Buy article online - an online subscription or single-article purchase is required to access this article.
In strychninium chlorate(VII) monohydrate, C
21H
23N
2O

·ClO
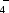
·H
2O, strychninium hydrogensulfate(VI) dihydrate, C
21H
23N
2O

·HSO
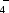
·2H
2O, and strychninium dihydrogenphosphate(V) dihydrate, C
21H
23N
2O

·H
2PO
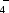
·2H
2O, the strychninium cations form bilayer sheets separated by water-anion sheets. The strychnine bilayer sheets in the three compounds are similar to one another. In all three structures, the surfaces of the cation and water-anion sheets exhibit donor-acceptor matching.
Supporting information
CCDC references: 268114; 268115; 268116
Strychnine (POCh, Poland) and a suitable acid were dissolved in water in a molar ratio of 1:1. Crystals of (I), (II) and (III) were grew after slow evaporation of the solvent.
Atoms O4A, O5A and O6A in (I) have an occupancy of 0.327 (7) and were refined isotropically. All H atoms were located from difference maps and then treated as riding, with C—H distances of 0.95–1.00 Å, N—H distances of 0.93 Å and O—H distances of 0.84–0.86 Å. The absolute structures of (I)–(III) were determined from the known absolute configuration of strychnine (Robertson & Beevers, 1951) and confirmed by the values of the Flack (1983) parameter.
For all compounds, data collection: CrysAlis CCD (Oxford Diffraction, 2001); cell refinement: CrysAlis RED (Oxford Diffraction, 2001); data reduction: CrysAlis RED; program(s) used to solve structure: SHELXS97 (Sheldrick, 1990); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: SHELXTL-NT (Bruker, 1999). Software used to prepare material for publication: SHELXL97 for (I), (II); SHELXL97 (Sheldrick, 1997) for (III).
(I) Strychninium chlorate(VII) monohydrate
top
Crystal data top
| C21H23N2O2+·ClO4−·H2O | F(000) = 476 |
| Mr = 452.88 | Dx = 1.525 Mg m−3 |
| Monoclinic, P21 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2yb | Cell parameters from 2062 reflections |
| a = 7.8016 (8) Å | θ = 3.5–29.0° |
| b = 7.6538 (9) Å | µ = 0.24 mm−1 |
| c = 16.5597 (17) Å | T = 100 K |
| β = 93.966 (9)° | Plate, colourless |
| V = 986.44 (18) Å3 | 0.10 × 0.10 × 0.05 mm |
| Z = 2 | |
Data collection top
Kuma KM-4 CCD
diffractometer | 3083 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.084 |
| Graphite monochromator | θmax = 29.0°, θmin = 3.5° |
| ω scans | h = −10→10 |
| 13754 measured reflections | k = −10→8 |
| 4042 independent reflections | l = −22→22 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.055 | H-atom parameters constrained |
| wR(F2) = 0.140 | w = 1/[σ2(Fo2) + (0.0818P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.01 | (Δ/σ)max < 0.001 |
| 4042 reflections | Δρmax = 0.91 e Å−3 |
| 293 parameters | Δρmin = −0.87 e Å−3 |
| 40 restraints | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
1225 Friedel pairs |
| Primary atom site location: structure-invariant direct methods | Absolute structure parameter: 0.09 (10) |
Crystal data top
| C21H23N2O2+·ClO4−·H2O | V = 986.44 (18) Å3 |
| Mr = 452.88 | Z = 2 |
| Monoclinic, P21 | Mo Kα radiation |
| a = 7.8016 (8) Å | µ = 0.24 mm−1 |
| b = 7.6538 (9) Å | T = 100 K |
| c = 16.5597 (17) Å | 0.10 × 0.10 × 0.05 mm |
| β = 93.966 (9)° | |
Data collection top
Kuma KM-4 CCD
diffractometer | 3083 reflections with I > 2σ(I) |
| 13754 measured reflections | Rint = 0.084 |
| 4042 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.055 | H-atom parameters constrained |
| wR(F2) = 0.140 | Δρmax = 0.91 e Å−3 |
| S = 1.01 | Δρmin = −0.87 e Å−3 |
| 4042 reflections | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
1225 Friedel pairs |
| 293 parameters | Absolute structure parameter: 0.09 (10) |
| 40 restraints | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| O1 | 1.0320 (3) | 0.9704 (4) | 0.21457 (14) | 0.0211 (5) | |
| O2 | 0.9146 (3) | 0.5133 (3) | 0.03046 (16) | 0.0256 (6) | |
| N1 | 0.7777 (3) | 0.5609 (4) | 0.14534 (16) | 0.0148 (6) | |
| N2 | 0.5137 (4) | 0.8238 (4) | 0.35552 (17) | 0.0176 (6) | |
| H22 | 0.4188 | 0.8176 | 0.3864 | 0.021* | |
| C1 | 0.5933 (5) | 0.3442 (5) | 0.0637 (2) | 0.0203 (7) | |
| H1 | 0.6772 | 0.3173 | 0.0267 | 0.024* | |
| C2 | 0.4300 (5) | 0.2668 (5) | 0.0573 (2) | 0.0219 (7) | |
| H2 | 0.4031 | 0.1860 | 0.0147 | 0.026* | |
| C3 | 0.3072 (5) | 0.3041 (5) | 0.1108 (2) | 0.0235 (8) | |
| H3A | 0.1972 | 0.2506 | 0.1043 | 0.028* | |
| C4 | 0.3440 (4) | 0.4201 (4) | 0.1744 (2) | 0.0188 (7) | |
| H4 | 0.2611 | 0.4439 | 0.2124 | 0.023* | |
| C5 | 0.5050 (4) | 0.5010 (4) | 0.18157 (19) | 0.0163 (6) | |
| C6 | 0.6268 (4) | 0.4621 (4) | 0.12664 (19) | 0.0167 (6) | |
| C7 | 0.5773 (4) | 0.6253 (4) | 0.2461 (2) | 0.0143 (6) | |
| C8 | 0.7456 (4) | 0.6905 (4) | 0.2096 (2) | 0.0138 (6) | |
| H8 | 0.8429 | 0.6889 | 0.2522 | 0.017* | |
| C9 | 0.8976 (4) | 0.5985 (4) | 0.0919 (2) | 0.0183 (7) | |
| C10 | 1.0118 (4) | 0.7532 (4) | 0.1146 (2) | 0.0197 (7) | |
| H10A | 1.0721 | 0.7878 | 0.0665 | 0.024* | |
| H10B | 1.1003 | 0.7149 | 0.1566 | 0.024* | |
| C11 | 0.9205 (4) | 0.9167 (4) | 0.1466 (2) | 0.0168 (7) | |
| H11 | 0.9148 | 1.0103 | 0.1044 | 0.020* | |
| C12 | 0.9844 (5) | 1.1313 (5) | 0.2512 (2) | 0.0238 (8) | |
| H12A | 0.9300 | 1.2092 | 0.2092 | 0.029* | |
| H12B | 1.0889 | 1.1902 | 0.2752 | 0.029* | |
| C13 | 0.8630 (4) | 1.1014 (5) | 0.3153 (2) | 0.0199 (7) | |
| H13 | 0.8996 | 1.1264 | 0.3700 | 0.024* | |
| C14 | 0.7045 (4) | 1.0407 (4) | 0.2984 (2) | 0.0188 (7) | |
| C15 | 0.5841 (4) | 1.0065 (4) | 0.3624 (2) | 0.0198 (7) | |
| H15A | 0.4882 | 1.0914 | 0.3573 | 0.024* | |
| H15B | 0.6450 | 1.0225 | 0.4164 | 0.024* | |
| C16 | 0.6416 (4) | 0.6876 (4) | 0.3873 (2) | 0.0192 (7) | |
| H16A | 0.6173 | 0.6511 | 0.4427 | 0.023* | |
| H16B | 0.7602 | 0.7339 | 0.3884 | 0.023* | |
| C17 | 0.6191 (4) | 0.5352 (4) | 0.3289 (2) | 0.0182 (7) | |
| H17A | 0.5238 | 0.4583 | 0.3431 | 0.022* | |
| H17B | 0.7258 | 0.4654 | 0.3284 | 0.022* | |
| C18 | 0.7374 (4) | 0.8668 (4) | 0.16764 (19) | 0.0140 (6) | |
| H18 | 0.6710 | 0.8470 | 0.1146 | 0.017* | |
| C19 | 0.6344 (4) | 1.0032 (4) | 0.2125 (2) | 0.0157 (6) | |
| H19 | 0.6309 | 1.1147 | 0.1810 | 0.019* | |
| C20 | 0.4520 (4) | 0.9315 (4) | 0.2140 (2) | 0.0161 (7) | |
| H20A | 0.3754 | 1.0218 | 0.2346 | 0.019* | |
| H20B | 0.4077 | 0.8993 | 0.1585 | 0.019* | |
| C21 | 0.4548 (4) | 0.7722 (4) | 0.2683 (2) | 0.0162 (6) | |
| H21 | 0.3357 | 0.7237 | 0.2679 | 0.019* | |
| Cl1 | 0.14780 (12) | 0.47191 (15) | 0.41366 (6) | 0.0361 (3) | |
| O3 | 0.0787 (4) | 0.5136 (5) | 0.33459 (17) | 0.0480 (9) | |
| O4 | 0.2722 (6) | 0.5986 (6) | 0.4434 (3) | 0.0331 (11) | 0.673 (7) |
| O5 | 0.2463 (5) | 0.3113 (5) | 0.4020 (3) | 0.0344 (12) | 0.673 (7) |
| O6 | 0.0206 (8) | 0.4325 (12) | 0.4667 (4) | 0.0734 (19) | 0.673 (7) |
| O4A | 0.3173 (8) | 0.5295 (16) | 0.4352 (7) | 0.038 (3)* | 0.327 (7) |
| O5A | 0.0523 (17) | 0.6000 (16) | 0.4575 (8) | 0.072 (4)* | 0.327 (7) |
| O6A | 0.0732 (13) | 0.3358 (12) | 0.4598 (6) | 0.038 (3)* | 0.327 (7) |
| O1W | 0.2410 (4) | 0.9535 (5) | 0.43438 (19) | 0.0451 (8) | |
