In hexakis(
m-toluidinium) cyclohexaphosphate, 6C
7H
10N
+·P
6O
186-, the atomic arrangement is typical of a layer structure. Layers including the centrosymmetric P
6O
18 ring anions develop around the (100) planes at
x =
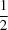
. The hydrogen-bond distribution is described.
Supporting information
CCDC reference: 143257
The title compound was prepared according to the following reaction:
H6P6O18 + 6(m-CH3C6H4NH2) ——>
(m-CH3C6H4NH3)6P6O18. An aqueous solution of
cyclohexaphosphoric acid was first obtained by passing a solution of
Li6P6O18 through an ion-exchange resin in its H-state (Amberlite IR
120). The lithium salt was prepared according to the process described by
Schulke & Kayser (1985), using "purum p.a. Fluka chemical reagents". This
solution was then added drop by drop to distilled p-toluidine (purum
p.a. Fluka) under continuous stirring until the solution exhibited a light
greenish aspect. The so-obtained solution was slowly evaporated until the
formation of large prismatic crystals of
(m-CH3C6H4NH3)6P6O18.
All H atoms were found by difference Fourier synthesis and fixed at their
positions with common isotropic displacement parameters (Uiso = 0.08 Å2).
Data collection: CAD4 Operations Manual (Enraf-Nonius, 1977); cell refinement: CAD4 Operations Manual (Enraf-Nonius, 1977); data reduction: TEXSAN for Windows (Molecular Structure Corporation, 1997); program(s) used to solve structure: SIR92 (Altomare et al., 1993); program(s) used to refine structure: TEXSAN for Windows; molecular graphics: MOLVIEW (Cense, 1990); software used to prepare material for publication: TEXSAN for Windows.
Crystal data top
| 6C7H10N+·O18P66− | Z = 1 |
| Mr = 1122.81 | F(000) = 588.00 |
| Triclinic, P1 | Dx = 1.408 Mg m−3 |
| a = 14.622 (3) Å | Mo Kα radiation, λ = 0.7107 Å |
| b = 10.656 (4) Å | Cell parameters from 25 reflections |
| c = 9.216 (4) Å | θ = 10.0–13.0° |
| α = 112.72 (3)° | µ = 0.28 mm−1 |
| β = 91.29 (3)° | T = 296 K |
| γ = 89.68 (3)° | Prism, colourless |
| V = 1324.2 (8) Å3 | 0.40 × 0.29 × 0.16 mm |
Data collection top
CAD4
diffractometer | Rint = 0.019 |
| Radiation source: X-ray tube | θmax = 25.0°, θmin = 3.5° |
| Graphite monochromator | h = −17→17 |
| ω–2θ scans | k = −12→12 |
| 9286 measured reflections | l = −10→10 |
| 4643 independent reflections | 2 standard reflections every 400 reflections |
| 3185 reflections with I > 3.00σ(I) | intensity decay: −4.2% |
Refinement top
| Refinement on F | 0 restraints |
| Least-squares matrix: full | 0 constraints |
| R[F2 > 2σ(F2)] = 0.035 | H-atom parameters not refined |
| wR(F2) = 0.050 | Weighting scheme based on measured s.u.'s w = 1/[σ2(Fo) + 0.00123|Fo|2] |
| S = 1.28 | (Δ/σ)max = 0.040 |
| 3185 reflections | Δρmax = 0.33 e Å−3 |
