Buy article online - an online subscription or single-article purchase is required to access this article.
The structures of the hexafluoridoiridates(IV) of calcium, Ca[IrF
6]·2H
2O [calcium hexafluoridoiridate(IV) dihydrate], strontium, Sr[IrF
6]·2H
2O [strontium hexafluoridoiridate(IV) dihydrate], and barium, Ba[IrF
6] [barium hexafluoridoiridate(IV)], have been determined by single-crystal X-ray analysis. The first two compounds are isomorphous. Their metal cations are eight-coordinated in a distorted square-antiprismatic coordination environment, and their anions are represented by an almost ideal octahedron. These two structures can be described as frameworks in which all atoms occupy general positions. Sr[RhF
6] and Ba[RhF
6] have a different space group (
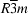
, from powder diffraction data) but similar cell dimensions. The structures are very close to that of Ba[IrF
6]. The cation is in a cuboctahedral coordination. The metal atoms are located on special positions of
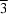
symmetry, while the F atoms are in general positions.
Supporting information
The title compounds were synthesized by the following method. Firstly, K2[IrCl6] was obtained by reaction between IrCl4·4H2O dissolved in concentrated HCl and KCl solution in the ratio K:Ir = 2:1. In the second stage, K2[IrF6] was obtained by the interaction of K2[IrCl6] with gaseous F2 at atmospheric pressure and 573 K in a flow reactor. An aqueous solution of K2[IrF6] was then treated with ion-exchange resin in the H form for one day. The mixture was filtered and a solution of H2[IrF6] acid was collected. Concentration by evaporation at room temperature yielded a solution of 0.11 M. Ca[IrF6]·2H2O, Sr[IrF6]·2H2O and Ba[IrF6] were prepared by the reaction between 0.11 M H2[IrF6] (5 ml) and the corresponding reagent, CaO (56 mg), SrCO3 (74 mg) or BaCO3 (99 mg). Crystals suitable for X-ray analysis were obtained by slow evaporation at room temperature.
The H atoms were located in difference electron-density maps and refined with O—H distances restrained to 0.82 (5) Å and H···H distances restrained to 1.30 (5) Å (to obtain H—O—H angles of about 105°).
For all compounds, data collection: APEX2 (Bruker, 2004); cell refinement: SAINT (Bruker, 2004); data reduction: SAINT (Bruker, 2004); program(s) used to solve structure: SHELXTL (Bruker, 2004); program(s) used to refine structure: SHELXTL (Bruker, 2004); molecular graphics: BS (Ozawa & Kang, 2004); software used to prepare material for publication: SHELXTL (Bruker, 2004).
(I) calcium hexafluoroiridate(IV) dihydrate
top
Crystal data top
| Ca[IrF6]·2H2O | F(000) = 684 |
| Mr = 382.31 | Dx = 4.148 Mg m−3 |
| Monoclinic, P21/n | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 2yn | Cell parameters from 3691 reflections |
| a = 5.9055 (2) Å | θ = 3.7–30.0° |
| b = 9.5369 (3) Å | µ = 22.72 mm−1 |
| c = 11.0140 (3) Å | T = 296 K |
| β = 99.262 (1)° | Needle, light pink |
| V = 612.22 (3) Å3 | 0.10 × 0.06 × 0.04 mm |
| Z = 4 | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 1790 independent reflections |
| Radiation source: fine-focus sealed tube | 1688 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.028 |
| Detector resolution: 25 pixels mm-1 | θmax = 30.0°, θmin = 2.8° |
| ϕ scans | h = −7→8 |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | k = −11→13 |
| Tmin = 0.210, Tmax = 0.464 | l = −11→15 |
| 5426 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.021 | Hydrogen site location: difference Fourier map |