| H11W | 0.1795 | 0.9016 | 0.4684 | 0.068* | |
| H12W | 0.2203 | 1.0640 | 0.4350 | 0.068* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O1 | 0.0138 (10) | 0.0210 (12) | 0.0282 (12) | −0.0025 (10) | −0.0002 (9) | −0.0054 (11) |
| O2 | 0.0265 (12) | 0.0265 (14) | 0.0245 (13) | −0.0009 (11) | 0.0068 (11) | −0.0037 (11) |
| N1 | 0.0164 (13) | 0.0112 (12) | 0.0168 (14) | −0.0013 (10) | 0.0015 (11) | −0.0030 (10) |
| N2 | 0.0169 (14) | 0.0172 (14) | 0.0192 (14) | −0.0029 (11) | 0.0042 (12) | −0.0007 (11) |
| C1 | 0.0263 (18) | 0.0171 (16) | 0.0171 (16) | −0.0001 (14) | 0.0000 (14) | 0.0028 (13) |
| C2 | 0.0309 (19) | 0.0143 (15) | 0.0190 (17) | −0.0043 (14) | −0.0074 (15) | 0.0004 (13) |
| C3 | 0.0250 (18) | 0.0155 (17) | 0.0293 (19) | −0.0068 (13) | −0.0042 (16) | 0.0037 (14) |
| C4 | 0.0171 (16) | 0.0151 (16) | 0.0240 (17) | −0.0015 (12) | −0.0006 (14) | 0.0045 (13) |
| C5 | 0.0197 (15) | 0.0128 (15) | 0.0156 (14) | −0.0008 (12) | −0.0042 (12) | 0.0053 (12) |
| C6 | 0.0195 (14) | 0.0107 (14) | 0.0194 (14) | −0.0024 (13) | −0.0023 (12) | 0.0039 (13) |
| C7 | 0.0146 (14) | 0.0103 (13) | 0.0177 (15) | −0.0007 (11) | −0.0005 (13) | 0.0038 (12) |
| C8 | 0.0137 (14) | 0.0107 (14) | 0.0169 (15) | 0.0000 (11) | 0.0000 (12) | 0.0004 (12) |
| C9 | 0.0185 (16) | 0.0161 (15) | 0.0202 (16) | 0.0049 (13) | 0.0002 (14) | 0.0022 (13) |
| C10 | 0.0161 (15) | 0.0157 (16) | 0.0274 (18) | −0.0020 (13) | 0.0019 (14) | 0.0010 (14) |
| C11 | 0.0126 (15) | 0.0153 (15) | 0.0222 (16) | −0.0019 (11) | 0.0000 (13) | 0.0010 (13) |
| C12 | 0.0168 (16) | 0.0206 (17) | 0.034 (2) | −0.0048 (14) | 0.0040 (15) | −0.0074 (15) |
| C13 | 0.0194 (16) | 0.0168 (16) | 0.0236 (17) | −0.0019 (13) | 0.0015 (14) | −0.0040 (14) |
| C14 | 0.0188 (16) | 0.0137 (15) | 0.0238 (17) | 0.0013 (13) | −0.0010 (14) | −0.0018 (13) |
| C15 | 0.0202 (16) | 0.0156 (17) | 0.0236 (17) | −0.0043 (12) | 0.0011 (14) | −0.0054 (13) |
| C16 | 0.0211 (17) | 0.0190 (17) | 0.0173 (16) | 0.0019 (13) | −0.0009 (14) | −0.0006 (13) |
| C17 | 0.0234 (16) | 0.0128 (14) | 0.0183 (16) | 0.0000 (12) | 0.0013 (14) | 0.0038 (12) |
| C18 | 0.0130 (14) | 0.0128 (15) | 0.0160 (15) | −0.0007 (12) | −0.0007 (12) | −0.0018 (12) |
| C19 | 0.0140 (13) | 0.0110 (15) | 0.0215 (15) | 0.0006 (11) | −0.0026 (12) | 0.0017 (13) |
| C20 | 0.0125 (15) | 0.0137 (16) | 0.0222 (15) | −0.0010 (11) | 0.0009 (13) | 0.0026 (12) |
| C21 | 0.0140 (15) | 0.0140 (14) | 0.0204 (16) | −0.0021 (12) | 0.0000 (13) | −0.0028 (13) |
| Cl1 | 0.0313 (5) | 0.0430 (6) | 0.0326 (5) | −0.0156 (4) | −0.0074 (4) | 0.0043 (5) |
| O3 | 0.0461 (17) | 0.054 (2) | 0.0403 (17) | 0.0195 (16) | −0.0203 (15) | −0.0117 (16) |
| O4 | 0.022 (2) | 0.039 (3) | 0.037 (2) | 0.000 (2) | −0.0042 (19) | −0.008 (2) |
| O5 | 0.030 (2) | 0.021 (2) | 0.050 (3) | −0.0011 (17) | −0.010 (2) | 0.0042 (19) |
| O6 | 0.058 (3) | 0.095 (5) | 0.072 (4) | −0.029 (3) | 0.041 (3) | −0.035 (3) |
| O1W | 0.0307 (15) | 0.053 (2) | 0.0543 (19) | −0.0033 (16) | 0.0183 (14) | −0.0129 (18) |
Geometric parameters (Å, º) top
| O1—C12 | 1.433 (4) | C12—C13 | 1.489 (5) |
| O1—C11 | 1.435 (4) | C12—H12A | 0.9900 |
| O2—C9 | 1.223 (4) | C12—H12B | 0.9900 |
| N1—C9 | 1.363 (4) | C13—C14 | 1.332 (5) |
| N1—C6 | 1.415 (4) | C13—H13 | 0.9500 |
| N1—C8 | 1.488 (4) | C14—C15 | 1.488 (5) |
| N2—C15 | 1.504 (4) | C14—C19 | 1.516 (5) |
| N2—C16 | 1.512 (4) | C15—H15A | 0.9900 |
| N2—C21 | 1.536 (4) | C15—H15B | 0.9900 |
| N2—H22 | 0.9300 | C16—C17 | 1.518 (5) |
| C1—C6 | 1.389 (5) | C16—H16A | 0.9900 |
| C1—C2 | 1.403 (5) | C16—H16B | 0.9900 |
| C1—H1 | 0.9500 | C17—H17A | 0.9900 |
| C2—C3 | 1.379 (6) | C17—H17B | 0.9900 |
| C2—H2 | 0.9500 | C18—C19 | 1.540 (4) |
| C3—C4 | 1.392 (5) | C18—H18 | 1.0000 |
| C3—H3A | 0.9500 | C19—C20 | 1.527 (4) |
| C4—C5 | 1.398 (5) | C19—H19 | 1.0000 |
| C4—H4 | 0.9500 | C20—C21 | 1.515 (5) |
| C5—C6 | 1.392 (5) | C20—H20A | 0.9900 |
| C5—C7 | 1.511 (4) | C20—H20B | 0.9900 |
| C7—C21 | 1.537 (4) | C21—H21 | 1.0000 |
| C7—C17 | 1.548 (5) | Cl1—O6 | 1.402 (3) |
| C7—C8 | 1.565 (4) | Cl1—O4A | 1.416 (4) |
| C8—C18 | 1.517 (4) | Cl1—O3 | 1.417 (3) |
| C8—H8 | 1.0000 | Cl1—O4 | 1.435 (3) |
| C9—C10 | 1.513 (5) | Cl1—O6A | 1.438 (4) |
| C10—C11 | 1.552 (5) | Cl1—O5A | 1.455 (5) |
| C10—H10A | 0.9900 | Cl1—O5 | 1.470 (3) |
| C10—H10B | 0.9900 | O1W—H11W | 0.86 |
| C11—C18 | 1.542 (4) | O1W—H12W | 0.86 |
| C11—H11 | 1.0000 | | |
| | | |
| C12—O1—C11 | 114.7 (3) | C14—C13—C12 | 122.0 (3) |
| C9—N1—C6 | 124.7 (3) | C14—C13—H13 | 119.0 |
| C9—N1—C8 | 118.7 (3) | C12—C13—H13 | 119.0 |
| C6—N1—C8 | 109.5 (2) | C13—C14—C15 | 122.2 (3) |
| C15—N2—C16 | 112.7 (3) | C13—C14—C19 | 122.2 (3) |
| C15—N2—C21 | 113.2 (3) | C15—C14—C19 | 115.5 (3) |
| C16—N2—C21 | 107.3 (2) | C14—C15—N2 | 110.7 (3) |
| C15—N2—H22 | 107.8 | C14—C15—H15A | 109.5 |
| C16—N2—H22 | 107.8 | N2—C15—H15A | 109.5 |
| C21—N2—H22 | 107.8 | C14—C15—H15B | 109.5 |
| C6—C1—C2 | 117.0 (3) | N2—C15—H15B | 109.5 |
| C6—C1—H1 | 121.5 | H15A—C15—H15B | 108.1 |
| C2—C1—H1 | 121.5 | N2—C16—C17 | 105.3 (3) |
| C3—C2—C1 | 122.1 (3) | N2—C16—H16A | 110.7 |
| C3—C2—H2 | 119.0 | C17—C16—H16A | 110.7 |
| C1—C2—H2 | 119.0 | N2—C16—H16B | 110.7 |
| C2—C3—C4 | 120.1 (3) | C17—C16—H16B | 110.7 |
| C2—C3—H3A | 119.9 | H16A—C16—H16B | 108.8 |
| C4—C3—H3A | 119.9 | C16—C17—C7 | 103.3 (3) |
| C3—C4—C5 | 118.9 (3) | C16—C17—H17A | 111.1 |
| C3—C4—H4 | 120.5 | C7—C17—H17A | 111.1 |
| C5—C4—H4 | 120.5 | C16—C17—H17B | 111.1 |
| C6—C5—C4 | 120.0 (3) | C7—C17—H17B | 111.1 |
| C6—C5—C7 | 111.0 (3) | H17A—C17—H17B | 109.1 |
| C4—C5—C7 | 128.9 (3) | C8—C18—C19 | 112.9 (3) |
| C1—C6—C5 | 121.8 (3) | C8—C18—C11 | 108.3 (3) |
| C1—C6—N1 | 128.5 (3) | C19—C18—C11 | 117.6 (3) |
| C5—C6—N1 | 109.7 (3) | C8—C18—H18 | 105.7 |
| C5—C7—C21 | 115.0 (3) | C19—C18—H18 | 105.7 |
| C5—C7—C17 | 113.0 (3) | C11—C18—H18 | 105.7 |
| C21—C7—C17 | 102.2 (3) | C14—C19—C20 | 109.2 (3) |
| C5—C7—C8 | 102.1 (3) | C14—C19—C18 | 114.5 (3) |
| C21—C7—C8 | 114.3 (3) | C20—C19—C18 | 106.4 (3) |
| C17—C7—C8 | 110.7 (3) | C14—C19—H19 | 108.9 |
| N1—C8—C18 | 105.6 (2) | C20—C19—H19 | 108.9 |
| N1—C8—C7 | 104.6 (2) | C18—C19—H19 | 108.9 |
| C18—C8—C7 | 116.8 (3) | C21—C20—C19 | 108.9 (3) |
| N1—C8—H8 | 109.9 | C21—C20—H20A | 109.9 |
| C18—C8—H8 | 109.9 | C19—C20—H20A | 109.9 |
| C7—C8—H8 | 109.9 | C21—C20—H20B | 109.9 |
| O2—C9—N1 | 123.1 (3) | C19—C20—H20B | 109.9 |
| O2—C9—C10 | 121.9 (3) | H20A—C20—H20B | 108.3 |
| N1—C9—C10 | 115.0 (3) | C20—C21—N2 | 110.1 (3) |
| C9—C10—C11 | 116.1 (3) | C20—C21—C7 | 115.5 (3) |
| C9—C10—H10A | 108.3 | N2—C21—C7 | 105.2 (3) |
| C11—C10—H10A | 108.3 | C20—C21—H21 | 108.6 |
| C9—C10—H10B | 108.3 | N2—C21—H21 | 108.6 |
| C11—C10—H10B | 108.3 | C7—C21—H21 | 108.6 |
| H10A—C10—H10B | 107.4 | O6—Cl1—O3 | 112.7 (4) |
| O1—C11—C18 | 114.5 (3) | O4A—Cl1—O3 | 116.8 (5) |
| O1—C11—C10 | 103.3 (3) | O6—Cl1—O4 | 114.9 (3) |
| C18—C11—C10 | 109.4 (3) | O3—Cl1—O4 | 111.7 (3) |
| O1—C11—H11 | 109.8 | O4A—Cl1—O6A | 119.8 (6) |
| C18—C11—H11 | 109.8 | O3—Cl1—O6A | 120.8 (5) |
| C10—C11—H11 | 109.8 | O4A—Cl1—O5A | 99.5 (8) |
| O1—C12—C13 | 111.5 (3) | O3—Cl1—O5A | 97.7 (6) |
| O1—C12—H12A | 109.3 | O6A—Cl1—O5A | 89.0 (7) |
| C13—C12—H12A | 109.3 | O6—Cl1—O5 | 107.5 (4) |