| 325 parameters | Δρmin = −0.45 e Å−3 |
Crystal data top
| 6C7H10N+·O18P66− | γ = 89.68 (3)° |
| Mr = 1122.81 | V = 1324.2 (8) Å3 |
| Triclinic, P1 | Z = 1 |
| a = 14.622 (3) Å | Mo Kα radiation |
| b = 10.656 (4) Å | µ = 0.28 mm−1 |
| c = 9.216 (4) Å | T = 296 K |
| α = 112.72 (3)° | 0.40 × 0.29 × 0.16 mm |
| β = 91.29 (3)° | |
Data collection top
CAD4
diffractometer | Rint = 0.019 |
| 9286 measured reflections | 2 standard reflections every 400 reflections |
| 4643 independent reflections | intensity decay: −4.2% |
| 3185 reflections with I > 3.00σ(I) | |
Refinement top
| R[F2 > 2σ(F2)] = 0.035 | 0 restraints |
| wR(F2) = 0.050 | H-atom parameters not refined |
| S = 1.28 | Δρmax = 0.33 e Å−3 |
| 3185 reflections | Δρmin = −0.45 e Å−3 |
| 325 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| P1 | 0.52332 (4) | 0.72456 (5) | 0.30379 (6) | 0.0273 (2) | |
| P2 | 0.54036 (4) | 0.82818 (5) | 0.64975 (7) | 0.0269 (2) | |
| P3 | 0.61895 (4) | 1.10173 (5) | 0.74688 (7) | 0.0291 (2) | |
| O1 | 0.50612 (11) | 0.5785 (2) | 0.2584 (2) | 0.0442 (5) | |
| O2 | 0.58288 (12) | 0.7653 (2) | 0.2020 (2) | 0.0394 (5) | |
| O3 | 0.57042 (11) | 0.7855 (2) | 0.4725 (2) | 0.0464 (5) | |
| O4 | 0.44352 (11) | 0.7892 (2) | 0.6536 (2) | 0.0359 (4) | |
| O5 | 0.61148 (12) | 0.7810 (2) | 0.7298 (2) | 0.0515 (6) | |
| O6 | 0.54059 (11) | 0.9880 (2) | 0.7084 (2) | 0.0435 (5) | |
| O7 | 0.70021 (11) | 1.0452 (2) | 0.6536 (2) | 0.0397 (5) | |
| O8 | 0.57060 (11) | 1.1972 (2) | 0.6686 (2) | 0.0398 (5) | |
| O9 | 0.6257 (1) | 1.1730 (2) | 0.9164 (2) | 0.0598 (6) | |
| N1 | 0.32595 (13) | 0.9947 (2) | 0.6556 (2) | 0.0354 (5) | |
| N2 | 0.63273 (12) | 0.3671 (2) | 0.2048 (2) | 0.0315 (5) | |
| N3 | 0.37931 (12) | 0.3783 (2) | 0.1084 (2) | 0.0311 (5) | |
| C1 | 0.2498 (2) | 1.0170 (2) | 0.7615 (3) | 0.0313 (6) | |
| C2 | 0.1644 (2) | 1.0410 (2) | 0.7135 (3) | 0.0386 (7) | |
| C3 | 0.0917 (2) | 1.0685 (3) | 0.8174 (3) | 0.0452 (7) | |
| C4 | 0.1095 (2) | 1.0705 (3) | 0.9656 (4) | 0.0517 (8) | |
| C5 | 0.1953 (2) | 1.0455 (3) | 1.0120 (3) | 0.0496 (8) | |
| C6 | 0.2669 (2) | 1.0179 (3) | 0.9090 (3) | 0.0411 (7) | |
| C7 | −0.0017 (2) | 1.0934 (4) | 0.7665 (5) | 0.0759 (12) | |
| C8 | 0.7265 (2) | 0.4120 (2) | 0.2555 (3) | 0.0314 (6) | |
| C9 | 0.7414 (2) | 0.5292 (2) | 0.3861 (3) | 0.0393 (7) | |
| C10 | 0.8294 (2) | 0.5739 (3) | 0.4361 (3) | 0.0481 (8) | |
| C11 | 0.9016 (2) | 0.4964 (4) | 0.3515 (4) | 0.0604 (9) | |
| C12 | 0.8856 (2) | 0.3796 (4) | 0.2214 (4) | 0.0639 (10) | |
| C13 | 0.7973 (2) | 0.3358 (3) | 0.1705 (3) | 0.0476 (7) | |
| C14 | 0.8456 (3) | 0.7027 (4) | 0.5785 (4) | 0.0777 (11) | |
| C15 | 0.2881 (1) | 0.4366 (2) | 0.1237 (3) | 0.0285 (6) | |
| C16 | 0.2668 (2) | 0.5439 (2) | 0.2594 (3) | 0.0348 (6) | |
