ATP-binding cassettes represent the motor domains in ABC transporters, a superfamily of integral membrane-protein pumps that couple the hydrolysis of ATP to transmembrane solute translocation. A crystal of a Mg-ADP complex of the MJ1267 ATP-binding cassette was obtained that produced a diffraction pattern characterized by pathological streaking of the spots in the a* × b* plane. While the Laue symmetry of the diffraction pattern was P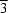 1m, the crystal was determined to be twinned based on intensity statistics, molecular-replacement analysis and difference Fourier analysis of an engineered single-site methylmercury derivative. The unit cell contains three similar 31 fibers, with two of them related by primarily translational non-crystallographic symmetry (NCS) and the third related to the first two by approximate twofold screw operations whose rotational components are very similar to the twinning operator. The promiscuous packing of these 31 fibers, which make both parallel and antiparallel interactions in the primary crystal lattice, can explain the twinning tendency based on the ability of the twin-related lattices to interact with one another while making only one slightly sub-optimal intermolecular contact per unit cell in the boundary region. The promiscuous fiber packing can also explain the streaking in the diffraction pattern based on the ability to form a variety of different lattices with similar inter-fiber packing interactions. The crystal structure was refined as a twin in space group P31 using the program CNS, yielding a free R factor of 28.9% at 2.6 Å and a refined twin fraction of 0.50. The structure shows a rigid-body rotation of the ABC-transporter-specific α-helical subdomain (ABCα subdomain) in MJ1267 compared with the conformation observed for the same protein in a C2 crystal lattice; this observation suggests that the ABCα subdomain is flexibly attached to the F1-type ATP-binding core of the ATP-binding cassette when Mg-ADP is bound at the active site.
1m, the crystal was determined to be twinned based on intensity statistics, molecular-replacement analysis and difference Fourier analysis of an engineered single-site methylmercury derivative. The unit cell contains three similar 31 fibers, with two of them related by primarily translational non-crystallographic symmetry (NCS) and the third related to the first two by approximate twofold screw operations whose rotational components are very similar to the twinning operator. The promiscuous packing of these 31 fibers, which make both parallel and antiparallel interactions in the primary crystal lattice, can explain the twinning tendency based on the ability of the twin-related lattices to interact with one another while making only one slightly sub-optimal intermolecular contact per unit cell in the boundary region. The promiscuous fiber packing can also explain the streaking in the diffraction pattern based on the ability to form a variety of different lattices with similar inter-fiber packing interactions. The crystal structure was refined as a twin in space group P31 using the program CNS, yielding a free R factor of 28.9% at 2.6 Å and a refined twin fraction of 0.50. The structure shows a rigid-body rotation of the ABC-transporter-specific α-helical subdomain (ABCα subdomain) in MJ1267 compared with the conformation observed for the same protein in a C2 crystal lattice; this observation suggests that the ABCα subdomain is flexibly attached to the F1-type ATP-binding core of the ATP-binding cassette when Mg-ADP is bound at the active site.
Supporting information
PDB reference: twinned trigonal crystal form of the MJ1267 ATP-binding cassette, 1g9x, r1g9xsf

 journal menu
journal menu