| O1—C12—H12B | 109.3 | O3—Cl1—O5 | 103.7 (2) |
| C13—C12—H12B | 109.3 | O4—Cl1—O5 | 105.3 (3) |
| H12A—C12—H12B | 108.0 | H11W—O1W—H12W | 109.4 |
| | | |
| C6—C1—C2—C3 | −0.3 (5) | C12—C13—C14—C15 | 178.5 (3) |
| C1—C2—C3—C4 | −0.9 (5) | C12—C13—C14—C19 | −2.0 (5) |
| C2—C3—C4—C5 | 1.8 (5) | C13—C14—C15—N2 | −127.3 (3) |
| C3—C4—C5—C6 | −1.4 (5) | C19—C14—C15—N2 | 53.1 (4) |
| C3—C4—C5—C7 | −177.6 (3) | C16—N2—C15—C14 | 76.5 (4) |
| C2—C1—C6—C5 | 0.6 (5) | C21—N2—C15—C14 | −45.6 (4) |
| C2—C1—C6—N1 | −178.2 (3) | C15—N2—C16—C17 | −140.5 (3) |
| C4—C5—C6—C1 | 0.2 (5) | C21—N2—C16—C17 | −15.2 (3) |
| C7—C5—C6—C1 | 177.0 (3) | N2—C16—C17—C7 | 34.9 (3) |
| C4—C5—C6—N1 | 179.2 (3) | C5—C7—C17—C16 | −165.2 (3) |
| C7—C5—C6—N1 | −4.0 (4) | C21—C7—C17—C16 | −41.1 (3) |
| C9—N1—C6—C1 | 21.0 (5) | C8—C7—C17—C16 | 81.0 (3) |
| C8—N1—C6—C1 | 170.9 (3) | N1—C8—C18—C19 | 156.1 (2) |
| C9—N1—C6—C5 | −158.0 (3) | C7—C8—C18—C19 | 40.5 (4) |
| C8—N1—C6—C5 | −8.0 (4) | N1—C8—C18—C11 | −71.8 (3) |
| C6—C5—C7—C21 | 137.6 (3) | C7—C8—C18—C11 | 172.5 (3) |
| C4—C5—C7—C21 | −45.9 (4) | O1—C11—C18—C8 | −73.9 (3) |
| C6—C5—C7—C17 | −105.6 (3) | C10—C11—C18—C8 | 41.5 (4) |
| C4—C5—C7—C17 | 70.9 (4) | O1—C11—C18—C19 | 55.5 (4) |
| C6—C5—C7—C8 | 13.3 (3) | C10—C11—C18—C19 | 170.9 (3) |
| C4—C5—C7—C8 | −170.3 (3) | C13—C14—C19—C20 | 178.7 (3) |
| C9—N1—C8—C18 | 44.2 (4) | C15—C14—C19—C20 | −1.8 (4) |
| C6—N1—C8—C18 | −107.8 (3) | C13—C14—C19—C18 | 59.5 (4) |
| C9—N1—C8—C7 | 167.9 (3) | C15—C14—C19—C18 | −121.0 (3) |
| C6—N1—C8—C7 | 16.0 (3) | C8—C18—C19—C14 | 59.6 (3) |
| C5—C7—C8—N1 | −17.0 (3) | C11—C18—C19—C14 | −67.6 (4) |
| C21—C7—C8—N1 | −141.8 (3) | C8—C18—C19—C20 | −61.1 (3) |
| C17—C7—C8—N1 | 103.5 (3) | C11—C18—C19—C20 | 171.6 (3) |
| C5—C7—C8—C18 | 99.2 (3) | C14—C19—C20—C21 | −55.9 (3) |
| C21—C7—C8—C18 | −25.5 (4) | C18—C19—C20—C21 | 68.2 (3) |
| C17—C7—C8—C18 | −140.2 (3) | C19—C20—C21—N2 | 62.7 (3) |
| C6—N1—C9—O2 | −21.4 (5) | C19—C20—C21—C7 | −56.1 (4) |
| C8—N1—C9—O2 | −168.8 (3) | C15—N2—C21—C20 | −10.6 (3) |
| C6—N1—C9—C10 | 159.6 (3) | C16—N2—C21—C20 | −135.7 (3) |
| C8—N1—C9—C10 | 12.2 (4) | C15—N2—C21—C7 | 114.4 (3) |
| O2—C9—C10—C11 | 137.3 (3) | C16—N2—C21—C7 | −10.7 (3) |
| N1—C9—C10—C11 | −43.7 (4) | C5—C7—C21—C20 | −84.0 (3) |
| C12—O1—C11—C18 | −66.7 (4) | C17—C7—C21—C20 | 153.1 (3) |
| C12—O1—C11—C10 | 174.5 (3) | C8—C7—C21—C20 | 33.5 (4) |
| C9—C10—C11—O1 | 136.4 (3) | C5—C7—C21—N2 | 154.4 (3) |
| C9—C10—C11—C18 | 14.0 (4) | C17—C7—C21—N2 | 31.6 (3) |
| C11—O1—C12—C13 | 88.7 (4) | C8—C7—C21—N2 | −88.0 (3) |
| O1—C12—C13—C14 | −66.1 (5) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O1W | 0.93 | 1.95 | 2.758 (4) | 145 |
| N2—H22···O4 | 0.93 | 2.27 | 3.004 (5) | 135 |
| N2—H22···O4A | 0.93 | 2.50 | 3.073 (10) | 120 |
| O1W—H11W···O6i | 0.86 | 1.97 | 2.709 (5) | 143 |
| O1W—H11W···O6Ai | 0.86 | 2.43 | 3.238 (12) | 158 |
| O1W—H12W···O5ii | 0.86 | 1.98 | 2.791 (5) | 156 |
| O1W—H12W···O6Aii | 0.86 | 2.42 | 3.244 (12) | 159 |
| Symmetry codes: (i) −x, y+1/2, −z+1; (ii) x, y+1, z. |
(II) Strychninium hydrogen sulfate (VI) dihydrate
top
Crystal data top
| C21H23N2O2+·HO4S−·2H2O | F(000) = 496 |
| Mr = 468.51 | Dx = 1.507 Mg m−3 |
| Monoclinic, P21 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2yb | Cell parameters from 1886 reflections |
| a = 7.7884 (8) Å | θ = 3.5–27.5° |
| b = 7.6686 (8) Å | µ = 0.21 mm−1 |
| c = 17.5780 (18) Å | T = 100 K |
| β = 100.388 (7)° | Needle, colourles |
| V = 1032.66 (18) Å3 | 0.35 × 0.25 × 0.15 mm |
| Z = 2 | |
Data collection top
Kuma KM-4 CCD
diffractometer | 3552 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.076 |
| Graphite monochromator | θmax = 27.5°, θmin = 3.5° |
| ω scans | h = −10→9 |
| 6972 measured reflections | k = −9→9 |
| 4009 independent reflections | l = −18→22 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.047 | H-atom parameters constrained |
| wR(F2) = 0.126 | w = 1/[σ2(Fo2) + (0.0753P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.05 | (Δ/σ)max < 0.001 |
| 4009 reflections | Δρmax = 0.33 e Å−3 |
| 289 parameters | Δρmin = −0.55 e Å−3 |
| 1 restraint | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
1465 Friedel pairs? |
| Primary atom site location: structure-invariant direct methods | Absolute structure parameter: −0.14 (9) |
Crystal data top
| C21H23N2O2+·HO4S−·2H2O | V = 1032.66 (18) Å3 |
| Mr = 468.51 | Z = 2 |
| Monoclinic, P21 | Mo Kα radiation |
| a = 7.7884 (8) Å | µ = 0.21 mm−1 |
| b = 7.6686 (8) Å | T = 100 K |
| c = 17.5780 (18) Å | 0.35 × 0.25 × 0.15 mm |
| β = 100.388 (7)° | |
Data collection top
Kuma KM-4 CCD
diffractometer | 3552 reflections with I > 2σ(I) |
| 6972 measured reflections | Rint = 0.076 |
| 4009 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.047 | H-atom parameters constrained |
| wR(F2) = 0.126 | Δρmax = 0.33 e Å−3 |
| S = 1.05 | Δρmin = −0.55 e Å−3 |
| 4009 reflections | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
1465 Friedel pairs? |
| 289 parameters | Absolute structure parameter: −0.14 (9) |
| 1 restraint | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O1 | 1.0840 (3) | 0.4310 (3) | 0.70225 (11) | 0.0171 (4) | |
| O2 | 0.9198 (3) | −0.0316 (3) | 0.52768 (12) | 0.0221 (5) | |
| N1 | 0.8127 (3) | 0.0140 (3) | 0.63901 (13) | 0.0146 (5) | |
| N2 | 0.6123 (3) | 0.2719 (3) | 0.84398 (13) | 0.0142 (5) | |
| H22 | 0.5288 | 0.2624 | 0.8755 | 0.017* | |
| C1 | 0.6044 (4) | −0.2025 (4) | 0.56144 (16) | 0.0177 (6) | |
| H1 | 0.6780 | −0.2299 | 0.5255 | 0.021* | |
| C2 | 0.4418 (4) | −0.2786 (4) | 0.55629 (17) | 0.0192 (6) | |
| H2 | 0.4034 | −0.3581 | 0.5153 | 0.023* | |
| C3 | 0.3318 (4) | −0.2436 (4) | 0.60875 (17) | 0.0195 (6) | |
| H3 | 0.2206 | −0.2980 | 0.6033 | 0.023* | |
| C4 | 0.3875 (4) | −0.1268 (4) | 0.66980 (16) | 0.0166 (6) | |
| H4 | 0.3151 | −0.1024 | 0.7066 | 0.020* | |
| C5 | 0.5502 (4) | −0.0471 (3) | 0.67583 (15) | 0.0130 (6) | |
| C6 | 0.6565 (4) | −0.0836 (4) | 0.62158 (14) | 0.0155 (5) | |
| C7 | 0.6405 (4) | 0.0756 (4) | 0.73712 (15) | 0.0124 (5) | |
| C8 | 0.7982 (4) | 0.1442 (4) | 0.70073 (15) | 0.0114 (5) | |
| H8 | 0.9073 | 0.1426 | 0.7407 | 0.014* | |
| C9 | 0.9200 (4) | 0.0542 (4) | 0.58699 (16) | 0.0156 (6) | |
| C10 | 1.0394 (4) | 0.2084 (4) | 0.60997 (17) | 0.0183 (6) | |
| H10A | 1.0911 | 0.2429 | 0.5647 | 0.022* | |
| H10B | 1.1362 | 0.1701 | 0.6511 | 0.022* | |
| C11 | 0.9540 (4) | 0.3712 (4) | 0.63923 (15) | 0.0135 (6) | |
| H11 | 0.9353 | 0.4617 | 0.5976 | 0.016* | |
| C12 | 1.0443 (4) | 0.5923 (4) | 0.73722 (17) | 0.0191 (6) | |
| H12A | 0.9740 | 0.6676 | 0.6976 | 0.023* | |
| H12B | 1.1540 | 0.6545 | 0.7582 | 0.023* | |
| C13 | 0.9443 (4) | 0.5575 (4) | 0.80131 (17) | 0.0183 (6) | |
| H13 | 0.9978 | 0.5824 | 0.8531 | 0.022* | |
| C14 | 0.7839 (4) | 0.4932 (4) | 0.78758 (16) | 0.0153 (6) | |
| C15 | 0.6824 (4) | 0.4573 (4) | 0.85108 (16) | 0.0169 (6) | |
| H15A | 0.5842 | 0.5407 | 0.8475 | 0.020* | |
| H15B | 0.7590 | 0.4730 | 0.9020 | 0.020* | |
| C16 | 0.7524 (4) | 0.1370 (4) | 0.87069 (16) | 0.0154 (6) | |
| H16A | 0.7501 | 0.1006 | 0.9245 | 0.018* | |
| H16B | 0.8694 | 0.1844 | 0.8681 | 0.018* | |
| C17 | 0.7089 (4) | −0.0164 (4) | 0.81521 (15) | 0.0140 (5) | |