| C17 | 0.1808 (2) | 0.5993 (3) | 0.2792 (3) | 0.0408 (7) | |
| C18 | 0.1162 (2) | 0.5425 (3) | 0.1599 (4) | 0.0532 (8) | |
| C19 | 0.1380 (2) | 0.4346 (3) | 0.0240 (4) | 0.0586 (9) | |
| C20 | 0.2246 (2) | 0.3804 (3) | 0.0038 (3) | 0.0454 (7) | |
| C21 | 0.1585 (2) | 0.7199 (3) | 0.4262 (4) | 0.0583 (9) | |
| H1 | 0.3647 | 1.0636 | 0.6918 | 0.080* | |
| H2 | 0.3599 | 0.9291 | 0.6501 | 0.080* | |
| H3 | 0.3055 | 0.9823 | 0.5566 | 0.080* | |
| H4 | 0.3229 | 1.0027 | 0.9380 | 0.080* | |
| H5 | 0.2040 | 1.0471 | 1.1132 | 0.080* | |
| H6 | 0.0590 | 1.0934 | 1.0432 | 0.080* | |
| H7 | 0.1560 | 1.0440 | 0.6098 | 0.080* | |
| H8 | −0.0435 | 1.1307 | 0.8403 | 0.080* | |
| H9 | −0.0288 | 1.0070 | 0.7272 | 0.080* | |
| H10 | −0.0008 | 1.1307 | 0.7043 | 0.080* | |
| H11 | 0.6275 | 0.3085 | 0.0986 | 0.080* | |
| H12 | 0.5926 | 0.4389 | 0.2226 | 0.080* | |
| H13 | 0.6076 | 0.3194 | 0.2548 | 0.080* | |
| H14 | 0.7852 | 0.2558 | 0.0832 | 0.080* | |
| H15 | 0.9338 | 0.3252 | 0.1572 | 0.080* | |
| H16 | 0.9629 | 0.5241 | 0.3820 | 0.080* | |
| H17 | 0.6863 | 0.5807 | 0.4391 | 0.080* | |
| H18 | 0.9034 | 0.7302 | 0.5782 | 0.080* | |
| H19 | 0.8562 | 0.6849 | 0.6451 | 0.080* | |
| H20 | 0.8003 | 0.7663 | 0.5944 | 0.080* | |
| H21 | 0.3829 | 0.3239 | 0.1556 | 0.080* | |
| H22 | 0.4201 | 0.4516 | 0.1598 | 0.080* | |
| H23 | 0.3896 | 0.3310 | 0.0006 | 0.080* | |
| H24 | 0.2396 | 0.3109 | −0.0928 | 0.080* | |
| H25 | 0.0946 | 0.3978 | −0.0674 | 0.080* | |
| H26 | 0.0531 | 0.5763 | 0.1766 | 0.080* | |
| H27 | 0.3091 | 0.5840 | 0.3365 | 0.080* | |
| H28 | 0.2011 | 0.7256 | 0.5172 | 0.080* | |
| H29 | 0.1718 | 0.8055 | 0.4129 | 0.080* | |
| H30 | 0.0974 | 0.7241 | 0.4511 | 0.080* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| P1 | 0.0340 (3) | 0.0208 (3) | 0.0253 (3) | −0.0005 (2) | 0.0028 (2) | 0.0068 (2) |
| P2 | 0.0343 (3) | 0.0223 (3) | 0.0260 (3) | 0.0015 (2) | 0.0024 (2) | 0.0112 (2) |
| P3 | 0.0379 (3) | 0.0244 (3) | 0.0238 (3) | −0.0015 (2) | −0.0024 (2) | 0.0081 (2) |
| O1 | 0.0412 (9) | 0.0216 (8) | 0.0642 (12) | −0.0027 (7) | −0.0055 (8) | 0.0107 (8) |
| O2 | 0.0496 (10) | 0.0351 (9) | 0.0327 (9) | 0.0032 (7) | 0.0149 (8) | 0.0115 (7) |
| O3 | 0.0389 (9) | 0.0648 (12) | 0.0255 (9) | −0.0132 (8) | 0.0028 (7) | 0.0063 (8) |
| O4 | 0.0376 (9) | 0.0346 (9) | 0.0399 (9) | −0.0021 (7) | 0.0077 (7) | 0.0189 (7) |
| O5 | 0.0498 (11) | 0.0601 (12) | 0.0638 (13) | 0.0076 (9) | −0.0028 (9) | 0.0453 (11) |
| O6 | 0.0387 (9) | 0.0233 (8) | 0.0641 (12) | 0.0013 (7) | 0.0058 (8) | 0.0117 (8) |
| O7 | 0.0354 (9) | 0.0388 (9) | 0.0456 (10) | 0.0047 (7) | −0.0000 (7) | 0.0170 (8) |
| O8 | 0.0445 (10) | 0.0419 (10) | 0.0438 (10) | 0.0127 (8) | 0.0162 (8) | 0.0279 (8) |
| O9 | 0.079 (1) | 0.0635 (13) | 0.0260 (10) | −0.0061 (11) | −0.0079 (9) | 0.0054 (9) |