| wR(F2) = 0.056 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.25 | w = 1/[σ2(Fo2) + (0.0253P)2]
where P = (Fo2 + 2Fc2)/3 |
| 1790 reflections | (Δ/σ)max = 0.001 |
| 107 parameters | Δρmax = 0.94 e Å−3 |
| 6 restraints | Δρmin = −2.84 e Å−3 |
Crystal data top
| Ca[IrF6]·2H2O | V = 612.22 (3) Å3 |
| Mr = 382.31 | Z = 4 |
| Monoclinic, P21/n | Mo Kα radiation |
| a = 5.9055 (2) Å | µ = 22.72 mm−1 |
| b = 9.5369 (3) Å | T = 296 K |
| c = 11.0140 (3) Å | 0.10 × 0.06 × 0.04 mm |
| β = 99.262 (1)° | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 1790 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | 1688 reflections with I > 2σ(I) |
| Tmin = 0.210, Tmax = 0.464 | Rint = 0.028 |
| 5426 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.021 | 6 restraints |
| wR(F2) = 0.056 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.25 | Δρmax = 0.94 e Å−3 |
| 1790 reflections | Δρmin = −2.84 e Å−3 |
| 107 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Ir1 | 0.40499 (2) | 0.539327 (15) | 0.265398 (13) | 0.00857 (7) | |
| Ca1 | 0.66063 (14) | 0.18588 (9) | 0.45306 (8) | 0.01119 (16) | |
| F2 | 0.6581 (4) | 0.6440 (3) | 0.2159 (2) | 0.0184 (5) | |
| F1 | 0.6254 (4) | 0.4059 (3) | 0.3455 (2) | 0.0199 (5) | |
| F4 | 0.1480 (5) | 0.4431 (3) | 0.3160 (3) | 0.0211 (6) | |
| F3 | 0.1815 (4) | 0.6693 (3) | 0.1806 (2) | 0.0198 (5) | |
| F6 | 0.3794 (5) | 0.4293 (3) | 0.1160 (2) | 0.0208 (6) | |
| F5 | 0.4356 (5) | 0.6502 (3) | 0.4133 (2) | 0.0210 (6) | |
| O1 | 1.0353 (6) | 0.3009 (4) | 0.5137 (4) | 0.0247 (7) | |
| O2 | 0.3779 (6) | 0.0763 (4) | 0.5856 (3) | 0.0167 (6) | |
| H1A | 1.065 (11) | 0.362 (6) | 0.563 (5) | 0.04 (2)* | |
| H1B | 1.095 (13) | 0.328 (8) | 0.457 (5) | 0.07 (3)* | |
| H2A | 0.280 (8) | 0.136 (5) | 0.561 (4) | 0.015 (14)* | |
| H2B | 0.423 (10) | 0.092 (7) | 0.655 (4) | 0.033 (18)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ir1 | 0.01050 (9) | 0.00778 (10) | 0.00746 (10) | −0.00010 (5) | 0.00158 (6) | −0.00031 (4) |
| Ca1 | 0.0131 (4) | 0.0109 (4) | 0.0095 (4) | 0.0003 (3) | 0.0015 (3) | 0.0000 (3) |
| F2 | 0.0194 (12) | 0.0188 (14) | 0.0191 (13) | −0.0046 (10) | 0.0097 (11) | 0.0033 (10) |
| F1 | 0.0208 (12) | 0.0166 (13) | 0.0208 (14) | 0.0043 (11) | −0.0010 (10) | 0.0061 (11) |
| F4 | 0.0193 (13) | 0.0226 (15) | 0.0225 (15) | −0.0081 (11) | 0.0063 (11) | 0.0044 (11) |
| F3 | 0.0184 (12) | 0.0140 (12) | 0.0248 (14) | 0.0045 (10) | −0.0037 (11) | 0.0032 (10) |
| F6 | 0.0231 (13) | 0.0231 (14) | 0.0151 (14) | −0.0004 (11) | −0.0004 (11) | −0.0100 (11) |
| F5 | 0.0288 (14) | 0.0217 (14) | 0.0139 (13) | −0.0008 (11) | 0.0078 (11) | −0.0099 (10) |
| O1 | 0.0251 (17) | 0.0260 (19) | 0.0227 (19) | −0.0078 (15) | 0.0031 (15) | 0.0036 (16) |
| O2 | 0.0175 (15) | 0.0173 (16) | 0.0142 (16) | 0.0036 (13) | −0.0009 (12) | −0.0008 (13) |
Geometric parameters (Å, º) top
| Ir1—F5 | 1.926 (2) | Ca1—O2 | 2.607 (3) |
| Ir1—F1 | 1.929 (3) | Ca1—Ca1v | 4.2273 (17) |
| Ir1—F4 | 1.931 (3) | Ca1—H2A | 2.75 (5) |
| Ir1—F6 | 1.937 (3) | F2—Ca1vi | 2.326 (3) |
| Ir1—F3 | 1.937 (2) | F3—Ca1vii | 2.306 (3) |
| Ir1—F2 | 1.947 (2) | F6—Ca1viii | 2.313 (3) |
| Ca1—F5i | 2.281 (3) | F5—Ca1i | 2.281 (3) |
| Ca1—F3ii | 2.306 (3) | O1—H1A | 0.79 (4) |
| Ca1—F6iii | 2.313 (3) | O1—H1B | 0.81 (4) |
| Ca1—F2iv | 2.326 (3) | O2—Ca1v | 2.540 (4) |
| Ca1—F1 | 2.402 (3) | O2—H2A | 0.83 (4) |
| Ca1—O1 | 2.464 (4) | O2—H2B | 0.78 (4) |
| Ca1—O2v | 2.540 (4) | | |
| | | |
| F5—Ir1—F1 | 90.62 (12) | F3ii—Ca1—O2 | 75.88 (10) |
| F5—Ir1—F4 | 89.31 (12) | F6iii—Ca1—O2 | 72.72 (10) |
| F1—Ir1—F4 | 93.33 (12) | F2iv—Ca1—O2 | 142.93 (11) |