| H17A | 0.6183 | −0.0921 | 0.8307 | 0.017* | |
| H17B | 0.8140 | −0.0871 | 0.8126 | 0.017* | |
| C18 | 0.7774 (4) | 0.3227 (4) | 0.66118 (15) | 0.0129 (6) | |
| H18 | 0.6960 | 0.3036 | 0.6110 | 0.016* | |
| C19 | 0.6855 (4) | 0.4564 (4) | 0.70598 (15) | 0.0130 (5) | |
| H19 | 0.6716 | 0.5683 | 0.6763 | 0.016* | |
| C20 | 0.5038 (4) | 0.3836 (4) | 0.70964 (16) | 0.0140 (6) | |
| H20A | 0.4338 | 0.4726 | 0.7311 | 0.017* | |
| H20B | 0.4425 | 0.3528 | 0.6570 | 0.017* | |
| C21 | 0.5242 (4) | 0.2221 (4) | 0.76095 (15) | 0.0132 (6) | |
| H21 | 0.4058 | 0.1732 | 0.7625 | 0.016* | |
| S1 | 0.24975 (9) | −0.08189 (9) | 0.89124 (4) | 0.01541 (17) | |
| O3 | 0.1683 (3) | −0.0051 (3) | 0.81895 (12) | 0.0218 (5) | |
| O4 | 0.4044 (3) | 0.0449 (3) | 0.92333 (12) | 0.0180 (5) | |
| H4A | 0.4634 | 0.0028 | 0.9639 | 0.027* | |
| O5 | 0.3280 (3) | −0.2524 (3) | 0.88275 (12) | 0.0237 (5) | |
| O6 | 0.1375 (3) | −0.0836 (3) | 0.95010 (12) | 0.0252 (5) | |
| O1W | 0.4017 (3) | 0.4373 (3) | 0.95295 (11) | 0.0187 (4) | |
| H11W | 0.3137 | 0.3822 | 0.9646 | 0.028* | |
| H12W | 0.3714 | 0.5426 | 0.9398 | 0.028* | |
| O2W | 0.1496 (3) | 0.2649 (3) | 1.00627 (13) | 0.0277 (5) | |
| H21W | 0.0551 | 0.3044 | 1.0189 | 0.042* | |
| H22W | 0.1282 | 0.1662 | 0.9831 | 0.042* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O1 | 0.0173 (9) | 0.0132 (9) | 0.0206 (9) | −0.0015 (9) | 0.0027 (8) | −0.0036 (9) |
| O2 | 0.0303 (11) | 0.0207 (11) | 0.0178 (10) | −0.0004 (10) | 0.0108 (9) | −0.0050 (8) |
| N1 | 0.0197 (12) | 0.0103 (11) | 0.0146 (11) | −0.0012 (10) | 0.0048 (10) | −0.0023 (9) |
| N2 | 0.0197 (12) | 0.0105 (12) | 0.0129 (11) | −0.0006 (10) | 0.0044 (10) | −0.0013 (9) |
| C1 | 0.0288 (16) | 0.0111 (14) | 0.0128 (13) | 0.0016 (13) | 0.0026 (11) | 0.0006 (11) |
| C2 | 0.0267 (16) | 0.0137 (14) | 0.0150 (13) | −0.0022 (13) | −0.0023 (12) | 0.0007 (11) |
| C3 | 0.0227 (15) | 0.0140 (14) | 0.0199 (14) | −0.0022 (13) | −0.0012 (12) | 0.0017 (12) |
| C4 | 0.0217 (15) | 0.0104 (13) | 0.0167 (13) | −0.0007 (12) | 0.0006 (11) | 0.0019 (10) |
| C5 | 0.0185 (13) | 0.0082 (14) | 0.0111 (11) | 0.0023 (11) | −0.0002 (10) | 0.0018 (10) |
| C6 | 0.0239 (14) | 0.0091 (12) | 0.0130 (11) | −0.0006 (13) | 0.0018 (10) | 0.0043 (12) |
| C7 | 0.0139 (13) | 0.0102 (13) | 0.0140 (12) | −0.0017 (11) | 0.0048 (10) | −0.0015 (11) |
| C8 | 0.0151 (13) | 0.0079 (12) | 0.0113 (12) | −0.0027 (11) | 0.0030 (10) | −0.0002 (10) |
| C9 | 0.0200 (14) | 0.0135 (14) | 0.0138 (13) | 0.0015 (12) | 0.0047 (11) | −0.0001 (11) |
| C10 | 0.0212 (14) | 0.0141 (14) | 0.0212 (14) | 0.0008 (13) | 0.0079 (12) | 0.0000 (12) |
| C11 | 0.0182 (14) | 0.0099 (13) | 0.0130 (12) | 0.0028 (11) | 0.0041 (10) | 0.0014 (10) |
| C12 | 0.0229 (16) | 0.0120 (14) | 0.0225 (15) | −0.0035 (13) | 0.0041 (12) | −0.0030 (12) |
| C13 | 0.0252 (16) | 0.0127 (14) | 0.0164 (13) | −0.0030 (12) | 0.0021 (12) | −0.0018 (11) |
| C14 | 0.0221 (14) | 0.0094 (12) | 0.0157 (13) | −0.0024 (12) | 0.0064 (11) | −0.0003 (11) |
| C15 | 0.0229 (14) | 0.0147 (14) | 0.0145 (12) | −0.0043 (12) | 0.0067 (11) | −0.0028 (11) |
| C16 | 0.0180 (14) | 0.0131 (14) | 0.0146 (13) | −0.0013 (12) | 0.0014 (11) | 0.0007 (11) |
| C17 | 0.0201 (14) | 0.0083 (12) | 0.0133 (12) | −0.0010 (11) | 0.0021 (10) | 0.0006 (10) |
| C18 | 0.0182 (14) | 0.0094 (13) | 0.0106 (12) | −0.0007 (11) | 0.0008 (10) | 0.0005 (10) |
| C19 | 0.0165 (12) | 0.0086 (13) | 0.0145 (12) | −0.0006 (11) | 0.0045 (10) | 0.0009 (10) |
| C20 | 0.0151 (13) | 0.0100 (14) | 0.0177 (13) | 0.0003 (11) | 0.0050 (10) | 0.0037 (10) |
| C21 | 0.0170 (13) | 0.0096 (13) | 0.0121 (12) | 0.0001 (11) | 0.0000 (11) | 0.0007 (10) |
| S1 | 0.0190 (3) | 0.0114 (3) | 0.0160 (3) | −0.0011 (3) | 0.0037 (2) | 0.0000 (3) |
| O3 | 0.0245 (11) | 0.0222 (11) | 0.0174 (10) | 0.0046 (10) | 0.0007 (8) | 0.0040 (9) |
| O4 | 0.0220 (11) | 0.0150 (11) | 0.0169 (10) | −0.0033 (9) | 0.0032 (8) | 0.0015 (8) |
| O5 | 0.0317 (12) | 0.0136 (11) | 0.0242 (11) | 0.0012 (10) | 0.0007 (10) | −0.0019 (9) |
| O6 | 0.0277 (11) | 0.0265 (12) | 0.0245 (10) | −0.0122 (11) | 0.0131 (9) | −0.0045 (10) |
| O1W | 0.0214 (10) | 0.0132 (10) | 0.0224 (10) | 0.0009 (9) | 0.0067 (8) | 0.0040 (9) |
| O2W | 0.0295 (12) | 0.0193 (12) | 0.0383 (13) | −0.0025 (11) | 0.0171 (11) | −0.0062 (10) |
Geometric parameters (Å, º) top
| O1—C11 | 1.435 (3) | C12—C13 | 1.504 (4) |
| O1—C12 | 1.439 (4) | C12—H12A | 0.9900 |
| O2—C9 | 1.233 (3) | C12—H12B | 0.9900 |
| N1—C9 | 1.380 (3) | C13—C14 | 1.324 (4) |
| N1—C6 | 1.414 (4) | C13—H13 | 0.9500 |
| N1—C8 | 1.493 (3) | C14—C15 | 1.505 (4) |
| N2—C16 | 1.515 (4) | C14—C19 | 1.526 (4) |
| N2—C15 | 1.520 (4) | C15—H15A | 0.9900 |
| N2—C21 | 1.545 (3) | C15—H15B | 0.9900 |
| N2—H22 | 0.9300 | C16—C17 | 1.527 (4) |
| C1—C2 | 1.383 (4) | C16—H16A | 0.9900 |
| C1—C6 | 1.399 (4) | C16—H16B | 0.9900 |
| C1—H1 | 0.9500 | C17—H17A | 0.9900 |
| C2—C3 | 1.393 (4) | C17—H17B | 0.9900 |
| C2—H2 | 0.9500 | C18—C19 | 1.545 (4) |
| C3—C4 | 1.405 (4) | C18—H18 | 1.0000 |
| C3—H3 | 0.9500 | C19—C20 | 1.534 (4) |
| C4—C5 | 1.393 (4) | C19—H19 | 1.0000 |
| C4—H4 | 0.9500 | C20—C21 | 1.523 (4) |
| C5—C6 | 1.399 (4) | C20—H20A | 0.9900 |
| C5—C7 | 1.506 (4) | C20—H20B | 0.9900 |
| C7—C21 | 1.548 (4) | C21—H21 | 1.0000 |
| C7—C17 | 1.550 (4) | S1—O3 | 1.440 (2) |
| C7—C8 | 1.574 (4) | S1—O5 | 1.462 (2) |
| C8—C18 | 1.531 (4) | S1—O6 | 1.470 (2) |
| C8—H8 | 1.0000 | S1—O4 | 1.572 (2) |
| C9—C10 | 1.513 (4) | O4—H4A | 0.8400 |
| C10—C11 | 1.546 (4) | O1W—H11W | 0.86 |
| C10—H10A | 0.9900 | O1W—H12W | 0.86 |
| C10—H10B | 0.9900 | O2W—H21W | 0.86 |
| C11—C18 | 1.539 (4) | O2W—H22W | 0.86 |
| C11—H11 | 1.0000 | | |
| | | |
| C11—O1—C12 | 115.4 (2) | C13—C12—H12B | 109.6 |
| C9—N1—C6 | 125.1 (2) | H12A—C12—H12B | 108.1 |
| C9—N1—C8 | 117.8 (2) | C14—C13—C12 | 121.9 (3) |
| C6—N1—C8 | 109.7 (2) | C14—C13—H13 | 119.1 |
| C16—N2—C15 | 112.7 (2) | C12—C13—H13 | 119.1 |
| C16—N2—C21 | 107.3 (2) | C13—C14—C15 | 122.5 (3) |
| C15—N2—C21 | 113.6 (2) | C13—C14—C19 | 122.5 (2) |
| C16—N2—H22 | 107.7 | C15—C14—C19 | 114.9 (2) |
| C15—N2—H22 | 107.7 | C14—C15—N2 | 109.8 (2) |
| C21—N2—H22 | 107.7 | C14—C15—H15A | 109.7 |
| C2—C1—C6 | 117.5 (3) | N2—C15—H15A | 109.7 |
| C2—C1—H1 | 121.2 | C14—C15—H15B | 109.7 |
| C6—C1—H1 | 121.2 | N2—C15—H15B | 109.7 |
| C1—C2—C3 | 122.7 (3) | H15A—C15—H15B | 108.2 |
| C1—C2—H2 | 118.6 | N2—C16—C17 | 105.3 (2) |
| C3—C2—H2 | 118.6 | N2—C16—H16A | 110.7 |
| C2—C3—C4 | 119.1 (3) | C17—C16—H16A | 110.7 |
| C2—C3—H3 | 120.5 | N2—C16—H16B | 110.7 |
| C4—C3—H3 | 120.5 | C17—C16—H16B | 110.7 |
| C5—C4—C3 | 119.3 (3) | H16A—C16—H16B | 108.8 |
| C5—C4—H4 | 120.4 | C16—C17—C7 | 102.5 (2) |
| C3—C4—H4 | 120.4 | C16—C17—H17A | 111.3 |
| C4—C5—C6 | 120.1 (3) | C7—C17—H17A | 111.3 |
| C4—C5—C7 | 129.0 (2) | C16—C17—H17B | 111.3 |
| C6—C5—C7 | 110.8 (2) | C7—C17—H17B | 111.3 |
| C1—C6—C5 | 121.2 (3) | H17A—C17—H17B | 109.2 |
| C1—C6—N1 | 129.0 (2) | C8—C18—C11 | 107.7 (2) |
| C5—C6—N1 | 109.8 (2) | C8—C18—C19 | 112.5 (2) |
| C5—C7—C21 | 115.2 (2) | C11—C18—C19 | 119.0 (2) |
| C5—C7—C17 | 113.0 (2) | C8—C18—H18 | 105.5 |
| C21—C7—C17 | 102.4 (2) | C11—C18—H18 | 105.5 |