| N1 | 0.0364 (11) | 0.0328 (10) | 0.0352 (11) | 0.0036 (8) | 0.0060 (9) | 0.0108 (9) |
| N2 | 0.0350 (10) | 0.0282 (9) | 0.0321 (10) | −0.0030 (8) | −0.0017 (8) | 0.0127 (8) |
| N3 | 0.0331 (10) | 0.0301 (10) | 0.0314 (10) | 0.0010 (8) | −0.0003 (8) | 0.0134 (8) |
| C1 | 0.0332 (12) | 0.0257 (11) | 0.0327 (12) | 0.0010 (9) | 0.0046 (9) | 0.0084 (9) |
| C2 | 0.0408 (13) | 0.0333 (12) | 0.0362 (13) | −0.0010 (10) | 0.0001 (11) | 0.0074 (10) |
| C3 | 0.0360 (13) | 0.0364 (13) | 0.056 (2) | 0.0009 (10) | 0.0039 (12) | 0.0092 (12) |
| C4 | 0.052 (2) | 0.049 (2) | 0.051 (2) | −0.0013 (13) | 0.0185 (13) | 0.0147 (13) |
| C5 | 0.062 (2) | 0.051 (2) | 0.036 (1) | −0.0010 (13) | 0.0100 (13) | 0.0165 (12) |
| C6 | 0.045 (1) | 0.042 (1) | 0.036 (1) | 0.0028 (11) | 0.0033 (11) | 0.0160 (11) |
| C7 | 0.045 (2) | 0.088 (3) | 0.094 (3) | 0.007 (2) | 0.002 (2) | 0.034 (2) |
| C8 | 0.0329 (12) | 0.0307 (11) | 0.0325 (12) | −0.0020 (9) | −0.0001 (9) | 0.0144 (10) |
| C9 | 0.042 (1) | 0.0332 (12) | 0.039 (1) | 0.0005 (10) | 0.0001 (11) | 0.0101 (11) |
| C10 | 0.048 (2) | 0.046 (2) | 0.046 (2) | −0.0124 (12) | −0.0064 (12) | 0.0129 (12) |
| C11 | 0.040 (2) | 0.075 (2) | 0.061 (2) | −0.013 (1) | −0.0033 (13) | 0.020 (2) |
| C12 | 0.039 (2) | 0.074 (2) | 0.067 (2) | 0.006 (1) | 0.009 (1) | 0.013 (2) |
| C13 | 0.046 (2) | 0.046 (2) | 0.043 (2) | 0.0032 (12) | 0.0074 (12) | 0.0070 (12) |
| C14 | 0.076 (2) | 0.064 (2) | 0.069 (2) | −0.018 (2) | −0.023 (2) | 0.001 (2) |
| C15 | 0.0320 (12) | 0.0239 (10) | 0.0315 (12) | −0.0019 (9) | −0.0016 (9) | 0.0130 (9) |
| C16 | 0.0377 (13) | 0.0328 (12) | 0.0318 (12) | −0.0042 (10) | −0.0028 (10) | 0.0102 (10) |
| C17 | 0.041 (1) | 0.0338 (12) | 0.046 (2) | 0.0025 (10) | 0.0058 (11) | 0.0135 (11) |
| C18 | 0.036 (1) | 0.044 (2) | 0.074 (2) | 0.0077 (12) | −0.0049 (13) | 0.018 (2) |
| C19 | 0.045 (2) | 0.052 (2) | 0.065 (2) | −0.0004 (13) | −0.024 (1) | 0.009 (2) |
| C20 | 0.050 (2) | 0.0329 (13) | 0.044 (2) | 0.0021 (11) | −0.0133 (12) | 0.0051 (11) |
| C21 | 0.059 (2) | 0.045 (2) | 0.057 (2) | 0.0100 (13) | 0.012 (1) | 0.004 (1) |
Geometric parameters (Å, º) top
| P1—O1 | 1.469 (2) | C4—C5 | 1.375 (5) |
| P1—O2 | 1.477 (2) | C4—H5 | 2.00 |
| P1—O3 | 1.578 (2) | C4—H6 | 1.00 |
| P1—O8i | 1.577 (2) | C5—C6 | 1.383 (4) |
| P2—O3 | 1.589 (2) | C5—H4 | 1.99 |
| P2—O4 | 1.483 (2) | C5—H5 | 0.93 |
| P2—O5 | 1.458 (2) | C6—H4 | 0.89 |
| P2—O6 | 1.576 (2) | C7—H8 | 0.89 |
| P3—O6 | 1.604 (2) | C7—H9 | 0.94 |
| P3—O7 | 1.467 (2) | C7—H10 | 0.81 |
| P3—O8 | 1.609 (2) | C8—C9 | 1.373 (4) |
| P3—O9 | 1.451 (2) | C8—C13 | 1.371 (4) |
| O1—H12 | 1.88 | C9—C10 | 1.380 (4) |
| O1—H22 | 1.80 | C9—H17 | 1.00 |
| O2—H1 | 1.86 | C10—C11 | 1.390 (5) |
| O2—H23 | 1.80 | C10—C14 | 1.504 (5) |