| F5—Ir1—F6 | 178.94 (11) | F1—Ca1—O2 | 127.87 (10) |
| F1—Ir1—F6 | 89.19 (12) | O1—Ca1—O2 | 130.91 (12) |
| F4—Ir1—F6 | 91.74 (13) | O2v—Ca1—O2 | 69.58 (12) |
| F5—Ir1—F3 | 91.15 (11) | F5i—Ca1—Ca1v | 104.40 (8) |
| F1—Ir1—F3 | 178.21 (12) | F3ii—Ca1—Ca1v | 73.57 (7) |
| F4—Ir1—F3 | 86.39 (12) | F6iii—Ca1—Ca1v | 68.19 (7) |
| F6—Ir1—F3 | 89.05 (12) | F2iv—Ca1—Ca1v | 109.52 (7) |
| F5—Ir1—F2 | 89.05 (11) | F1—Ca1—Ca1v | 147.84 (7) |
| F1—Ir1—F2 | 88.54 (11) | O1—Ca1—Ca1v | 136.56 (10) |
| F4—Ir1—F2 | 177.53 (11) | O2v—Ca1—Ca1v | 35.31 (7) |
| F6—Ir1—F2 | 89.90 (12) | O2—Ca1—Ca1v | 34.28 (8) |
| F3—Ir1—F2 | 91.79 (11) | F5i—Ca1—H2A | 62.2 (11) |
| F5i—Ca1—F3ii | 100.10 (10) | F3ii—Ca1—H2A | 64.6 (9) |
| F5i—Ca1—F6iii | 89.47 (11) | F6iii—Ca1—H2A | 88.6 (8) |
| F3ii—Ca1—F6iii | 141.76 (11) | F2iv—Ca1—H2A | 146.1 (11) |
| F5i—Ca1—F2iv | 145.96 (10) | F1—Ca1—H2A | 110.7 (8) |
| F3ii—Ca1—F2iv | 86.90 (10) | O1—Ca1—H2A | 136.2 (10) |
| F6iii—Ca1—F2iv | 105.53 (10) | O2v—Ca1—H2A | 81.1 (10) |
| F5i—Ca1—F1 | 73.07 (10) | O2—Ca1—H2A | 17.5 (8) |
| F3ii—Ca1—F1 | 75.37 (10) | Ca1v—Ca1—H2A | 47.0 (10) |
| F6iii—Ca1—F1 | 142.25 (11) | Ir1—F2—Ca1vi | 141.72 (14) |
| F2iv—Ca1—F1 | 76.70 (9) | Ir1—F1—Ca1 | 143.17 (14) |
| F5i—Ca1—O1 | 79.99 (12) | Ir1—F3—Ca1vii | 144.14 (14) |
| F3ii—Ca1—O1 | 149.31 (12) | Ir1—F6—Ca1viii | 150.91 (15) |
| F6iii—Ca1—O1 | 68.66 (11) | Ir1—F5—Ca1i | 157.35 (15) |
| F2iv—Ca1—O1 | 77.55 (12) | Ca1—O1—H1A | 127 (5) |
| F1—Ca1—O1 | 75.37 (11) | Ca1—O1—H1B | 114 (5) |
| F5i—Ca1—O2v | 139.27 (11) | H1A—O1—H1B | 103 (6) |
| F3ii—Ca1—O2v | 77.27 (11) | Ca1v—O2—Ca1 | 110.42 (12) |
| F6iii—Ca1—O2v | 71.74 (11) | Ca1v—O2—H2A | 126 (4) |
| F2iv—Ca1—O2v | 74.76 (10) | Ca1—O2—H2A | 91 (4) |
| F1—Ca1—O2v | 141.14 (11) | Ca1v—O2—H2B | 111 (5) |
| O1—Ca1—O2v | 122.42 (12) | Ca1—O2—H2B | 109 (5) |
| F5i—Ca1—O2 | 70.41 (10) | H2A—O2—H2B | 107 (5) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+1/2, y−1/2, −z+1/2; (iii) x+1/2, −y+1/2, z+1/2; (iv) −x+3/2, y−1/2, −z+1/2; (v) −x+1, −y, −z+1; (vi) −x+3/2, y+1/2, −z+1/2; (vii) −x+1/2, y+1/2, −z+1/2; (viii) x−1/2, −y+1/2, z−1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1B···F4ix | 0.81 (4) | 1.96 (5) | 2.736 (4) | 159 (8) |
| O2—H2B···F4iii | 0.78 (4) | 2.07 (4) | 2.777 (5) | 152 (6) |
| O2—H2A···O1x | 0.83 (4) | 2.14 (4) | 2.966 (5) | 175 (5) |
| Symmetry codes: (iii) x+1/2, −y+1/2, z+1/2; (ix) x+1, y, z; (x) x−1, y, z. |
(II) strontium hexafluoroiridate(IV) dihydrate
top
Crystal data top
| Sr[IrF6]·2H2O | F(000) = 756 |
| Mr = 429.85 | Dx = 4.354 Mg m−3 |
| Monoclinic, P21/n | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 2yn | Cell parameters from 3998 reflections |
| a = 6.0328 (2) Å | θ = 2.8–30.0° |
| b = 9.8264 (4) Å | µ = 28.47 mm−1 |
| c = 11.2123 (5) Å | T = 296 K |
| β = 99.363 (1)° | Needle, light pink |
| V = 655.82 (5) Å3 | 0.18 × 0.08 × 0.04 mm |
| Z = 4 | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 1895 independent reflections |
| Radiation source: fine-focus sealed tube | 1753 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.018 |
| Detector resolution: 25 pixels mm-1 | θmax = 30.0°, θmin = 2.8° |
| ϕ scans | h = −4→8 |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | k = −13→13 |
| Tmin = 0.080, Tmax = 0.336 | l = −15→15 |
| 5732 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.019 | Hydrogen site location: difference Fourier map |
| wR(F2) = 0.053 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.12 | w = 1/[σ2(Fo2) + (0.0251P)2 + 3.0724P]
where P = (Fo2 + 2Fc2)/3 |
| 1895 reflections | (Δ/σ)max = 0.003 |