| C5—C7—C8 | 102.5 (2) | C19—C18—H18 | 105.5 |
| C21—C7—C8 | 113.9 (2) | C14—C19—C20 | 109.8 (2) |
| C17—C7—C8 | 110.1 (2) | C14—C19—C18 | 113.7 (2) |
| N1—C8—C18 | 106.5 (2) | C20—C19—C18 | 107.2 (2) |
| N1—C8—C7 | 103.8 (2) | C14—C19—H19 | 108.7 |
| C18—C8—C7 | 117.2 (2) | C20—C19—H19 | 108.7 |
| N1—C8—H8 | 109.7 | C18—C19—H19 | 108.7 |
| C18—C8—H8 | 109.7 | C21—C20—C19 | 108.8 (2) |
| C7—C8—H8 | 109.7 | C21—C20—H20A | 109.9 |
| O2—C9—N1 | 122.2 (3) | C19—C20—H20A | 109.9 |
| O2—C9—C10 | 123.4 (3) | C21—C20—H20B | 109.9 |
| N1—C9—C10 | 114.5 (2) | C19—C20—H20B | 109.9 |
| C9—C10—C11 | 116.1 (2) | H20A—C20—H20B | 108.3 |
| C9—C10—H10A | 108.3 | C20—C21—N2 | 109.9 (2) |
| C11—C10—H10A | 108.3 | C20—C21—C7 | 115.7 (2) |
| C9—C10—H10B | 108.3 | N2—C21—C7 | 104.6 (2) |
| C11—C10—H10B | 108.3 | C20—C21—H21 | 108.8 |
| H10A—C10—H10B | 107.4 | N2—C21—H21 | 108.8 |
| O1—C11—C18 | 115.0 (2) | C7—C21—H21 | 108.8 |
| O1—C11—C10 | 103.2 (2) | O3—S1—O5 | 113.66 (13) |
| C18—C11—C10 | 110.2 (2) | O3—S1—O6 | 113.67 (14) |
| O1—C11—H11 | 109.4 | O5—S1—O6 | 112.05 (14) |
| C18—C11—H11 | 109.4 | O3—S1—O4 | 104.52 (13) |
| C10—C11—H11 | 109.4 | O5—S1—O4 | 106.49 (13) |
| O1—C12—C13 | 110.4 (2) | O6—S1—O4 | 105.53 (12) |
| O1—C12—H12A | 109.6 | S1—O4—H4A | 109.5 |
| C13—C12—H12A | 109.6 | H11W—O1W—H12W | 109.4 |
| O1—C12—H12B | 109.6 | H21W—O2W—H22W | 109.2 |
| | | |
| C6—C1—C2—C3 | −1.1 (4) | C12—C13—C14—C15 | 179.8 (3) |
| C1—C2—C3—C4 | −0.2 (5) | C12—C13—C14—C19 | −2.5 (5) |
| C2—C3—C4—C5 | 0.8 (4) | C13—C14—C15—N2 | −128.5 (3) |
| C3—C4—C5—C6 | −0.1 (4) | C19—C14—C15—N2 | 53.7 (3) |
| C3—C4—C5—C7 | −177.3 (3) | C16—N2—C15—C14 | 75.3 (3) |
| C2—C1—C6—C5 | 1.9 (4) | C21—N2—C15—C14 | −47.0 (3) |
| C2—C1—C6—N1 | −178.7 (3) | C15—N2—C16—C17 | −142.5 (2) |
| C4—C5—C6—C1 | −1.3 (4) | C21—N2—C16—C17 | −16.8 (3) |
| C7—C5—C6—C1 | 176.4 (3) | N2—C16—C17—C7 | 36.6 (3) |
| C4—C5—C6—N1 | 179.2 (2) | C5—C7—C17—C16 | −166.8 (2) |
| C7—C5—C6—N1 | −3.2 (3) | C21—C7—C17—C16 | −42.2 (3) |
| C9—N1—C6—C1 | 22.2 (5) | C8—C7—C17—C16 | 79.2 (3) |
| C8—N1—C6—C1 | 171.0 (3) | N1—C8—C18—C11 | −71.2 (3) |
| C9—N1—C6—C5 | −158.3 (3) | C7—C8—C18—C11 | 173.2 (2) |
| C8—N1—C6—C5 | −9.5 (3) | N1—C8—C18—C19 | 155.8 (2) |
| C4—C5—C7—C21 | −44.9 (4) | C7—C8—C18—C19 | 40.2 (3) |
| C6—C5—C7—C21 | 137.7 (2) | O1—C11—C18—C8 | −77.0 (3) |
| C4—C5—C7—C17 | 72.4 (4) | C10—C11—C18—C8 | 39.2 (3) |
| C6—C5—C7—C17 | −105.0 (3) | O1—C11—C18—C19 | 52.5 (3) |
| C4—C5—C7—C8 | −169.2 (3) | C10—C11—C18—C19 | 168.7 (2) |
| C6—C5—C7—C8 | 13.5 (3) | C13—C14—C19—C20 | −179.8 (3) |
| C9—N1—C8—C18 | 44.4 (3) | C15—C14—C19—C20 | −2.0 (3) |
| C6—N1—C8—C18 | −107.0 (2) | C13—C14—C19—C18 | 60.1 (4) |
| C9—N1—C8—C7 | 168.7 (2) | C15—C14—C19—C18 | −122.1 (3) |
| C6—N1—C8—C7 | 17.3 (3) | C8—C18—C19—C14 | 61.1 (3) |
| C5—C7—C8—N1 | −17.9 (3) | C11—C18—C19—C14 | −66.2 (3) |
| C21—C7—C8—N1 | −143.1 (2) | C8—C18—C19—C20 | −60.5 (3) |
| C17—C7—C8—N1 | 102.6 (2) | C11—C18—C19—C20 | 172.3 (2) |
| C5—C7—C8—C18 | 99.1 (3) | C14—C19—C20—C21 | −56.1 (3) |
| C21—C7—C8—C18 | −26.0 (3) | C18—C19—C20—C21 | 67.8 (3) |
| C17—C7—C8—C18 | −140.4 (2) | C19—C20—C21—N2 | 62.1 (3) |
| C6—N1—C9—O2 | −22.1 (5) | C19—C20—C21—C7 | −56.0 (3) |
| C8—N1—C9—O2 | −168.7 (3) | C16—N2—C21—C20 | −134.5 (2) |
| C6—N1—C9—C10 | 159.3 (3) | C15—N2—C21—C20 | −9.4 (3) |
| C8—N1—C9—C10 | 12.7 (4) | C16—N2—C21—C7 | −9.7 (3) |
| O2—C9—C10—C11 | 135.6 (3) | C15—N2—C21—C7 | 115.4 (2) |
| N1—C9—C10—C11 | −45.8 (3) | C5—C7—C21—C20 | −83.9 (3) |
| C12—O1—C11—C18 | −64.6 (3) | C17—C7—C21—C20 | 152.9 (2) |
| C12—O1—C11—C10 | 175.3 (2) | C8—C7—C21—C20 | 34.1 (3) |
| C9—C10—C11—O1 | 140.0 (2) | C5—C7—C21—N2 | 155.0 (2) |
| C9—C10—C11—C18 | 16.7 (3) | C17—C7—C21—N2 | 31.9 (3) |
| C11—O1—C12—C13 | 88.9 (3) | C8—C7—C21—N2 | −86.9 (2) |
| O1—C12—C13—C14 | −66.9 (4) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O4 | 0.93 | 2.17 | 2.900 (3) | 134 |
| N2—H22···O1W | 0.93 | 2.26 | 3.017 (3) | 138 |
| O1W—H11W···O2W | 0.86 | 1.82 | 2.673 (3) | 170 |
| O1W—H12W···O5i | 0.86 | 1.86 | 2.694 (3) | 162 |
| O2W—H21W···O6ii | 0.86 | 1.89 | 2.749 (3) | 173 |
| O2W—H22W···O6 | 0.86 | 2.01 | 2.845 (3) | 164 |
| O4—H4A···O1Wiii | 0.84 | 1.71 | 2.551 (3) | 174 |
| Symmetry codes: (i) x, y+1, z; (ii) −x, y+1/2, −z+2; (iii) −x+1, y−1/2, −z+2. |
(III) Strychninium dihydrogen phosphate(V) dihydrate
top
Crystal data top
| C21H23N2O2+·H2O4P−·2H2O | F(000) = 496 |
| Mr = 468.43 | Dx = 1.477 Mg m−3 |
| Monoclinic, P21 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2yb | Cell parameters from 1674 reflections |
| a = 7.6407 (12) Å | θ = 3.5–27.0° |
| b = 7.6624 (9) Å | µ = 0.18 mm−1 |
| c = 17.992 (2) Å | T = 100 K |
| β = 91.203 (12)° | Plate, colourless |
| V = 1053.1 (2) Å3 | 0.15 × 0.10 × 0.06 mm |
| Z = 2 | |
Data collection top
Kuma KM-4 CCD
diffractometer | 2421 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.056 |
| Graphite monochromator | θmax = 27.0°, θmin = 3.5° |
| ω scans | h = −9→9 |
| 7082 measured reflections | k = −9→8 |
| 3353 independent reflections | l = −22→22 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.054 | H-atom parameters constrained |
| wR(F2) = 0.083 | w = 1/[σ2(Fo2) + (0.0241P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.00 | (Δ/σ)max < 0.001 |
| 3353 reflections | Δρmax = 0.23 e Å−3 |
| 289 parameters | Δρmin = −0.24 e Å−3 |
| 1 restraint | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
886 Friedel pairs |
| Primary atom site location: structure-invariant direct methods | Absolute structure parameter: −0.05 (14) |
Crystal data top
| C21H23N2O2+·H2O4P−·2H2O | V = 1053.1 (2) Å3 |
| Mr = 468.43 | Z = 2 |
| Monoclinic, P21 | Mo Kα radiation |
| a = 7.6407 (12) Å | µ = 0.18 mm−1 |
| b = 7.6624 (9) Å | T = 100 K |
| c = 17.992 (2) Å | 0.15 × 0.10 × 0.06 mm |
| β = 91.203 (12)° | |
Data collection top
Kuma KM-4 CCD
diffractometer | 2421 reflections with I > 2σ(I) |
| 7082 measured reflections | Rint = 0.056 |
| 3353 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.054 | H-atom parameters constrained |
| wR(F2) = 0.083 | Δρmax = 0.23 e Å−3 |
| S = 1.00 | Δρmin = −0.24 e Å−3 |
| 3353 reflections | Absolute structure: from known structure (Robertson & Beevers, 1951), Flack (1983),
886 Friedel pairs |
| 289 parameters | Absolute structure parameter: −0.05 (14) |
| 1 restraint | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O1 | 1.0260 (3) | 0.3554 (3) | 0.70326 (12) | 0.0195 (6) | |
| O2 | 0.9172 (3) | −0.0852 (3) | 0.52588 (12) | 0.0212 (6) | |
| N1 | 0.7684 (3) | −0.0447 (4) | 0.63359 (14) | 0.0133 (7) | |
| N2 | 0.4909 (3) | 0.2025 (4) | 0.83230 (14) | 0.0143 (7) | |
| H22 | 0.3939 | 0.1899 | 0.8622 | 0.017* | |
| C1 | 0.5888 (5) | −0.2645 (5) | 0.55887 (18) | 0.0193 (8) | |
| H1 | 0.6780 | −0.2907 | 0.5246 | 0.023* | |
| C2 | 0.4247 (5) | −0.3436 (5) | 0.55375 (19) | 0.0204 (9) | |
| H2 | 0.4018 | −0.4248 | 0.5148 | 0.024* | |
| C3 | 0.2936 (5) | −0.3073 (5) | 0.60392 (18) | 0.0205 (9) | |
| H3 | 0.1821 | −0.3612 | 0.5983 | 0.025* | |
| C4 | 0.3260 (5) | −0.1914 (5) | 0.66256 (19) | 0.0159 (8) | |
| H4 | 0.2386 | −0.1693 | 0.6982 | 0.019* | |
| C5 | 0.4884 (4) | −0.1085 (5) | 0.66822 (17) | 0.0140 (8) | |