| O4—H2 | 1.93 | C10—H18 | 1.98 |
| O4—H13 | 1.84 | C10—H19 | 1.86 |
| O5—H21 | 1.81 | C11—C12 | 1.371 (5) |
| O7—H3 | 1.84 | C11—H16 | 0.95 |
| O9—H11 | 1.74 | C12—C13 | 1.384 (5) |
| N1—C1 | 1.454 (3) | C12—H15 | 0.97 |
| N1—H1 | 0.88 | C13—H14 | 0.93 |
| N1—H2 | 0.84 | C14—H18 | 0.90 |
| N1—H3 | 0.91 | C14—H19 | 0.72 |
| N2—C8 | 1.461 (3) | C14—H20 | 0.92 |
| N2—H11 | 0.94 | C15—C16 | 1.370 (4) |
| N2—H12 | 0.93 | C15—C20 | 1.375 (4) |
| N2—H13 | 0.89 | C15—H21 | 1.92 |
| N3—C15 | 1.455 (3) | C15—H22 | 1.95 |
| N3—H21 | 0.85 | C15—H23 | 1.96 |
| N3—H22 | 0.95 | C16—C17 | 1.372 (4) |
| N3—H23 | 0.94 | C16—H27 | 0.91 |
| C1—C2 | 1.371 (4) | C17—C18 | 1.383 (4) |
| C1—C6 | 1.372 (4) | C17—C21 | 1.505 (4) |
| C1—H1 | 1.95 | C17—H27 | 1.96 |
| C1—H2 | 1.96 | C18—C19 | 1.376 (5) |
| C1—H3 | 1.97 | C18—H26 | 0.98 |
| C1—H4 | 1.98 | C19—C20 | 1.375 (4) |
| C2—C3 | 1.400 (4) | C19—H25 | 1.00 |
| C2—H7 | 0.97 | C20—H24 | 0.94 |
| C3—C4 | 1.377 (5) | C21—H28 | 1.02 |
| C3—C7 | 1.489 (4) | C21—H29 | 0.99 |
| C3—H9 | 1.94 | C21—H30 | 0.92 |
| C3—H10 | 1.95 | | |
| | | |
| O1···N3 | 2.748 (3) | O4···N1 | 2.771 (3) |
| O1···N2 | 2.807 (3) | O5···N3iii | 2.657 (3) |
| O2···N1i | 2.721 (3) | O7···N1i | 2.733 (3) |
| O2···N3ii | 2.741 (3) | O9···N2iv | 2.664 (3) |
| O4···N2iii | 2.735 (3) | | |
| | | |
| O1—P1—O2 | 117.50 (12) | C5—C4—H5 | 24.1 |
| O1—P1—O3 | 109.84 (13) | C5—C4—H6 | 118.8 |
| O1—P1—O8i | 109.48 (11) | H5—C4—H6 | 94.7 |
| O2—P1—O3 | 105.23 (11) | C4—C5—C6 | 120.0 (3) |
| O2—P1—O8i | 111.29 (10) | C4—C5—H4 | 142.5 |
| O3—P1—O8i | 102.33 (11) | C4—C5—H5 | 118.9 |
| O3—P2—O4 | 109.77 (11) | C6—C5—H4 | 22.5 |
| O3—P2—O5 | 107.09 (12) | C6—C5—H5 | 121.1 |
| O3—P2—O6 | 101.21 (12) | H4—C5—H5 | 98.6 |
| O4—P2—O5 | 120.20 (11) | C1—C6—C5 | 118.3 (3) |
| O4—P2—O6 | 105.54 (10) | C1—C6—H4 | 120.8 |
| O5—P2—O6 | 111.40 (13) | C5—C6—H4 | 120.9 |
| O6—P3—O7 | 110.61 (11) | C3—C7—H8 | 118.4 |
| O6—P3—O8 | 98.70 (11) | C3—C7—H9 | 103.6 |
| O6—P3—O9 | 107.92 (13) | C3—C7—H10 | 112.6 |
| O7—P3—O8 | 105.95 (11) | H8—C7—H9 | 94.7 |
| O7—P3—O9 | 121.22 (13) | H8—C7—H10 | 111.5 |
| O8—P3—O9 | 110.11 (13) | H9—C7—H10 | 114.6 |
| P1—O1—H12 | 127.9 | N2—C8—C9 | 119.3 (2) |
| P1—O1—H22 | 140.1 | N2—C8—C13 | 118.8 (2) |
| H12—O1—H22 | 89.3 | C9—C8—C13 | 121.9 (3) |
| P1—O2—H1 | 112.3 | C8—C9—C10 | 120.3 (3) |
| P1—O2—H23 | 128.0 | C8—C9—H17 | 117.0 |
| H1i—O2i—H23i | 119.7 | C10—C9—H17 | 122.6 |
| P1—O3—P2 | 137.20 (13) | C9—C10—C11 | 118.1 (3) |
| P2—O4—H2 | 112.1 | C9—C10—C14 | 120.3 (3) |
| P2—O4—H13 | 130.0 | C9—C10—H18 | 144.1 |
| H2—O4—H13 | 111.6 | C9—C10—H19 | 121.4 |
| P2—O5—H21 | 136.6 | C11—C10—C14 | 121.6 (3) |
| P2—O6—P3 | 134.46 (12) | C11—C10—H18 | 97.1 |
| P3—O7—H3 | 117.3 | C11—C10—H19 | 115.8 |