| 107 parameters | Δρmax = 2.33 e Å−3 |
| 6 restraints | Δρmin = −1.25 e Å−3 |
Crystal data top
| Sr[IrF6]·2H2O | V = 655.82 (5) Å3 |
| Mr = 429.85 | Z = 4 |
| Monoclinic, P21/n | Mo Kα radiation |
| a = 6.0328 (2) Å | µ = 28.47 mm−1 |
| b = 9.8264 (4) Å | T = 296 K |
| c = 11.2123 (5) Å | 0.18 × 0.08 × 0.04 mm |
| β = 99.363 (1)° | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 1895 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | 1753 reflections with I > 2σ(I) |
| Tmin = 0.080, Tmax = 0.336 | Rint = 0.018 |
| 5732 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.019 | 6 restraints |
| wR(F2) = 0.053 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.12 | Δρmax = 2.33 e Å−3 |
| 1895 reflections | Δρmin = −1.25 e Å−3 |
| 107 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Ir1 | 0.40999 (3) | 0.536839 (17) | 0.267134 (15) | 0.01472 (6) | |
| Sr1 | 0.66440 (7) | 0.18841 (4) | 0.45219 (4) | 0.01520 (9) | |
| F2 | 0.6486 (5) | 0.6473 (3) | 0.2231 (3) | 0.0263 (7) | |
| F1 | 0.6320 (6) | 0.4104 (3) | 0.3390 (3) | 0.0269 (7) | |
| F4 | 0.1699 (6) | 0.4326 (3) | 0.3126 (3) | 0.0291 (7) | |
| F3 | 0.1867 (6) | 0.6603 (3) | 0.1904 (3) | 0.0319 (7) | |
| F6 | 0.3908 (6) | 0.4395 (4) | 0.1175 (3) | 0.0287 (7) | |
| F5 | 0.4344 (6) | 0.6358 (3) | 0.4146 (3) | 0.0290 (7) | |
| O1 | 1.0533 (8) | 0.3064 (5) | 0.5159 (4) | 0.0328 (9) | |
| O2 | 0.3823 (6) | 0.0811 (4) | 0.5915 (4) | 0.0205 (7) | |
| H1B | 1.095 (11) | 0.344 (6) | 0.459 (4) | 0.022 (16)* | |
| H2A | 0.263 (7) | 0.118 (6) | 0.580 (5) | 0.016 (15)* | |
| H2B | 0.426 (9) | 0.089 (6) | 0.656 (4) | 0.011 (14)* | |
| H1A | 1.016 (16) | 0.364 (7) | 0.557 (7) | 0.06 (3)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ir1 | 0.01644 (10) | 0.01326 (9) | 0.01493 (9) | −0.00037 (6) | 0.00393 (6) | −0.00003 (6) |
| Sr1 | 0.01645 (19) | 0.01469 (19) | 0.01464 (18) | −0.00055 (14) | 0.00308 (14) | 0.00093 (14) |
| F2 | 0.0276 (15) | 0.0283 (16) | 0.0258 (16) | −0.0048 (13) | 0.0123 (13) | 0.0043 (12) |
| F1 | 0.0275 (16) | 0.0221 (15) | 0.0294 (16) | 0.0040 (12) | −0.0003 (13) | 0.0072 (13) |
| F4 | 0.0280 (16) | 0.0305 (16) | 0.0305 (17) | −0.0123 (13) | 0.0097 (13) | 0.0018 (13) |
| F3 | 0.0254 (16) | 0.0227 (15) | 0.044 (2) | 0.0043 (13) | −0.0048 (14) | 0.0040 (14) |
| F6 | 0.0291 (17) | 0.0340 (17) | 0.0216 (15) | −0.0007 (14) | 0.0000 (13) | −0.0124 (13) |
| F5 | 0.0351 (18) | 0.0279 (16) | 0.0264 (16) | −0.0042 (14) | 0.0121 (13) | −0.0124 (13) |
| O1 | 0.027 (2) | 0.040 (2) | 0.030 (2) | −0.0099 (18) | 0.0001 (17) | 0.0052 (19) |
| O2 | 0.0157 (16) | 0.0228 (17) | 0.0230 (18) | 0.0007 (13) | 0.0032 (14) | 0.0015 (14) |
Geometric parameters (Å, º) top
| Ir1—F5 | 1.903 (3) | Sr1—O2v | 2.700 (4) |
| Ir1—F1 | 1.907 (3) | Sr1—O2 | 2.704 (4) |
| Ir1—F4 | 1.909 (3) | Sr1—Sr1v | 4.4143 (8) |
| Ir1—F3 | 1.910 (3) | F2—Sr1vi | 2.454 (3) |
| Ir1—F6 | 1.919 (3) | F3—Sr1vii | 2.453 (3) |
| Ir1—F2 | 1.930 (3) | F6—Sr1viii | 2.461 (3) |
| Sr1—F5i | 2.421 (3) | F5—Sr1i | 2.421 (3) |
| Sr1—F2ii | 2.454 (3) | O1—H1B | 0.81 (4) |
| Sr1—F3iii | 2.453 (3) | O1—H1A | 0.79 (4) |
| Sr1—F6iv | 2.461 (3) | O2—Sr1v | 2.700 (4) |
| Sr1—F1 | 2.515 (3) | O2—H2A | 0.80 (4) |
| Sr1—O1 | 2.610 (4) | O2—H2B | 0.73 (4) |
| | | |
| F5—Ir1—F1 | 90.91 (15) | F6iv—Sr1—O2v | 70.39 (12) |
| F5—Ir1—F4 | 89.84 (15) | F1—Sr1—O2v | 139.65 (12) |
| F1—Ir1—F4 | 92.79 (15) | O1—Sr1—O2v | 123.23 (14) |
| F5—Ir1—F3 | 90.93 (15) | F5i—Sr1—O2 | 71.37 (11) |
| F1—Ir1—F3 | 178.15 (15) | F2ii—Sr1—O2 | 144.39 (11) |
| F4—Ir1—F3 | 87.20 (15) | F3iii—Sr1—O2 | 76.96 (12) |
| F5—Ir1—F6 | 178.70 (15) | F6iv—Sr1—O2 | 71.96 (11) |
