 (13) | 0.0417 (11) | 0.0092 (10) | −0.0033 (10) | −0.0034 (10) |
| C4 | 0.0426 (12) | 0.0420 (13) | 0.0574 (13) | 0.0057 (10) | −0.0003 (10) | −0.0032 (11) |
| C5 | 0.0517 (14) | 0.0528 (16) | 0.0728 (16) | 0.0015 (12) | −0.0072 (12) | −0.0078 (12) |
| C6 | 0.0546 (14) | 0.0484 (15) | 0.100 (2) | −0.0031 (12) | −0.0056 (14) | −0.0041 (14) |
| C7 | 0.0561 (15) | 0.0515 (16) | 0.097 (2) | −0.0004 (13) | 0.0033 (13) | 0.0178 (15) |
| C8 | 0.0582 (14) | 0.0515 (16) | 0.0695 (15) | 0.0071 (12) | 0.0038 (12) | 0.0127 (12) |
| C9 | 0.0444 (12) | 0.0437 (13) | 0.0555 (12) | 0.0093 (10) | −0.0017 (10) | 0.0066 (11) |
| C10 | 0.0519 (12) | 0.0502 (14) | 0.0383 (10) | 0.0068 (12) | 0.0005 (9) | 0.0028 (10) |
| C11 | 0.0639 (14) | 0.0461 (14) | 0.0408 (11) | −0.0024 (12) | 0.0016 (10) | 0.0027 (10) |
| C12 | 0.0581 (14) | 0.0523 (15) | 0.0455 (12) | 0.0067 (12) | 0.0022 (10) | 0.0081 (10) |
| C13 | 0.0481 (13) | 0.0457 (13) | 0.0551 (14) | 0.0055 (11) | 0.0070 (10) | 0.0085 (10) |
| C14 | 0.0510 (12) | 0.0497 (15) | 0.0577 (14) | 0.0017 (12) | 0.0068 (10) | 0.0017 (11) |
| C15 | 0.0459 (14) | 0.0591 (18) | 0.0842 (19) | 0.0015 (12) | 0.0056 (12) | −0.0146 (15) |
| C16 | 0.0586 (17) | 0.0544 (17) | 0.119 (3) | −0.0049 (13) | 0.0114 (16) | −0.0084 (18) |
| C17 | 0.0700 (19) | 0.0600 (19) | 0.110 (2) | −0.0077 (14) | 0.0194 (16) | 0.0198 (19) |
| C18 | 0.0592 (15) | 0.0585 (17) | 0.0679 (16) | 0.0033 (13) | 0.0104 (13) | 0.0178 (13) |
| C19 | 0.075 (2) | 0.098 (2) | 0.095 (2) | −0.0014 (18) | 0.0004 (15) | −0.0370 (19) |
| C20 | 0.0790 (17) | 0.0703 (17) | 0.0343 (11) | 0.0017 (14) | −0.0012 (10) | −0.0031 (12) |
| C21 | 0.095 (2) | 0.078 (2) | 0.0519 (16) | −0.0002 (18) | −0.0067 (12) | −0.0168 (14) |
Geometric parameters (Å, º) top
| O1—C1 | 1.356 (3) | C15—C16 | 1.378 (4) |
| O1—C20 | 1.444 (2) | C15—C19 | 1.504 (4) |
| O2—C12 | 1.249 (3) | C16—C17 | 1.366 (4) |
| O3—C18 | 1.349 (3) | C17—C18 | 1.388 (4) |
| O3—H33 | 0.8200 | C20—C21 | 1.497 (4) |
| N1—C1 | 1.302 (3) | C3—H3 | 0.9300 |
| N1—C9 | 1.375 (3) | C5—H5 | 0.9300 |
| C1—C2 | 1.440 (3) | C6—H6 | 0.9300 |
| C2—C3 | 1.370 (3) | C7—H7 | 0.9300 |
| C2—C10 | 1.460 (3) | C8—H8 | 0.9300 |
| C3—C4 | 1.407 (3) | C10—H10 | 0.9300 |
| C4—C5 | 1.412 (3) | C11—H11 | 0.9300 |
| C4—C9 | 1.416 (3) | C14—H14 | 0.9300 |
| C5—C6 | 1.363 (4) | C16—H16 | 0.9300 |
| C6—C7 | 1.396 (4) | C17—H17 | 0.9300 |
| C7—C8 | 1.364 (4) | C19—H19A | 0.9600 |
| C8—C9 | 1.411 (3) | C19—H19B | 0.9600 |
| C10—C11 | 1.326 (3) | C19—H19C | 0.9600 |
| C11—C12 | 1.462 (3) | C20—H13A | 0.9700 |
| C12—C13 | 1.474 (3) | C20—H13B | 0.9700 |
| C13—C14 | 1.401 (3) | C21—H21A | 0.9600 |
| C13—C18 | 1.414 (4) | C21—H21B | 0.9600 |
| C14—C15 | 1.379 (4) | C21—H21C | 0.9600 |
| | | |
| O1···C11 | 2.813 (3) | C12···H33 | 2.4000 |
| O2···O3 | 2.541 (3) | C14···H11 | 2.6200 |