| P1i—O8—P3 | 133.27 (12) | C14—C10—H18 | 25.3 |
| P3—O9—H11 | 158.8 | C14—C10—H19 | 21.5 |
| C1—N1—H1 | 110.3 | H18—C10—H19 | 34.9 |
| C1—N1—H2 | 114.5 | C10—C11—C12 | 120.9 (3) |
| C1—N1—H3 | 110.8 | C10—C11—H16 | 120.5 |
| H1—N1—H2 | 101.5 | C12—C11—H16 | 118.6 |
| H1—N1—H3 | 110.5 | C11—C12—C13 | 120.9 (3) |
| H2—N1—H3 | 108.8 | C11—C12—H15 | 123.3 |
| C8—N2—H11 | 113.3 | C13—C12—H15 | 115.8 |
| C8—N2—H12 | 112.7 | C8—C13—C12 | 117.9 (3) |
| C8—N2—H13 | 114.5 | C8—C13—H14 | 120.0 |
| H11—N2—H12 | 108.5 | C12—C13—H14 | 122.0 |
| H11—N2—H13 | 103.3 | C10—C14—H18 | 108.8 |
| H12—N2—H13 | 103.6 | C10—C14—H19 | 108.6 |
| C15—N3—H21 | 110.1 | C10—C14—H20 | 113.9 |
| C15—N3—H22 | 106.5 | H18—C14—H19 | 90.7 |
| C15—N3—H23 | 107.6 | H18—C14—H20 | 117.4 |
| H21—N3—H22 | 108.1 | H19—C14—H20 | 115.1 |
| H21—N3—H23 | 109.9 | N3—C15—C16 | 119.1 (2) |
| H22—N3—H23 | 114.6 | N3—C15—C20 | 119.3 (2) |
| N1—C1—C2 | 119.3 (2) | N3—C15—H21 | 24.5 |
| N1—C1—C6 | 118.4 (2) | N3—C15—H22 | 27.8 |
| N1—C1—H1 | 25.2 | N3—C15—H23 | 27.3 |
| N1—C1—H2 | 23.0 | C16—C15—C20 | 121.6 (2) |
| N1—C1—H3 | 25.6 | C16—C15—H21 | 112.6 |
| N1—C1—H4 | 95.8 | C16—C15—H22 | 95.7 |
| C2—C1—C6 | 122.2 (2) | C16—C15—H23 | 143.8 |
| C2—C1—H1 | 125.3 | C20—C15—H21 | 119.4 |
| C2—C1—H2 | 133.8 | C20—C15—H22 | 140.3 |
| C2—C1—H3 | 94.1 | C20—C15—H23 | 93.8 |
| C2—C1—H4 | 144.9 | H21—C15—H22 | 44.2 |
| C6—C1—H1 | 106.4 | H21—C15—H23 | 44.4 |
| C6—C1—H2 | 101.8 | H22—C15—H23 | 48.0 |
| C6—C1—H3 | 143.6 | C15—C16—C17 | 120.6 (3) |
| C6—C1—H4 | 22.7 | C15—C16—H27 | 121.9 |
| H1—C1—H2 | 40.0 | C17—C16—H27 | 117.4 |
| H1—C1—H3 | 44.2 | C16—C17—C18 | 118.2 (3) |
| H1—C1—H4 | 85.8 | C16—C17—C21 | 120.5 (3) |
| H2—C1—H3 | 42.6 | C16—C17—H27 | 24.2 |
| H2—C1—H4 | 79.9 | C18—C17—C21 | 121.3 (3) |
| H3—C1—H4 | 121.0 | C18—C17—H27 | 142.4 |
| C1—C2—C3 | 119.6 (3) | C21—C17—H27 | 96.3 |
| C1—C2—H7 | 119.4 | C17—C18—C19 | 120.9 (3) |
| C3—C2—H7 | 120.9 | C17—C18—H26 | 118.8 |
| C2—C3—C4 | 117.9 (3) | C19—C18—H26 | 120.2 |
| C2—C3—C7 | 120.2 (3) | C18—C19—C20 | 120.7 (3) |
| C2—C3—H9 | 117.2 | C18—C19—H25 | 122.2 |
| C2—C3—H10 | 100.6 | C20—C19—H25 | 116.8 |
| C4—C3—C7 | 121.8 (3) | C15—C20—C19 | 118.0 (3) |
| C4—C3—H9 | 116.7 | C15—C20—H24 | 122.3 |
| C4—C3—H10 | 139.8 | C19—C20—H24 | 119.6 |
| C7—C3—H9 | 28.0 | C17—C21—H28 | 111.2 |
| C7—C3—H10 | 22.6 | C17—C21—H29 | 110.5 |
| H9—C3—H10 | 44.6 | C17—C21—H30 | 113.1 |
| C3—C4—C5 | 122.0 (3) | H28—C21—H29 | 102.5 |
| C3—C4—H5 | 146.0 | H28—C21—H30 | 113.1 |
| C3—C4—H6 | 119.3 | H29—C21—H30 | 105.8 |
| Symmetry codes: (i) −x+1, −y+2, −z+1; (ii) −x+1, −y+1, −z; (iii) −x+1, −y+1, −z+1; (iv) x, y+1, z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O2i | 0.88 | 1.86 | 2.721 (3) | 164 |
| N1—H2···O4 | 0.88 | 1.93 | 2.771 (3) | 175 |