| C6 | 0.6166 (4) | −0.1450 (4) | 0.61658 (17) | 0.0140 (8) | |
| C7 | 0.5575 (4) | 0.0136 (4) | 0.72794 (19) | 0.0132 (8) | |
| C8 | 0.7318 (4) | 0.0804 (4) | 0.69469 (18) | 0.0129 (8) | |
| H8 | 0.8276 | 0.0742 | 0.7333 | 0.015* | |
| C9 | 0.8962 (4) | −0.0026 (5) | 0.5831 (2) | 0.0160 (8) | |
| C10 | 1.0105 (4) | 0.1502 (5) | 0.60675 (19) | 0.0173 (8) | |
| H10A | 1.0752 | 0.1902 | 0.5628 | 0.021* | |
| H10B | 1.0981 | 0.1073 | 0.6437 | 0.021* | |
| C11 | 0.9174 (4) | 0.3096 (5) | 0.64035 (18) | 0.0160 (8) | |
| H11 | 0.9167 | 0.4077 | 0.6037 | 0.019* | |
| C12 | 0.9796 (5) | 0.5157 (5) | 0.7401 (2) | 0.0213 (9) | |
| H12A | 0.9306 | 0.5991 | 0.7031 | 0.026* | |
| H12B | 1.0859 | 0.5688 | 0.7630 | 0.026* | |
| C13 | 0.8476 (5) | 0.4826 (5) | 0.79900 (19) | 0.0186 (9) | |
| H13 | 0.8799 | 0.5029 | 0.8495 | 0.022* | |
| C14 | 0.6865 (4) | 0.4257 (4) | 0.78276 (19) | 0.0150 (8) | |
| C15 | 0.5587 (4) | 0.3869 (5) | 0.84205 (18) | 0.0175 (8) | |
| H15A | 0.4601 | 0.4705 | 0.8389 | 0.021* | |
| H15B | 0.6163 | 0.3993 | 0.8916 | 0.021* | |
| C16 | 0.6245 (5) | 0.0676 (4) | 0.85649 (19) | 0.0159 (8) | |
| H16A | 0.6050 | 0.0297 | 0.9083 | 0.019* | |
| H16B | 0.7448 | 0.1143 | 0.8529 | 0.019* | |
| C17 | 0.5950 (4) | −0.0820 (5) | 0.80253 (17) | 0.0155 (8) | |
| H17A | 0.4941 | −0.1546 | 0.8170 | 0.019* | |
| H17B | 0.7004 | −0.1567 | 0.7995 | 0.019* | |
| C18 | 0.7293 (4) | 0.2622 (4) | 0.65908 (19) | 0.0146 (8) | |
| H18 | 0.6661 | 0.2482 | 0.6102 | 0.018* | |
| C19 | 0.6212 (4) | 0.3947 (5) | 0.70332 (18) | 0.0141 (8) | |
| H19A | 0.6201 | 0.5086 | 0.6762 | 0.017* | |
| C20 | 0.4340 (4) | 0.3226 (5) | 0.70426 (19) | 0.0153 (8) | |
| H20A | 0.3540 | 0.4109 | 0.7249 | 0.018* | |
| H20B | 0.3935 | 0.2946 | 0.6530 | 0.018* | |
| C21 | 0.4327 (4) | 0.1595 (5) | 0.75169 (18) | 0.0160 (8) | |
| H21 | 0.3108 | 0.1121 | 0.7521 | 0.019* | |
| P1 | 0.16440 (15) | 0.22066 (15) | 0.98020 (6) | 0.0333 (3) | |
| O3 | 0.2926 (4) | 0.2198 (4) | 1.05036 (13) | 0.0492 (9) | |
| H3A | 0.3945 | 0.2437 | 1.0370 | 0.074* | |
| O4 | 0.2377 (3) | 0.1028 (3) | 0.92117 (13) | 0.0276 (7) | |
| O5 | 0.1703 (3) | 0.4093 (4) | 0.94618 (15) | 0.0472 (9) | |
| H5A | 0.1079 | 0.4765 | 0.9713 | 0.071* | |
| O6 | −0.0144 (4) | 0.1752 (4) | 1.00773 (18) | 0.0589 (10) | |
| O1W | 0.3748 (4) | −0.2095 (4) | 0.97335 (14) | 0.0510 (10) | |
| H11W | 0.3503 | −0.1080 | 0.9557 | 0.077* | |
| H12W | 0.2802 | −0.2634 | 0.9841 | 0.077* | |
| O2W | 0.0398 (3) | −0.1224 (4) | 0.83054 (13) | 0.0355 (7) | |
| H21W | −0.0019 | −0.1976 | 0.8609 | 0.053* | |
| H22W | 0.1036 | −0.0482 | 0.8546 | 0.053* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O1 | 0.0144 (13) | 0.0220 (15) | 0.0220 (14) | −0.0020 (11) | −0.0002 (10) | −0.0074 (12) |
| O2 | 0.0224 (14) | 0.0238 (15) | 0.0175 (14) | 0.0002 (12) | 0.0057 (11) | −0.0053 (12) |
| N1 | 0.0156 (16) | 0.0123 (17) | 0.0122 (15) | −0.0021 (13) | 0.0032 (13) | −0.0005 (13) |
| N2 | 0.0131 (16) | 0.0155 (18) | 0.0144 (15) | 0.0010 (14) | 0.0055 (12) | −0.0007 (14) |
| C1 | 0.026 (2) | 0.0161 (19) | 0.0162 (19) | 0.0013 (19) | 0.0047 (16) | −0.0021 (17) |
| C2 | 0.033 (2) | 0.014 (2) | 0.0139 (19) | 0.0014 (18) | −0.0048 (17) | −0.0003 (16) |
| C3 | 0.021 (2) | 0.019 (2) | 0.022 (2) | 0.0004 (18) | −0.0029 (16) | 0.0041 (17) |
| C4 | 0.0180 (19) | 0.0125 (19) | 0.0175 (19) | 0.0007 (16) | 0.0024 (16) | 0.0012 (16) |
| C5 | 0.0182 (19) | 0.0122 (19) | 0.0115 (18) | −0.0004 (17) | 0.0004 (15) | 0.0014 (16) |
| C6 | 0.017 (2) | 0.016 (2) | 0.0084 (18) | 0.0001 (17) | −0.0022 (15) | 0.0054 (16) |
| C7 | 0.019 (2) | 0.0102 (19) | 0.0110 (18) | 0.0022 (16) | 0.0004 (15) | −0.0013 (15) |
| C8 | 0.0129 (19) | 0.014 (2) | 0.0120 (19) | −0.0023 (16) | 0.0011 (15) | −0.0024 (16) |
| C9 | 0.015 (2) | 0.015 (2) | 0.018 (2) | 0.0056 (17) | −0.0009 (16) | 0.0039 (17) |
| C10 | 0.0126 (19) | 0.019 (2) | 0.021 (2) | −0.0021 (16) | 0.0035 (16) | 0.0019 (17) |
| C11 | 0.016 (2) | 0.017 (2) | 0.0149 (19) | −0.0015 (17) | 0.0026 (16) | 0.0031 (16) |
| C12 | 0.022 (2) | 0.016 (2) | 0.027 (2) | −0.0006 (18) | 0.0001 (18) | −0.0068 (18) |
| C13 | 0.022 (2) | 0.017 (2) | 0.017 (2) | −0.0016 (17) | 0.0025 (16) | −0.0044 (16) |
| C14 | 0.0179 (19) | 0.0068 (19) | 0.0206 (19) | −0.0013 (16) | 0.0070 (16) | 0.0002 (16) |
| C15 | 0.0179 (19) | 0.014 (2) | 0.020 (2) | −0.0030 (17) | 0.0028 (16) | −0.0039 (17) |
| C16 | 0.021 (2) | 0.013 (2) | 0.014 (2) | −0.0005 (17) | −0.0017 (16) | 0.0013 (16) |
| C17 | 0.0171 (19) | 0.013 (2) | 0.0164 (19) | 0.0022 (17) | 0.0043 (15) | 0.0022 (16) |
| C18 | 0.016 (2) | 0.015 (2) | 0.0122 (17) | 0.0001 (16) | −0.0024 (15) | −0.0025 (15) |
| C19 | 0.0138 (18) | 0.0110 (19) | 0.0174 (18) | 0.0020 (16) | −0.0007 (15) | 0.0028 (16) |
| C20 | 0.0111 (19) | 0.015 (2) | 0.020 (2) | −0.0004 (16) | −0.0002 (15) | 0.0010 (16) |
| C21 | 0.0157 (19) | 0.018 (2) | 0.0144 (19) | 0.0003 (16) | −0.0012 (15) | −0.0034 (16) |
| P1 | 0.0432 (7) | 0.0283 (7) | 0.0293 (6) | −0.0048 (6) | 0.0221 (5) | −0.0039 (6) |
| O3 | 0.103 (3) | 0.0310 (17) | 0.0134 (14) | −0.022 (2) | 0.0013 (15) | −0.0023 (15) |
| O4 | 0.0221 (15) | 0.0384 (18) | 0.0227 (15) | −0.0092 (13) | 0.0088 (12) | −0.0097 (13) |
| O5 | 0.050 (2) | 0.0336 (19) | 0.059 (2) | 0.0100 (17) | 0.0330 (17) | 0.0188 (17) |
| O6 | 0.055 (2) | 0.034 (2) | 0.090 (2) | −0.0016 (16) | 0.0607 (18) | 0.0010 (18) |
| O1W | 0.057 (2) | 0.048 (2) | 0.0468 (19) | −0.0339 (17) | −0.0206 (16) | 0.0231 (16) |
| O2W | 0.0351 (16) | 0.0399 (19) | 0.0314 (16) | −0.0118 (15) | −0.0007 (13) | −0.0073 (15) |
Geometric parameters (Å, º) top
| O1—C11 | 1.433 (4) | C12—C13 | 1.500 (5) |
| O1—C12 | 1.443 (4) | C12—H12A | 0.9900 |
| O2—C9 | 1.222 (4) | C12—H12B | 0.9900 |
| N1—C9 | 1.385 (4) | C13—C14 | 1.333 (4) |
| N1—C6 | 1.419 (4) | C13—H13 | 0.9500 |
| N1—C8 | 1.490 (4) | C14—C15 | 1.491 (4) |
| N2—C16 | 1.511 (4) | C14—C19 | 1.522 (4) |
| N2—C15 | 1.514 (4) | C15—H15A | 0.9900 |
| N2—C21 | 1.543 (4) | C15—H15B | 0.9900 |
| N2—H22 | 0.9300 | C16—C17 | 1.516 (5) |
| C1—C2 | 1.394 (5) | C16—H16A | 0.9900 |
| C1—C6 | 1.397 (5) | C16—H16B | 0.9900 |
| C1—H1 | 0.9500 | C17—H17A | 0.9900 |
| C2—C3 | 1.391 (5) | C17—H17B | 0.9900 |
| C2—H2 | 0.9500 | C18—C19 | 1.540 (4) |
| C3—C4 | 1.397 (5) | C18—H18 | 1.0000 |
| C3—H3 | 0.9500 | C19—C20 | 1.534 (4) |
| C4—C5 | 1.396 (5) | C19—H19A | 1.0000 |
| C4—H4 | 0.9500 | C20—C21 | 1.513 (5) |
| C5—C6 | 1.393 (4) | C20—H20A | 0.9900 |
| C5—C7 | 1.511 (5) | C20—H20B | 0.9900 |
| C7—C21 | 1.537 (4) | C21—H21 | 1.0000 |
| C7—C17 | 1.550 (4) | P1—O6 | 1.504 (3) |
| C7—C8 | 1.558 (5) | P1—O4 | 1.511 (3) |
| C8—C18 | 1.534 (4) | P1—O5 | 1.570 (3) |
| C8—H8 | 1.0000 | P1—O3 | 1.582 (3) |
| C9—C10 | 1.516 (5) | O3—H3A | 0.8400 |
| C10—C11 | 1.543 (5) | O5—H5A | 0.8400 |
| C10—H10A | 0.9900 | O1W—H11W | 0.86 |
| C10—H10B | 0.9900 | O1W—H12W | 0.86 |
| C11—C18 | 1.527 (4) | O2W—H21W | 0.86 |
| C11—H11 | 1.0000 | O2W—H22W | 0.86 |
| | | |
| C11—O1—C12 | 115.3 (3) | H12A—C12—H12B | 108.1 |
| C9—N1—C6 | 124.7 (3) | C14—C13—C12 | 122.2 (3) |
| C9—N1—C8 | 118.7 (3) | C14—C13—H13 | 118.9 |
| C6—N1—C8 | 110.0 (2) | C12—C13—H13 | 118.9 |
| C16—N2—C15 | 112.2 (3) | C13—C14—C15 | 121.6 (3) |
