Buy article online - an online subscription or single-article purchase is required to access this article.
In the title molecule, C20H22N2O, the pyridine heterocycle has an envelope conformation. The crystal packing is stabilized by intermolecular N—H O hydrogen bonds, which link the molecules into chains along the [1
O hydrogen bonds, which link the molecules into chains along the [1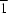 0] direction.
0] direction.
 O hydrogen bonds, which link the molecules into chains along the [1
O hydrogen bonds, which link the molecules into chains along the [1Supporting information
Crystallographic Information File (CIF) https://doi.org/10.1107/S160053680502413X/cv6556sup1.cif | |
Structure factor file (CIF format) https://doi.org/10.1107/S160053680502413X/cv6556Isup2.hkl |
CCDC reference: 283786
Key indicators
- Single-crystal X-ray study
- T = 296 K
- Mean
![[sigma]](//journals.iucr.org/logos/entities/sigma_rmgif.gif) (C-C) = 0.004 Å
(C-C) = 0.004 Å - R factor = 0.034
- wR factor = 0.090
- Data-to-parameter ratio = 8.2
checkCIF/PLATON results
No syntax errors found
Alert level C PLAT152_ALERT_1_C Supplied and Calc Volume s.u. Inconsistent ..... ? PLAT230_ALERT_2_C Hirshfeld Test Diff for C12 - C13 .. 5.26 su PLAT241_ALERT_2_C Check High Ueq as Compared to Neighbors for C12 PLAT241_ALERT_2_C Check High Ueq as Compared to Neighbors for C19
Alert level G REFLT03_ALERT_4_G Please check that the estimate of the number of Friedel pairs is correct. If it is not, please give the correct count in the _publ_section_exptl_refinement section of the submitted CIF. From the CIF: _diffrn_reflns_theta_max 26.25 From the CIF: _reflns_number_total 1757 Count of symmetry unique reflns 1761 Completeness (_total/calc) 99.77% TEST3: Check Friedels for noncentro structure Estimate of Friedel pairs measured 0 Fraction of Friedel pairs measured 0.000 Are heavy atom types Z>Si present no
0 ALERT level A = In general: serious problem 0 ALERT level B = Potentially serious problem 4 ALERT level C = Check and explain 1 ALERT level G = General alerts; check 1 ALERT type 1 CIF construction/syntax error, inconsistent or missing data 3 ALERT type 2 Indicator that the structure model may be wrong or deficient 0 ALERT type 3 Indicator that the structure quality may be low 1 ALERT type 4 Improvement, methodology, query or suggestion
Computing details top
Data collection: XSCANS (Siemens, 1996); cell refinement: XSCANS; data reduction: XSCANS; program(s) used to solve structure: SHELXTL-Plus (Sheldrick, 1998); program(s) used to refine structure: SHELXTL-Plus; molecular graphics: SHELXTL-Plus; software used to prepare material for publication: SHELXTL-Plus.
(-)-1-Benzyl-4-[1(S)-phenylethylamino]-5,6-dihydropyridin-2(1H)-one top
Crystal data top
| C20H22N2O | Z = 1 |
| Mr = 306.40 | F(000) = 164 |
| Triclinic, P1 | Dx = 1.168 Mg m−3 |
| Hall symbol: P 1 | Melting point: 434 K |
| a = 6.0091 (7) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 6.4829 (8) Å | Cell parameters from 77 reflections |
| c = 12.2030 (15) Å | θ = 3.6–14.1° |
| α = 81.136 (10)° | µ = 0.07 mm−1 |
| β = 80.67 (1)° | T = 296 K |
| γ = 69.04 (1)° | Prism, colourless |