| N1—H3···O7i | 0.91 | 1.84 | 2.733 (3) | 164 |
| N2—H11···O9v | 0.94 | 1.74 | 2.664 (3) | 168 |
| N2—H12···O1 | 0.93 | 1.88 | 2.807 (3) | 177 |
| N2—H13···O4iii | 0.89 | 1.84 | 2.735 (3) | 176 |
| N3—H21···O5iii | 0.85 | 1.81 | 2.657 (3) | 176 |
| N3—H22···O1 | 0.95 | 1.80 | 2.748 (3) | 174 |
| N3—H23···O2ii | 0.94 | 1.80 | 2.741 (3) | 176 |
| Symmetry codes: (i) −x+1, −y+2, −z+1; (ii) −x+1, −y+1, −z; (iii) −x+1, −y+1, −z+1; (v) x, y−1, z−1. |
Experimental details
| Crystal data |
| Chemical formula | 6C7H10N+·O18P66− |
| Mr | 1122.81 |
| Crystal system, space group | Triclinic, P1 |
| Temperature (K) | 296 |
| a, b, c (Å) | 14.622 (3), 10.656 (4), 9.216 (4) |
| α, β, γ (°) | 112.72 (3), 91.29 (3), 89.68 (3) |
| V (Å3) | 1324.2 (8) |
| Z | 1 |
| Radiation type | Mo Kα |
| µ (mm−1) | 0.28 |
| Crystal size (mm) | 0.40 × 0.29 × 0.16 |
| |
| Data collection |
| Diffractometer | CAD4
diffractometer |
| Absorption correction | – |
No. of measured, independent and
observed [I > 3.00σ(I)] reflections | 9286, 4643, 3185 |
| Rint | 0.019 |
| (sin θ/λ)max (Å−1) | 0.594 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.035, 0.050, 1.28 |
| No. of reflections | 3185 |
| No. of parameters | 325 |
| H-atom treatment | H-atom parameters not refined |
| Δρmax, Δρmin (e Å−3) | 0.33, −0.45 |
Selected geometric parameters (Å, º) top| P1—O1 | 1.469 (2) | C3—C4 | 1.377 (5) |
| P1—O2 | 1.477 (2) | C3—C7 | 1.489 (4) |
| P1—O3 | 1.578 (2) | C4—C5 | 1.375 (5) |
| P1—O8i | 1.577 (2) | C5—C6 | 1.383 (4) |
| P2—O3 | 1.589 (2) | C8—C9 | 1.373 (4) |
| P2—O4 | 1.483 (2) | C8—C13 | 1.371 (4) |
| P2—O5 | 1.458 (2) | C9—C10 | 1.380 (4) |
| P2—O6 | 1.576 (2) | C10—C11 | 1.390 (5) |
| P3—O6 | 1.604 (2) | C10—C14 | 1.504 (5) |
| P3—O7 | 1.467 (2) | C11—C12 | 1.371 (5) |
| P3—O8 | 1.609 (2) | C12—C13 | 1.384 (5) |
| P3—O9 | 1.451 (2) | C15—C16 | 1.370 (4) |
| N1—C1 | 1.454 (3) | C15—C20 | 1.375 (4) |
| N2—C8 | 1.461 (3) | C16—C17 | 1.372 (4) |
| N3—C15 | 1.455 (3) | C17—C18 | 1.383 (4) |
| C1—C2 | 1.371 (4) | C17—C21 | 1.505 (4) |
| C1—C6 | 1.372 (4) | C18—C19 | 1.376 (5) |
| C2—C3 | 1.400 (4) | C19—C20 | 1.375 (4) |
| | | |
| O1—P1—O2 | 117.50 (12) | O5—P2—O6 | 111.40 (13) |
| O1—P1—O3 | 109.84 (13) | O6—P3—O7 | 110.61 (11) |
| O1—P1—O8i | 109.48 (11) | O6—P3—O8 | 98.70 (11) |
| O2—P1—O3 | 105.23 (11) | O6—P3—O9 | 107.92 (13) |
| O2—P1—O8i | 111.29 (10) | O7—P3—O8 | 105.95 (11) |
| O3—P1—O8i | 102.33 (11) | O7—P3—O9 | 121.22 (13) |
| O3—P2—O4 | 109.77 (11) | O8—P3—O9 | 110.11 (13) |
| O3—P2—O5 | 107.09 (12) | P1—O3—P2 | 137.20 (13) |
| O3—P2—O6 | 101.21 (12) | P2—O6—P3 | 134.46 (12) |
| O4—P2—O5 | 120.20 (11) | P1i—O8—P3 | 133.27 (12) |
| O4—P2—O6 | 105.54 (10) | | |