| C16—N2—C21 | 107.7 (2) | C13—C14—C19 | 122.7 (3) |
| C15—N2—C21 | 113.5 (2) | C15—C14—C19 | 115.7 (3) |
| C16—N2—H22 | 107.7 | C14—C15—N2 | 109.3 (3) |
| C15—N2—H22 | 107.7 | C14—C15—H15A | 109.8 |
| C21—N2—H22 | 107.7 | N2—C15—H15A | 109.8 |
| C2—C1—C6 | 117.2 (3) | C14—C15—H15B | 109.8 |
| C2—C1—H1 | 121.4 | N2—C15—H15B | 109.8 |
| C6—C1—H1 | 121.4 | H15A—C15—H15B | 108.3 |
| C3—C2—C1 | 122.0 (3) | N2—C16—C17 | 104.0 (3) |
| C3—C2—H2 | 119.0 | N2—C16—H16A | 110.9 |
| C1—C2—H2 | 119.0 | C17—C16—H16A | 110.9 |
| C2—C3—C4 | 119.9 (3) | N2—C16—H16B | 110.9 |
| C2—C3—H3 | 120.0 | C17—C16—H16B | 110.9 |
| C4—C3—H3 | 120.0 | H16A—C16—H16B | 109.0 |
| C5—C4—C3 | 119.2 (3) | C16—C17—C7 | 102.7 (3) |
| C5—C4—H4 | 120.4 | C16—C17—H17A | 111.2 |
| C3—C4—H4 | 120.4 | C7—C17—H17A | 111.2 |
| C6—C5—C4 | 119.8 (3) | C16—C17—H17B | 111.2 |
| C6—C5—C7 | 111.0 (3) | C7—C17—H17B | 111.2 |
| C4—C5—C7 | 129.1 (3) | H17A—C17—H17B | 109.1 |
| C5—C6—C1 | 122.0 (3) | C11—C18—C8 | 107.7 (3) |
| C5—C6—N1 | 109.3 (3) | C11—C18—C19 | 118.3 (3) |
| C1—C6—N1 | 128.7 (3) | C8—C18—C19 | 112.6 (3) |
| C5—C7—C21 | 116.0 (3) | C11—C18—H18 | 105.7 |
| C5—C7—C17 | 112.3 (3) | C8—C18—H18 | 105.7 |
| C21—C7—C17 | 102.0 (3) | C19—C18—H18 | 105.7 |
| C5—C7—C8 | 102.7 (3) | C14—C19—C20 | 109.5 (3) |
| C21—C7—C8 | 114.1 (3) | C14—C19—C18 | 114.8 (3) |
| C17—C7—C8 | 110.1 (3) | C20—C19—C18 | 106.2 (3) |
| N1—C8—C18 | 106.1 (2) | C14—C19—H19A | 108.8 |
| N1—C8—C7 | 104.3 (3) | C20—C19—H19A | 108.8 |
| C18—C8—C7 | 117.1 (3) | C18—C19—H19A | 108.8 |
| N1—C8—H8 | 109.7 | C21—C20—C19 | 108.7 (3) |
| C18—C8—H8 | 109.7 | C21—C20—H20A | 109.9 |
| C7—C8—H8 | 109.7 | C19—C20—H20A | 109.9 |
| O2—C9—N1 | 122.6 (3) | C21—C20—H20B | 109.9 |
| O2—C9—C10 | 123.4 (3) | C19—C20—H20B | 109.9 |
| N1—C9—C10 | 114.0 (3) | H20A—C20—H20B | 108.3 |
| C9—C10—C11 | 117.0 (3) | C20—C21—C7 | 115.6 (3) |
| C9—C10—H10A | 108.1 | C20—C21—N2 | 110.4 (3) |
| C11—C10—H10A | 108.1 | C7—C21—N2 | 104.4 (3) |
| C9—C10—H10B | 108.1 | C20—C21—H21 | 108.7 |
| C11—C10—H10B | 108.1 | C7—C21—H21 | 108.7 |
| H10A—C10—H10B | 107.3 | N2—C21—H21 | 108.7 |
| O1—C11—C18 | 114.6 (3) | O6—P1—O4 | 116.46 (17) |
| O1—C11—C10 | 103.9 (3) | O6—P1—O5 | 112.05 (17) |
| C18—C11—C10 | 110.0 (3) | O4—P1—O5 | 105.20 (15) |
| O1—C11—H11 | 109.4 | O6—P1—O3 | 106.72 (19) |
| C18—C11—H11 | 109.4 | O4—P1—O3 | 109.02 (17) |
| C10—C11—H11 | 109.4 | O5—P1—O3 | 107.02 (17) |
| O1—C12—C13 | 110.8 (3) | P1—O3—H3A | 109.5 |
| O1—C12—H12A | 109.5 | P1—O5—H5A | 109.5 |
| C13—C12—H12A | 109.5 | H11W—O1W—H12W | 109.9 |
| O1—C12—H12B | 109.5 | H21W—O2W—H22W | 109.7 |
| C13—C12—H12B | 109.5 | | |
| | | |
| C6—C1—C2—C3 | −0.5 (5) | C12—C13—C14—C15 | 177.6 (3) |
| C1—C2—C3—C4 | −1.5 (5) | C12—C13—C14—C19 | −1.9 (5) |
| C2—C3—C4—C5 | 2.4 (5) | C13—C14—C15—N2 | −125.8 (3) |
| C3—C4—C5—C6 | −1.4 (5) | C19—C14—C15—N2 | 53.7 (4) |
| C3—C4—C5—C7 | −176.5 (3) | C16—N2—C15—C14 | 74.7 (3) |
| C4—C5—C6—C1 | −0.6 (5) | C21—N2—C15—C14 | −47.7 (3) |
| C7—C5—C6—C1 | 175.4 (3) | C15—N2—C16—C17 | −144.8 (3) |
| C4—C5—C6—N1 | 179.6 (3) | C21—N2—C16—C17 | −19.2 (3) |
| C7—C5—C6—N1 | −4.4 (4) | N2—C16—C17—C7 | 38.8 (3) |
| C2—C1—C6—C5 | 1.5 (5) | C5—C7—C17—C16 | −168.6 (3) |
| C2—C1—C6—N1 | −178.8 (3) | C21—C7—C17—C16 | −43.7 (3) |
| C9—N1—C6—C5 | −157.6 (3) | C8—C7—C17—C16 | 77.7 (3) |
| C8—N1—C6—C5 | −6.8 (4) | O1—C11—C18—C8 | −74.2 (3) |
| C9—N1—C6—C1 | 22.6 (5) | C10—C11—C18—C8 | 42.3 (3) |
| C8—N1—C6—C1 | 173.4 (3) | O1—C11—C18—C19 | 55.0 (4) |
| C6—C5—C7—C21 | 138.1 (3) | C10—C11—C18—C19 | 171.5 (3) |
| C4—C5—C7—C21 | −46.5 (5) | N1—C8—C18—C11 | −71.8 (3) |
| C6—C5—C7—C17 | −105.2 (3) | C7—C8—C18—C11 | 172.4 (3) |
| C4—C5—C7—C17 | 70.3 (5) | N1—C8—C18—C19 | 155.9 (3) |
| C6—C5—C7—C8 | 13.0 (4) | C7—C8—C18—C19 | 40.0 (4) |
| C4—C5—C7—C8 | −171.6 (3) | C13—C14—C19—C20 | 177.7 (3) |
| C9—N1—C8—C18 | 43.1 (4) | C15—C14—C19—C20 | −1.9 (4) |
| C6—N1—C8—C18 | −109.7 (3) | C13—C14—C19—C18 | 58.4 (5) |
| C9—N1—C8—C7 | 167.4 (3) | C15—C14—C19—C18 | −121.1 (3) |
| C6—N1—C8—C7 | 14.6 (3) | C11—C18—C19—C14 | −66.4 (4) |
| C5—C7—C8—N1 | −16.0 (3) | C8—C18—C19—C14 | 60.5 (4) |
| C21—C7—C8—N1 | −142.3 (3) | C11—C18—C19—C20 | 172.6 (3) |
| C17—C7—C8—N1 | 103.7 (3) | C8—C18—C19—C20 | −60.6 (3) |
| C5—C7—C8—C18 | 100.9 (3) | C14—C19—C20—C21 | −55.8 (4) |
| C21—C7—C8—C18 | −25.5 (4) | C18—C19—C20—C21 | 68.6 (3) |
| C17—C7—C8—C18 | −139.4 (3) | C19—C20—C21—C7 | −57.1 (4) |
| C6—N1—C9—O2 | −20.9 (5) | C19—C20—C21—N2 | 61.1 (3) |
| C8—N1—C9—O2 | −169.4 (3) | C5—C7—C21—C20 | −84.8 (4) |
| C6—N1—C9—C10 | 161.1 (3) | C17—C7—C21—C20 | 152.9 (3) |
| C8—N1—C9—C10 | 12.6 (4) | C8—C7—C21—C20 | 34.2 (4) |
| O2—C9—C10—C11 | 138.8 (3) | C5—C7—C21—N2 | 153.7 (3) |
| N1—C9—C10—C11 | −43.2 (4) | C17—C7—C21—N2 | 31.4 (3) |
| C12—O1—C11—C18 | −67.2 (4) | C8—C7—C21—N2 | −87.3 (3) |
| C12—O1—C11—C10 | 172.8 (2) | C16—N2—C21—C20 | −133.0 (3) |
| C9—C10—C11—O1 | 136.0 (3) | C15—N2—C21—C20 | −8.1 (3) |
| C9—C10—C11—C18 | 12.9 (4) | C16—N2—C21—C7 | −8.1 (3) |
| C11—O1—C12—C13 | 88.5 (3) | C15—N2—C21—C7 | 116.7 (3) |
| O1—C12—C13—C14 | −65.0 (4) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O4 | 0.93 | 1.75 | 2.648 (3) | 163 |
| O1W—H11W···O4 | 0.86 | 1.93 | 2.768 (4) | 166 |
| O1W—H12W···O6i | 0.86 | 2.09 | 2.919 (4) | 161 |
| O2W—H21W···O6i | 0.86 | 2.56 | 3.307 (4) | 146 |
| O2W—H22W···O4 | 0.86 | 1.94 | 2.797 (3) | 172 |
| O3—H3A···O1Wii | 0.84 | 1.81 | 2.642 (4) | 169 |
| O5—H5A···O6iii | 0.84 | 1.73 | 2.510 (4) | 154 |
| Symmetry codes: (i) −x, y−1/2, −z+2; (ii) −x+1, y+1/2, −z+2; (iii) −x, y+1/2, −z+2. |
Experimental details
| | (I) | (II) | (III) |
| Crystal data |
| Chemical formula | C21H23N2O2+·ClO4−·H2O | C21H23N2O2+·HO4S−·2H2O | C21H23N2O2+·H2O4P−·2H2O |
| Mr | 452.88 | 468.51 | 468.43 |
| Crystal system, space group | Monoclinic, P21 | Monoclinic, P21 | Monoclinic, P21 |
| Temperature (K) | 100 | 100 | 100 |
| a, b, c (Å) | 7.8016 (8), 7.6538 (9), 16.5597 (17) | 7.7884 (8), 7.6686 (8), 17.5780 (18) | 7.6407 (12), 7.6624 (9), 17.992 (2) |
| β (°) | 93.966 (9) | 100.388 (7) | 91.203 (12) |
| V (Å3) | 986.44 (18) | 1032.66 (18) | 1053.1 (2) |
| Z | 2 | 2 | 2 |
| Radiation type | Mo Kα | Mo Kα | Mo Kα |
| µ (mm−1) | 0.24 | 0.21 | 0.18 |
| Crystal size (mm) | 0.10 × 0.10 × 0.05 | 0.35 × 0.25 × 0.15 | 0.15 × 0.10 × 0.06 |
| |
| Data collection |
| Diffractometer | Kuma KM-4 CCD
diffractometer | Kuma KM-4 CCD
diffractometer | Kuma KM-4 CCD
diffractometer |
| Absorption correction | – | – | – |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 13754, 4042, 3083 | 6972, 4009, 3552 | 7082, 3353, 2421 |
| Rint | 0.084 | 0.076 | 0.056 |
| (sin θ/λ)max (Å−1) | 0.682 | 0.649 | 0.639 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.055, 0.140, 1.01 | 0.047, 0.126, 1.05 | 0.054, 0.083, 1.00 |
| No. of reflections | 4042 | 4009 | 3353 |
| No. of parameters | 293 | 289 | 289 |
| No. of restraints | 40 | 1 | 1 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.91, −0.87 | 0.33, −0.55 | 0.23, −0.24 |
| Absolute structure | From known structure (Robertson & Beevers, 1951), Flack (1983),
1225 Friedel pairs | From known structure (Robertson & Beevers, 1951), Flack (1983),