| F1—Ir1—F6 | 89.03 (15) | F1—Sr1—O2 | 128.68 (11) |
| F4—Ir1—F6 | 91.45 (15) | O1—Sr1—O2 | 129.60 (13) |
| F3—Ir1—F6 | 89.13 (16) | O2v—Sr1—O2 | 70.45 (13) |
| F5—Ir1—F2 | 88.80 (14) | F5i—Sr1—Sr1v | 106.45 (8) |
| F1—Ir1—F2 | 88.44 (14) | F2ii—Sr1—Sr1v | 109.86 (8) |
| F4—Ir1—F2 | 178.18 (15) | F3iii—Sr1—Sr1v | 71.89 (8) |
| F3—Ir1—F2 | 91.62 (14) | F6iv—Sr1—Sr1v | 66.74 (8) |
| F6—Ir1—F2 | 89.90 (15) | F1—Sr1—Sr1v | 148.09 (8) |
| F5i—Sr1—F2ii | 143.44 (11) | O1—Sr1—Sr1v | 136.52 (10) |
| F5i—Sr1—F3iii | 102.07 (12) | O2v—Sr1—Sr1v | 35.26 (8) |
| F2ii—Sr1—F3iii | 85.43 (12) | O2—Sr1—Sr1v | 35.19 (8) |
| F5i—Sr1—F6iv | 93.55 (12) | Ir1—F2—Sr1vi | 139.16 (16) |
| F2ii—Sr1—F6iv | 104.37 (11) | Ir1—F1—Sr1 | 140.36 (17) |
| F3iii—Sr1—F6iv | 138.41 (12) | Ir1—F3—Sr1vii | 146.71 (18) |
| F5i—Sr1—F1 | 71.68 (12) | Ir1—F6—Sr1viii | 149.37 (18) |
| F2ii—Sr1—F1 | 75.33 (11) | Ir1—F5—Sr1i | 155.32 (19) |
| F3iii—Sr1—F1 | 77.34 (11) | Sr1—O1—H1B | 112 (5) |
| F6iv—Sr1—F1 | 144.19 (12) | Sr1—O1—H1A | 99 (7) |
| F5i—Sr1—O1 | 79.32 (14) | H1B—O1—H1A | 106 (6) |
| F2ii—Sr1—O1 | 77.44 (14) | Sr1v—O2—Sr1 | 109.56 (13) |
| F3iii—Sr1—O1 | 150.55 (13) | Sr1v—O2—H2A | 111 (4) |
| F6iv—Sr1—O1 | 69.93 (13) | Sr1—O2—H2A | 111 (4) |
| F1—Sr1—O1 | 75.25 (12) | Sr1v—O2—H2B | 107 (5) |
| F5i—Sr1—O2v | 141.54 (11) | Sr1—O2—H2B | 112 (5) |
| F2ii—Sr1—O2v | 75.00 (11) | H2A—O2—H2B | 106 (5) |
| F3iii—Sr1—O2v | 73.61 (12) | | |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+3/2, y−1/2, −z+1/2; (iii) −x+1/2, y−1/2, −z+1/2; (iv) x+1/2, −y+1/2, z+1/2; (v) −x+1, −y, −z+1; (vi) −x+3/2, y+1/2, −z+1/2; (vii) −x+1/2, y+1/2, −z+1/2; (viii) x−1/2, −y+1/2, z−1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1B···F4ix | 0.81 (4) | 1.97 (4) | 2.784 (6) | 175 (7) |
| O2—H2B···F4iv | 0.73 (4) | 2.11 (4) | 2.788 (5) | 154 (6) |
| O2—H2A···O1x | 0.80 (4) | 2.29 (5) | 3.002 (6) | 149 (6) |
| Symmetry codes: (iv) x+1/2, −y+1/2, z+1/2; (ix) x+1, y, z; (x) x−1, y, z. |
(III) barium hexafluoroiridate(IV)
top
Crystal data top
| Ba[IrF6] | Dx = 6.404 Mg m−3 |
| Mr = 443.54 | Mo Kα radiation, λ = 0.71073 Å |
| Trigonal, R3 | Cell parameters from 967 reflections |
| Hall symbol: -R 3 | θ = 4.2–30.0° |
| a = 7.3965 (5) Å | µ = 37.44 mm−1 |
| c = 7.2826 (7) Å | T = 293 K |
| V = 345.04 (5) Å3 | Needle, light pink |
| Z = 3 | 0.12 × 0.04 × 0.04 mm |
| F(000) = 561 | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 182 independent reflections |
| Radiation source: fine-focus sealed tube | 182 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.037 |
| Detector resolution: 25 pixels mm-1 | θmax = 27.4°, θmin = 4.2° |
| ϕ scans | h = −9→9 |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | k = −7→9 |
| Tmin = 0.178, Tmax = 0.216 | l = −9→9 |
| 889 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.024 | w = 1/[σ2(Fo2) + (0.0408P)2 + 6.1383P]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.066 | (Δ/σ)max < 0.001 |
| S = 1.22 | Δρmax = 1.91 e Å−3 |
| 182 reflections | Δρmin = −2.46 e Å−3 |
| 15 parameters | Extinction correction: SHELXTL (Bruker, 2004), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.0108 (11) |
Crystal data top
| Ba[IrF6] | Z = 3 |
| Mr = 443.54 | Mo Kα radiation |
| Trigonal, R3 | µ = 37.44 mm−1 |
| a = 7.3965 (5) Å | T = 293 K |
| c = 7.2826 (7) Å | 0.12 × 0.04 × 0.04 mm |
| V = 345.04 (5) Å3 | |
Data collection top
Bruker Nonius X8 APEX CCD area-detector
diffractometer | 182 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker, 2004) | 182 reflections with I > 2σ(I) |
| Tmin = 0.178, Tmax = 0.216 | Rint = 0.037 |