| O3···O2 | 2.541 (3) | C17···H19Cii | 2.9400 |
| O1···H11 | 2.1900 | C18···H19Biv | 2.9700 |
| O2···H10 | 2.4400 | C20···H19Cv | 2.9700 |
| O2···H8i | 2.9000 | H3···H5 | 2.5300 |
| O2···H13Aii | 2.8700 | H3···H10 | 2.3500 |
| O2···H33 | 1.8100 | H3···O3vi | 2.5600 |
| O3···H3iii | 2.5600 | H5···H3 | 2.5300 |
| N1···H13A | 2.6600 | H6···H16vii | 2.5700 |
| N1···H13B | 2.5600 | H8···O2viii | 2.9000 |
| C1···C11iv | 3.562 (3) | H10···O2 | 2.4400 |
| C1···C3ii | 3.454 (3) | H10···H3 | 2.3500 |
| C1···C10iv | 3.451 (3) | H11···O1 | 2.1900 |
| C2···C4ii | 3.592 (3) | H11···C1 | 2.8800 |
| C3···C9ii | 3.504 (3) | H11···C14 | 2.6200 |
| C3···C1iv | 3.454 (3) | H11···H14 | 2.0300 |
| C4···C2iv | 3.592 (3) | H13A···N1 | 2.6600 |
| C4···C6ii | 3.571 (3) | H13A···O2iv | 2.8700 |
| C5···C9iv | 3.446 (3) | H13B···N1 | 2.5600 |
| C5···C7ii | 3.560 (3) | H13B···H19Cv | 2.5200 |
| C6···C4iv | 3.571 (3) | H14···C11 | 2.6200 |
| C7···C5iv | 3.560 (3) | H14···H11 | 2.0300 |
| C9···C5ii | 3.446 (3) | H14···H19A | 2.3900 |
| C9···C3iv | 3.504 (3) | H16···H6ix | 2.5700 |
| C10···C1ii | 3.451 (3) | H16···H19B | 2.5200 |
| C11···C1ii | 3.562 (3) | H19A···H14 | 2.3900 |
| C11···O1 | 2.813 (3) | H19B···H16 | 2.5200 |
| C14···C15iv | 3.571 (3) | H19B···C18ii | 2.9700 |
| C15···C16iv | 3.557 (4) | H19C···C20x | 2.9700 |
| C15···C14ii | 3.571 (3) | H19C···H13Bx | 2.5200 |
| C16···C15ii | 3.557 (4) | H19C···C17iv | 2.9400 |
| C1···H11 | 2.8800 | H33···O2 | 1.8100 |
| C11···H14 | 2.6200 | H33···C12 | 2.4000 |
| | | |
| C1—O1—C20 | 117.53 (18) | C2—C3—H3 | 119.00 |
| C18—O3—H33 | 109.00 | C4—C3—H3 | 119.00 |
| C1—N1—C9 | 118.00 (18) | C4—C5—H5 | 120.00 |
| O1—C1—N1 | 118.71 (17) | C6—C5—H5 | 120.00 |
| N1—C1—C2 | 125.4 (2) | C5—C6—H6 | 120.00 |
| O1—C1—C2 | 115.87 (19) | C7—C6—H6 | 120.00 |
| C1—C2—C3 | 115.5 (2) | C6—C7—H7 | 120.00 |
| C3—C2—C10 | 119.76 (17) | C8—C7—H7 | 120.00 |
| C1—C2—C10 | 124.7 (2) | C7—C8—H8 | 120.00 |
| C2—C3—C4 | 121.83 (19) | C9—C8—H8 | 120.00 |
| C3—C4—C9 | 117.3 (2) | C2—C10—H10 | 115.00 |
| C5—C4—C9 | 119.1 (2) | C11—C10—H10 | 115.00 |
| C3—C4—C5 | 123.6 (2) | C10—C11—H11 | 119.00 |
| C4—C5—C6 | 120.2 (2) | C12—C11—H11 | 119.00 |
| C5—C6—C7 | 120.8 (2) | C13—C14—H14 | 118.00 |
| C6—C7—C8 | 120.6 (3) | C15—C14—H14 | 118.00 |
| C7—C8—C9 | 120.3 (3) | C15—C16—H16 | 119.00 |
| N1—C9—C8 | 119.0 (2) | C17—C16—H16 | 119.00 |
| C4—C9—C8 | 119.1 (2) | C16—C17—H17 | 120.00 |
| N1—C9—C4 | 121.9 (2) | C18—C17—H17 | 119.00 |
| C2—C10—C11 | 130.1 (2) | C15—C19—H19A | 110.00 |
| C10—C11—C12 | 122.6 (2) | C15—C19—H19B | 110.00 |
| O2—C12—C11 | 120.0 (2) | C15—C19—H19C | 110.00 |
| C11—C12—C13 | 119.54 (19) | H19A—C19—H19B | 109.00 |
| O2—C12—C13 | 120.4 (2) | H19A—C19—H19C | 109.00 |
| C12—C13—C18 | 119.6 (2) | H19B—C19—H19C | 109.00 |
| C14—C13—C18 | 117.4 (2) | O1—C20—H13A | 110.00 |