1465 Friedel pairs? | From known structure (Robertson & Beevers, 1951), Flack (1983),
886 Friedel pairs |
| Absolute structure parameter | 0.09 (10) | −0.14 (9) | −0.05 (14) |
Hydrogen-bond geometry (Å, º) for (I) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O1W | 0.93 | 1.95 | 2.758 (4) | 145 |
| N2—H22···O4 | 0.93 | 2.27 | 3.004 (5) | 135 |
| N2—H22···O4A | 0.93 | 2.50 | 3.073 (10) | 120 |
| O1W—H11W···O6i | 0.86 | 1.97 | 2.709 (5) | 143 |
| O1W—H11W···O6Ai | 0.86 | 2.43 | 3.238 (12) | 158 |
| O1W—H12W···O5ii | 0.86 | 1.98 | 2.791 (5) | 156 |
| O1W—H12W···O6Aii | 0.86 | 2.42 | 3.244 (12) | 159 |
| Symmetry codes: (i) −x, y+1/2, −z+1; (ii) x, y+1, z. |
Hydrogen-bond geometry (Å, º) for (II) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O4 | 0.93 | 2.17 | 2.900 (3) | 134 |
| N2—H22···O1W | 0.93 | 2.26 | 3.017 (3) | 138 |
| O1W—H11W···O2W | 0.86 | 1.82 | 2.673 (3) | 170 |
| O1W—H12W···O5i | 0.86 | 1.86 | 2.694 (3) | 162 |
| O2W—H21W···O6ii | 0.86 | 1.89 | 2.749 (3) | 173 |
| O2W—H22W···O6 | 0.86 | 2.01 | 2.845 (3) | 164 |
| O4—H4A···O1Wiii | 0.84 | 1.71 | 2.551 (3) | 174 |
| Symmetry codes: (i) x, y+1, z; (ii) −x, y+1/2, −z+2; (iii) −x+1, y−1/2, −z+2. |
Hydrogen-bond geometry (Å, º) for (III) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N2—H22···O4 | 0.93 | 1.75 | 2.648 (3) | 163 |
| O1W—H11W···O4 | 0.86 | 1.93 | 2.768 (4) | 166 |
| O1W—H12W···O6i | 0.86 | 2.09 | 2.919 (4) | 161 |
| O2W—H21W···O6i | 0.86 | 2.56 | 3.307 (4) | 146 |
| O2W—H22W···O4 | 0.86 | 1.94 | 2.797 (3) | 172 |
| O3—H3A···O1Wii | 0.84 | 1.81 | 2.642 (4) | 169 |
| O5—H5A···O6iii | 0.84 | 1.73 | 2.510 (4) | 154 |
| Symmetry codes: (i) −x, y−1/2, −z+2; (ii) −x+1, y+1/2, −z+2; (iii) −x, y+1/2, −z+2. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2005/03/00/gd1357/gd1357fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2005/03/00/gd1357/gd1357fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2005/03/00/gd1357/gd1357fig3thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



The recently described racemic resolution of alanine derivatives by the strychnine (and brucine) has revealed that enantiomeric recognition of the N-protected alanine (Gould & Walkinshaw, 1984; Bialonska & Ciunik, 2004) is dependent on the type of alkaloid self-assembly, which is determined by the donor/acceptor properties of the resolved substrate. Cocrystallization of strychnine with the chloric(VII), sulfuric(VI) or phosphoric(V) acid gives an opportunity to control, by the number of the donor/acceptor sites, the molecular recognition.
Views of the molecules of C21H23N2O2+·ClO4−·H2O, (I), C21H23N2O2+·HSO4−·2H2O, (II), and C21H23N2O2+·H2PO4−·2H2O, (III), with the atom-numbering schemes, are presented in Fig. 1. The strychnine molecules in (I), (II) and (III) form, in a similar way to many other crystals (NO3−, SO42−, Cl−, Br− and I−) (Mostad, 1985; Borkhoven et al., 1951; Ghosh et al., 1989; Robertson & Beevers, 1951; Yuan et al., 1994], herring-bone bilayer sheets separated by anions and water molecules (Fig. 2). The carbonyl O atom and arene ring of the strychnine molecule are directed inwards in the bilayer sheet. This configuration, coupled with protonation of the tertiary amine N atom, causes the surfaces of the strychnine bilayer sheets in all of the crystals under investigation to act as donors.
In the crystal structure of (I), the chlorate(VII) anion adopts two arrangements (in a 2:1 ratio) around the Cl1—O3 bond. Atom O3 atom of the anion is directed towards a flat hole at the surface of the strychnine layer (see Fig. 2a). As presented in Fig. 3(a), protonated amine atom N2 of the strychnine molecule acts as a donor in a biffurcated hydrogen bond, where the water molecule O1W, as well as atom O4 or O4A of the disordered anion, act as acceptors. Both the anion and the water molecule are also directly linked by one branch [O1W—H11W···O4 (or O5A)] of the biffurcated hydrogen bond. Atom O6 (or O6A) of the anion at (−x, y + 1/2, −z + 1) is the other acceptor. The O1W—H12W···O5(x, y + 1, z) (or O6A) interaction complete the hydrogen-bonded network, resulting in anion/water tapes along the [010] direction (Table 1).
Contrary to the crystal structure of (I), in the crystal structure of (II) two water molecules and a hydrogensulfate(VI) anion form, through symmetry operations, a hydrogen-bonded sheet. However, the chlorate(VII) anion/water tapes in (I) and the hydrogensulfate(VI) anion/water layers in (II) exhibit largely similar hydrogen-bonding patterns. This similarity results from (i) similar occupancy of the holes at the surface of the strychnine sheet by the anion (see Fig. 2b) and (ii) the similar environment of the protonated tertiary amine atom, N2, of the strychnine molecule, which in (II) is again the donor in a biffurcated hydrogen bond to the anion and water molecule (Fig. 3b). However, whereas two anions and a water molecule are connected by the biffurcated hydrogen bond in (I), the equivalent interaction in (II) also involves the other water molecule (O2W). The layer results from the fact that besides participating in the N2—H22···O4 hydrogen bond, atom O4 of the hydrogensulfate(VI) anion also acts as a donor in a hydrogen bond to the O1W water molecule from the (−x + 1, y − 1/2, −z + 2) position (Table 2).
The holes at the surface of the strychnine bilayer sheet in (III) are occupied by the O2W water molecules (Fig. 2c), and atom N2 of the strychnine molecule acts as a donor of a hydrogen bond of which atom O4 of the dihydrogenphosphate(V) anion is the only acceptor (Fig. 3c). The anion/water layer that results from the hydrogen-bond network formed by the dihydrogenphosphate(V) anions and water molecules is quite different from the layers in (II). In (III), each O atom of the anion participates in short hydrogen bonds (Table 3). Atoms O3 and O5 atoms of the anion act as hydrogen-bond donors to water molecule O1W and atom O6 in the adjacent anion at (−x, y + 1/2, −z + 2), respectively. Each of the two remaining O atoms (O4 and O6) acts as an acceptor of three hydrogen bonds; besides the above-mentioned interactions with atom O5 or N2 as donor, water molecules O1W and O2W, as bridges, are the donors of the other two bonds.
In summary, the chlorate(VII), hydrogensulfate(VI) and dihydrogenphosphate(V) anions are recognized by similar strychnine self-assembled bilayers in (I), (I) and (III), respectively. However, the molecular recognition of the anions, resulting from the control of their donor/acceptor sites, shows a great diversity of non-covalent interactions inside the water/anion sheet. The various hydrogen-bonding patterns in the anion/water sheet are determined by the different numbers of water molecules and especially by the different occupancy of the holes at the surface of the strychnine bilayer, by the anion in (I) and (II), and by a water molecule in (III). Furthermore, the formation of bifurcated hydrogen bonds or participation of one acceptor in several intermolecular interactions cause the surfaces of both the water/anion and the strychnine sheets to fit well in accordance with their donor/acceptor properties.