| 889 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.024 | 15 parameters |
| wR(F2) = 0.066 | 0 restraints |
| S = 1.22 | Δρmax = 1.91 e Å−3 |
| 182 reflections | Δρmin = −2.46 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Ir1 | 0.0000 | 0.0000 | 0.0000 | 0.0074 (4) | |
| F1 | 0.0729 (6) | 0.2325 (6) | 0.1640 (5) | 0.0154 (10) | |
| Ba1 | 0.3333 | 0.6667 | 0.1667 | 0.0099 (4) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ir1 | 0.0064 (4) | 0.0064 (4) | 0.0093 (5) | 0.0032 (2) | 0.000 | 0.000 |
| F1 | 0.017 (2) | 0.0127 (19) | 0.015 (2) | 0.0068 (16) | −0.0006 (14) | −0.0042 (15) |
| Ba1 | 0.0092 (5) | 0.0092 (5) | 0.0112 (6) | 0.0046 (2) | 0.000 | 0.000 |
Geometric parameters (Å, º) top
| Ir1—F1i | 1.935 (4) | Ba1—F1ix | 2.800 (4) |
| Ir1—F1 | 1.935 (4) | Ba1—F1x | 2.800 (4) |
| Ir1—F1ii | 1.935 (4) | Ba1—F1xi | 2.800 (4) |
| Ir1—F1iii | 1.935 (4) | Ba1—F1xii | 2.800 (4) |
| Ir1—F1iv | 1.935 (4) | Ba1—F1xiii | 2.883 (4) |
| Ir1—F1v | 1.935 (4) | Ba1—F1xiv | 2.883 (4) |
| Ir1—Ba1vi | 3.6413 (4) | Ba1—F1xv | 2.883 (4) |
| Ir1—Ba1vii | 3.6413 (4) | Ba1—F1xvi | 2.883 (4) |
| Ba1—F1 | 2.800 (4) | Ba1—F1xvii | 2.883 (4) |
| F1—Ba1vii | 2.883 (4) | Ba1—F1xviii | 2.883 (4) |
| Ba1—F1viii | 2.800 (4) | | |
| | | |
| F1i—Ir1—F1 | 180.0 (3) | F1xi—Ba1—F1xiii | 119.63 (13) |
| F1i—Ir1—F1ii | 85.93 (18) | F1—Ba1—F1xiii | 96.28 (14) |
| F1—Ir1—F1ii | 94.07 (18) | F1xii—Ba1—F1xiii | 113.77 (12) |
| F1i—Ir1—F1iii | 94.07 (18) | F1viii—Ba1—F1xiv | 96.28 (14) |
| F1—Ir1—F1iii | 85.93 (18) | F1ix—Ba1—F1xiv | 119.63 (13) |
| F1ii—Ir1—F1iii | 180.0 (3) | F1x—Ba1—F1xiv | 66.23 (12) |
| F1i—Ir1—F1iv | 94.07 (18) | F1xi—Ba1—F1xiv | 83.72 (14) |
| F1—Ir1—F1iv | 85.93 (18) | F1—Ba1—F1xiv | 113.77 (12) |
| F1ii—Ir1—F1iv | 94.07 (18) | F1xii—Ba1—F1xiv | 60.37 (13) |
| F1iii—Ir1—F1iv | 85.93 (18) | F1xiii—Ba1—F1xiv | 54.47 (12) |
| F1i—Ir1—F1v | 85.93 (18) | F1viii—Ba1—F1xv | 113.77 (12) |
| F1—Ir1—F1v | 94.07 (18) | F1ix—Ba1—F1xv | 83.72 (14) |
| F1ii—Ir1—F1v | 85.93 (18) | F1x—Ba1—F1xv | 119.63 (13) |
| F1iii—Ir1—F1v | 94.07 (18) | F1xi—Ba1—F1xv | 66.23 (12) |
| F1iv—Ir1—F1v | 180.0 (5) | F1—Ba1—F1xv | 60.37 (13) |
| F1i—Ir1—Ba1vi | 51.91 (12) | F1xii—Ba1—F1xv | 96.28 (14) |
| F1—Ir1—Ba1vi | 128.09 (12) | F1xiii—Ba1—F1xv | 54.47 (12) |
| F1ii—Ir1—Ba1vi | 51.91 (12) | F1xiv—Ba1—F1xv | 54.47 (12) |
| F1iii—Ir1—Ba1vi | 128.09 (12) | F1viii—Ba1—F1xvi | 66.23 (12) |
| F1iv—Ir1—Ba1vi | 128.09 (12) | F1ix—Ba1—F1xvi | 96.28 (14) |
| F1v—Ir1—Ba1vi | 51.91 (12) | F1x—Ba1—F1xvi | 60.37 (13) |
| F1i—Ir1—Ba1vii | 128.09 (12) | F1xi—Ba1—F1xvi | 113.77 (12) |
| F1—Ir1—Ba1vii | 51.91 (12) | F1—Ba1—F1xvi | 119.63 (13) |
| F1ii—Ir1—Ba1vii | 128.09 (12) | F1xii—Ba1—F1xvi | 83.72 (14) |
| F1iii—Ir1—Ba1vii | 51.91 (12) | F1xiii—Ba1—F1xvi | 125.53 (12) |
| F1iv—Ir1—Ba1vii | 51.91 (12) | F1xiv—Ba1—F1xvi | 125.53 (13) |
| F1v—Ir1—Ba1vii | 128.09 (12) | F1xv—Ba1—F1xvi | 180.0 |
| Ba1vi—Ir1—Ba1vii | 180.0 | F1viii—Ba1—F1xvii | 83.72 (14) |
| Ir1—F1—Ba1 | 138.56 (19) | F1ix—Ba1—F1xvii | 60.37 (13) |
| Ir1—F1—Ba1vii | 96.20 (15) | F1x—Ba1—F1xvii | 113.77 (12) |
| Ba1—F1—Ba1vii | 119.63 (13) | F1xi—Ba1—F1xvii | 96.28 (14) |
| F1viii—Ba1—F1ix | 60.004 (3) | F1—Ba1—F1xvii | 66.23 (12) |
| F1viii—Ba1—F1x | 60.004 (3) | F1xii—Ba1—F1xvii | 119.63 (13) |
| F1ix—Ba1—F1x | 119.994 (2) | F1xiii—Ba1—F1xvii | 125.53 (12) |
| F1viii—Ba1—F1xi | 180.0 | F1xiv—Ba1—F1xvii | 180.0 |
| F1ix—Ba1—F1xi | 119.996 (3) | F1xv—Ba1—F1xvii | 125.53 (13) |
| F1x—Ba1—F1xi | 119.996 (3) | F1xvi—Ba1—F1xvii | 54.47 (12) |
| F1viii—Ba1—F1 | 119.995 (3) | F1viii—Ba1—F1xviii | 119.63 (13) |