| Symmetry code: (i) −x+1, −y+2, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O2i | 0.88 | 1.86 | 2.721 (3) | 164 |
| N1—H2···O4 | 0.88 | 1.93 | 2.771 (3) | 175 |
| N1—H3···O7i | 0.91 | 1.84 | 2.733 (3) | 164 |
| N2—H11···O9ii | 0.94 | 1.74 | 2.664 (3) | 168 |
| N2—H12···O1 | 0.93 | 1.88 | 2.807 (3) | 177 |
| N2—H13···O4iii | 0.89 | 1.84 | 2.735 (3) | 176 |
| N3—H21···O5iii | 0.85 | 1.81 | 2.657 (3) | 176 |
| N3—H22···O1 | 0.95 | 1.80 | 2.748 (3) | 174 |
| N3—H23···O2iv | 0.94 | 1.80 | 2.741 (3) | 176 |
| Symmetry codes: (i) −x+1, −y+2, −z+1; (ii) x, y−1, z−1; (iii) −x+1, −y+1, −z+1; (iv) −x+1, −y+1, −z. |

 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2000/03/00/gs1058/gs1058fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2000/03/00/gs1058/gs1058fig2thm.gif)




The title compound, (I), has been isolated as part of an investigation into salts formed between toluidine and cyclohexaphosphoric acid. According to the relative position of the methyl group with respect to the amine function, we can separate three phases with different crystal structures. Two of these have already been studied: (o-CH3C6H4NH3)6P6O18·2H2O (Laarafa et al., 1997) and (p-CH3C6H4NH3)6P6O18·8H2O (Ben Nasr & Rzaigui, 1999). We report in this contribution the crystal structure of m-toluidine and cyclohexaphosphoric acid salt. \sch
The main geometrical features such as intramolecular bond distances and angles are reported in Table 1. A view of the asymmetric unit is shown in Fig. 1. The atomic arrangement is typical of a layer structure. Layers including the P6O18 ring anions develop around the (100) planes at x = 1/2. Between these layers, separated by a distance of 14.622 (3) Å, organic cations establish hydrogen bonds (Table 2) to interconnect the different P6O18 anions (Fig. 2). The N(1)H3 groups perform the internal P6O18 ring cohesion through hydrogen bonds involving external oxygen atoms of each PO4 tetrahedron. The two other groups, N(2)H3 and N(3)H3, are linked to three different P6O18 rings and so contribute to the intralayer cohesion of this compound. Inside such a layer, the phosphoric ring develops around the inversion centre located at (1/2, 0, 1/2) and thus presents a -1 internal symmetry. The main geometrical features of this ring are reported in Table 1. This atomic arrangement is the third type of structure obtained with toluidine and cyclohexaphosphoric acid in aqueous solution. It is worth noticing that according to the relative position of the methyl group on the phenyl ring the toluidine cyclohexaphosphate crystallizes with different degrees of hydration: the meta isomer gives an anhydrous compound, whereas the ortho and para isomers lead to di- and octahydrate salts respectively, giving additional hydrogen bonds inside the anionic layers.