| C12—C13—C14 | 123.0 (2) | O1—C20—H13B | 110.00 |
| C13—C14—C15 | 123.1 (2) | C21—C20—H13A | 110.00 |
| C14—C15—C16 | 117.6 (2) | C21—C20—H13B | 110.00 |
| C14—C15—C19 | 121.6 (3) | H13A—C20—H13B | 109.00 |
| C16—C15—C19 | 120.9 (3) | C20—C21—H21A | 109.00 |
| C15—C16—C17 | 121.8 (3) | C20—C21—H21B | 109.00 |
| C16—C17—C18 | 121.0 (3) | C20—C21—H21C | 110.00 |
| O3—C18—C13 | 121.8 (2) | H21A—C21—H21B | 109.00 |
| C13—C18—C17 | 119.2 (2) | H21A—C21—H21C | 109.00 |
| O3—C18—C17 | 119.0 (3) | H21B—C21—H21C | 109.00 |
| O1—C20—C21 | 106.80 (19) | | |
| | | |
| C20—O1—C1—N1 | −1.5 (3) | C5—C6—C7—C8 | 0.1 (4) |
| C20—O1—C1—C2 | 177.77 (19) | C6—C7—C8—C9 | 0.2 (4) |
| C1—O1—C20—C21 | −175.3 (2) | C7—C8—C9—C4 | −0.1 (3) |
| C9—N1—C1—O1 | 179.7 (2) | C7—C8—C9—N1 | 179.6 (2) |
| C9—N1—C1—C2 | 0.6 (4) | C2—C10—C11—C12 | 180.0 (2) |
| C1—N1—C9—C8 | −179.7 (2) | C10—C11—C12—O2 | −1.1 (4) |
| C1—N1—C9—C4 | 0.0 (4) | C10—C11—C12—C13 | 179.3 (2) |
| O1—C1—C2—C10 | −0.5 (3) | O2—C12—C13—C14 | −179.4 (2) |
| N1—C1—C2—C3 | −0.9 (4) | O2—C12—C13—C18 | −2.5 (3) |
| O1—C1—C2—C3 | 179.9 (2) | C11—C12—C13—C14 | 0.3 (3) |
| N1—C1—C2—C10 | 178.6 (2) | C11—C12—C13—C18 | 177.1 (2) |
| C10—C2—C3—C4 | −178.9 (2) | C12—C13—C14—C15 | 176.6 (2) |
| C1—C2—C3—C4 | 0.6 (3) | C18—C13—C14—C15 | −0.3 (3) |
| C3—C2—C10—C11 | −178.9 (2) | C12—C13—C18—O3 | 2.6 (4) |
| C1—C2—C10—C11 | 1.6 (4) | C12—C13—C18—C17 | −175.7 (2) |
| C2—C3—C4—C9 | −0.1 (3) | C14—C13—C18—O3 | 179.7 (2) |
| C2—C3—C4—C5 | 179.5 (2) | C14—C13—C18—C17 | 1.3 (3) |
| C3—C4—C5—C6 | −179.1 (2) | C13—C14—C15—C16 | −0.6 (4) |
| C3—C4—C9—C8 | 179.4 (2) | C13—C14—C15—C19 | −179.0 (2) |
| C9—C4—C5—C6 | 0.5 (3) | C14—C15—C16—C17 | 0.6 (4) |
| C3—C4—C9—N1 | −0.3 (3) | C19—C15—C16—C17 | 178.9 (3) |
| C5—C4—C9—C8 | −0.2 (3) | C15—C16—C17—C18 | 0.5 (5) |
| C5—C4—C9—N1 | −179.9 (2) | C16—C17—C18—O3 | −179.8 (3) |
| C4—C5—C6—C7 | −0.4 (4) | C16—C17—C18—C13 | −1.5 (4) |
| Symmetry codes: (i) −x+1/2, y−1/2, z+1/2; (ii) x−1/2, y, −z+1/2; (iii) −x, y−1/2, −z+1; (iv) x+1/2, y, −z+1/2; (v) −x, y+1/2, −z; (vi) −x, y+1/2, −z+1; (vii) x+1, y+1, z; (viii) −x+1/2, y+1/2, z−1/2; (ix) x−1, y−1, z; (x) −x, y−1/2, −z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O3—H33···O2 | 0.8200 | 1.8100 | 2.541 (3) | 147.00 |
| C3—H3···O3vi | 0.9300 | 2.5600 | 3.441 (3) | 159.00 |
| C10—H10···O2 | 0.9300 | 2.4400 | 2.801 (3) | 103.00 |
| C11—H11···O1 | 0.9300 | 2.1900 | 2.813 (3) | 124.00 |
| Symmetry code: (vi) −x, y+1/2, −z+1. |
 O and O—H
O and O—H O intra- and intermolecular hydrogen bonds, resulting in the formation of a two-dimensional network.
O intra- and intermolecular hydrogen bonds, resulting in the formation of a two-dimensional network.
 journal menu
journal menu











 Alert level C
Alert level C
 Alert level G
Alert level G