| F1ix—Ba1—F1 | 60.005 (3) | F1ix—Ba1—F1xviii | 113.77 (12) |
| F1x—Ba1—F1 | 180.0 | F1x—Ba1—F1xviii | 96.28 (14) |
| F1xi—Ba1—F1 | 60.005 (2) | F1xi—Ba1—F1xviii | 60.37 (13) |
| F1viii—Ba1—F1xii | 119.995 (3) | F1—Ba1—F1xviii | 83.72 (14) |
| F1ix—Ba1—F1xii | 180.0 | F1xii—Ba1—F1xviii | 66.23 (12) |
| F1x—Ba1—F1xii | 60.006 (2) | F1xiii—Ba1—F1xviii | 180.0 |
| F1xi—Ba1—F1xii | 60.005 (3) | F1xiv—Ba1—F1xviii | 125.53 (12) |
| F1—Ba1—F1xii | 119.995 (2) | F1xv—Ba1—F1xviii | 125.53 (12) |
| F1viii—Ba1—F1xiii | 60.37 (13) | F1xvi—Ba1—F1xviii | 54.47 (12) |
| F1ix—Ba1—F1xiii | 66.23 (12) | F1xvii—Ba1—F1xviii | 54.47 (12) |
| F1x—Ba1—F1xiii | 83.72 (14) | | |
| Symmetry codes: (i) −x, −y, −z; (ii) x−y, x, −z; (iii) −x+y, −x, z; (iv) −y, x−y, z; (v) y, −x+y, −z; (vi) x−1/3, y−2/3, z−2/3; (vii) x−1/3, y−2/3, z+1/3; (viii) −x+y, −x+1, z; (ix) y−1/3, −x+y+1/3, −z+1/3; (x) −x+2/3, −y+4/3, −z+1/3; (xi) x−y+2/3, x+1/3, −z+1/3; (xii) −y+1, x−y+1, z; (xiii) x−y+1/3, x+2/3, −z+2/3; (xiv) y+1/3, −x+y+2/3, −z+2/3; (xv) −x+1/3, −y+2/3, −z+2/3; (xvi) x+1/3, y+2/3, z−1/3; (xvii) −y+1/3, x−y+2/3, z−1/3; (xviii) −x+y+1/3, −x+2/3, z−1/3. |
Experimental details
| | (I) | (II) | (III) |
| Crystal data |
| Chemical formula | Ca[IrF6]·2H2O | Sr[IrF6]·2H2O | Ba[IrF6] |
| Mr | 382.31 | 429.85 | 443.54 |
| Crystal system, space group | Monoclinic, P21/n | Monoclinic, P21/n | Trigonal, R3 |
| Temperature (K) | 296 | 296 | 293 |
| a, b, c (Å) | 5.9055 (2), 9.5369 (3), 11.0140 (3) | 6.0328 (2), 9.8264 (4), 11.2123 (5) | 7.3965 (5), 7.3965 (5), 7.2826 (7) |
| α, β, γ (°) | 90, 99.262 (1), 90 | 90, 99.363 (1), 90 | 90, 90, 120 |
| V (Å3) | 612.22 (3) | 655.82 (5) | 345.04 (5) |
| Z | 4 | 4 | 3 |
| Radiation type | Mo Kα | Mo Kα | Mo Kα |
| µ (mm−1) | 22.72 | 28.47 | 37.44 |
| Crystal size (mm) | 0.10 × 0.06 × 0.04 | 0.18 × 0.08 × 0.04 | 0.12 × 0.04 × 0.04 |
| |
| Data collection |
| Diffractometer | Bruker Nonius X8 APEX CCD area-detector
diffractometer | Bruker Nonius X8 APEX CCD area-detector
diffractometer | Bruker Nonius X8 APEX CCD area-detector
diffractometer |
| Absorption correction | Multi-scan
(SADABS; Bruker, 2004) | Multi-scan
(SADABS; Bruker, 2004) | Multi-scan
(SADABS; Bruker, 2004) |
| Tmin, Tmax | 0.210, 0.464 | 0.080, 0.336 | 0.178, 0.216 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 5426, 1790, 1688 | 5732, 1895, 1753 | 889, 182, 182 |
| Rint | 0.028 | 0.018 | 0.037 |
| (sin θ/λ)max (Å−1) | 0.704 | 0.703 | 0.648 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.021, 0.056, 1.25 | 0.019, 0.053, 1.12 | 0.024, 0.066, 1.22 |
| No. of reflections | 1790 | 1895 | 182 |
| No. of parameters | 107 | 107 | 15 |
| No. of restraints | 6 | 6 | 0 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement | H atoms treated by a mixture of independent and constrained refinement | – |
| Δρmax, Δρmin (e Å−3) | 0.94, −2.84 | 2.33, −1.25 | 1.91, −2.46 |
Selected bond lengths (Å) for (I) top| Ir1—F5 | 1.926 (2) | Ca1—F3ii | 2.306 (3) |
| Ir1—F1 | 1.929 (3) | Ca1—F6iii | 2.313 (3) |
| Ir1—F4 | 1.931 (3) | Ca1—F2iv | 2.326 (3) |
| Ir1—F6 | 1.937 (3) | Ca1—F1 | 2.402 (3) |
| Ir1—F3 | 1.937 (2) | Ca1—O1 | 2.464 (4) |
| Ir1—F2 | 1.947 (2) | Ca1—O2v | 2.540 (4) |
| Ca1—F5i | 2.281 (3) | Ca1—O2 | 2.607 (3) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+1/2, y−1/2, −z+1/2; (iii) x+1/2, −y+1/2, z+1/2; (iv) −x+3/2, y−1/2, −z+1/2; (v) −x+1, −y, −z+1. |
Hydrogen-bond geometry (Å, º) for (I) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1B···F4vi | 0.81 (4) | 1.96 (5) | 2.736 (4) | 159 (8) |
| O2—H2B···F4iii | 0.78 (4) | 2.07 (4) | 2.777 (5) | 152 (6) |
| O2—H2A···O1vii | 0.83 (4) | 2.14 (4) | 2.966 (5) | 175 (5) |
| Symmetry codes: (iii) x+1/2, −y+1/2, z+1/2; (vi) x+1, y, z; (vii) x−1, y, z. |
Selected bond lengths (Å) for (II) top| Ir1—F5 | 1.903 (3) | Sr1—F2ii | 2.454 (3) |
| Ir1—F1 | 1.907 (3) | Sr1—F3iii | 2.453 (3) |
| Ir1—F4 | 1.909 (3) | Sr1—F6iv | 2.461 (3) |
| Ir1—F3 | 1.910 (3) | Sr1—F1 | 2.515 (3) |
| Ir1—F6 | 1.919 (3) | Sr1—O1 | 2.610 (4) |
| Ir1—F2 | 1.930 (3) | Sr1—O2v | 2.700 (4) |
| Sr1—F5i | 2.421 (3) | Sr1—O2 | 2.704 (4) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x+3/2, y−1/2, −z+1/2; (iii) −x+1/2, y−1/2, −z+1/2; (iv) x+1/2, −y+1/2, z+1/2; (v) −x+1, −y, −z+1. |
Hydrogen-bond geometry (Å, º) for (II) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1B···F4vi | 0.81 (4) | 1.97 (4) | 2.784 (6) | 175 (7) |
| O2—H2B···F4iv | 0.73 (4) | 2.11 (4) | 2.788 (5) | 154 (6) |
| O2—H2A···O1vii | 0.80 (4) | 2.29 (5) | 3.002 (6) | 149 (6) |
| Symmetry codes: (iv) x+1/2, −y+1/2, z+1/2; (vi) x+1, y, z; (vii) x−1, y, z. |
Selected bond lengths (Å) for (III) top| Ir1—F1 | 1.935 (4) | Ba1—F1 | 2.800 (4) |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2007/11/00/iz3025/iz3025fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2007/11/00/iz3025/iz3025fig2thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



Fluorine-containing complexes of noble metals attract attention in various areas of chemistry, such as chemical analysis, electrochemistry and applied chemistry (Mitkin, 2001). In particular, this concerns hexafluorometallates. However, structure determination using single-crystal X-ray analysis for many of these compounds has not been carried out yet. Among the complexes of IrIV, several have been characterized by X-ray powder diffraction (Babel, 1967) but only one structure solved by the single-crystal method (K2[IrF6]; Fitz et al., 2002) is currently available. A new contribution to the crystal chemistry of these compounds seemed useful. We focused our attention on the investigation of alkali and alkaline earth metal hexafluoroiridates. In this paper, the syntheses and structures of three complexes, namely Ca[IrF6]·2H2O, (I), Sr[IrF6]·2H2O, (II), and Ba[IrF6], (III), are reported.
The first two title compounds, (I) and (II), are isostructural. The coordination environment of the alkaline earth metal cations includes five F atoms and three H2O molecules, forming a distorted square-antiprism (Fig. 1). The complex anion, [IrF6]2−, has the shape of an almost ideal octahedron, with average Ir—F distances of 1.934 (7) and 1.913 (10) Å for the Ca and Sr compounds, respectively. In each anion, five F atoms coordinate to the metal cation and one is involved in hydrogen bonding. There are two types of H2O molecules, each with a different function. The molecules of one type bridge two cations, while the others coordinate only to single cations. Both types also form strong hydrogen bonds with each other and with the F atoms. In addition, there are longer O—H···F contacts, with distances of about 3.4 Å.
The barium compound, (III), is isostructural with the Sr[RhF6] and Ba[RhF6] complexes (Wilhelm & Hoppe, 1974). They all belong to the Ba[GeF6] structural type. The coordination environment of the Ba2+ cation includes 12 equidistant F atoms, forming a slightly distorted cuboctahedron. Each F atom is shared between two cuboctahedra and forms one vertex of the [IrF6]2− octahedron. Thus, the anion interconnects eight Ba2+ cations. The Ba and Ir atoms form two rhombohedral sublattices separated by c/2 (Fig. 2).