| V = 435.67 (9) Å3 | 0.48 × 0.40 × 0.16 mm |
Data collection top
| Bruker P4 diffractometer | Rint = 0.020 |
| Radiation source: fine-focus sealed tube, FN4 | θmax = 26.3°, θmin = 3.4° |
| Graphite monochromator | h = −7→7 |
| ω scans | k = −8→7 |
| 3488 measured reflections | l = −15→15 |
| 1757 independent reflections | 3 standard reflections every 97 reflections |
| 1481 reflections with I > 2σ(I) | intensity decay: 1% |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: geom and difmap |
| R[F2 > 2σ(F2)] = 0.034 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.090 | w = 1/[σ2(Fo2) + (0.0444P)2 + 0.0309P] where P = (Fo2 + 2Fc2)/3 |
| S = 1.01 | (Δ/σ)max < 0.001 |
| 1757 reflections | Δρmax = 0.11 e Å−3 |
| 214 parameters | Δρmin = −0.09 e Å−3 |
| 3 restraints | Extinction correction: SHELXTL-Plus (Sheldrick, 1998), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.091 (12) |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top
| x | y | z | Uiso*/Ueq | ||
| N1 | 0.5371 (3) | 0.0310 (3) | 0.79695 (16) | 0.0506 (5) | |
| O1 | 0.8481 (3) | −0.1208 (3) | 0.89904 (16) | 0.0681 (5) | |
| C2 | 0.6715 (4) | 0.0429 (4) | 0.87423 (18) | 0.0463 (5) | |
| C3 | 0.5983 (4) | 0.2452 (4) | 0.92672 (18) | 0.0451 (5) | |
| H3A | 0.7045 | 0.2687 | 0.9669 | 0.054* | |
| C4 | 0.3778 (4) | 0.4027 (3) | 0.91891 (17) | 0.0429 (5) | |
| C5 | 0.2177 (4) | 0.3773 (4) | 0.8433 (2) | 0.0547 (6) | |
| H5A | 0.1050 | 0.3118 | 0.8869 | 0.066* | |
| H5B | 0.1264 | 0.5227 | 0.8097 | 0.066* | |
| C6 | 0.3584 (5) | 0.2339 (4) | 0.7527 (2) | 0.0607 (7) | |
| H6A | 0.2496 | 0.1951 | 0.7159 | 0.073* | |
| H6B | 0.4384 | 0.3168 | 0.6975 | 0.073* | |
| N7 | 0.2809 (4) | 0.5786 (3) | 0.97903 (17) | 0.0505 (5) | |
| H7A | 0.139 (6) | 0.676 (5) | 0.961 (3) | 0.067 (8)* | |
| C8 | 0.4060 (5) | 0.6408 (4) | 1.0536 (2) | 0.0536 (6) | |
| H8A | 0.5501 | 0.6623 | 1.0107 | 0.064* | |
| C9 | 0.2412 (8) | 0.8635 (5) | 1.0945 (3) | 0.0897 (11) | |
| H9A | 0.2002 | 0.9726 | 1.0315 | 0.135* | |
| H9B | 0.3226 | 0.9110 | 1.1418 | 0.135* | |
| H9C | 0.0976 | 0.8462 | 1.1358 | 0.135* | |
| C10 | 0.4819 (4) | 0.4702 (4) | 1.15329 (19) | 0.0489 (5) | |
| C11 | 0.6715 (6) | 0.4612 (6) | 1.2054 (3) | 0.0812 (9) | |
| H11A | 0.7563 | 0.5571 | 1.1782 | 0.097* | |
| C12 | 0.7384 (7) | 0.3101 (8) | 1.2986 (3) | 0.1045 (13) | |
| H12A | 0.8658 | 0.3079 | 1.3336 | 0.125* | |
| C13 | 0.6194 (6) | 0.1652 (6) | 1.3392 (3) | 0.0876 (10) | |
| H13A | 0.6664 | 0.0628 | 1.4007 | 0.105* | |
| C14 | 0.4310 (6) | 0.1728 (5) | 1.2883 (2) | 0.0738 (8) | |
| H14A | 0.3476 | 0.0757 | 1.3156 | 0.089* | |
| C15 | 0.3624 (5) | 0.3236 (4) | 1.1963 (2) | 0.0561 (6) | |
| H15A | 0.2329 | 0.3265 | 1.1627 | 0.067* | |
| C16 | 0.6019 (4) | −0.1712 (4) | 0.7444 (2) | 0.0539 (6) | |
| H16A | 0.4595 | −0.1762 | 0.7187 | 0.065* | |
| H16B | 0.6533 | −0.2966 | 0.8005 | 0.065* | |
| C17 | 0.7982 (4) | −0.1998 (4) | 0.6465 (2) | 0.0495 (5) | |
| C18 | 0.8451 (6) | −0.3710 (5) | 0.5818 (3) | 0.0778 (9) | |
| H18A | 0.7493 | −0.4596 | 0.5952 | 0.093* | |
| C19 | 1.0351 (8) | −0.4111 (6) | 0.4969 (3) | 0.0999 (12) | |
| H19A | 1.0672 | −0.5289 | 0.4550 | 0.120* | |
| C20 | 1.1742 (6) | −0.2818 (6) | 0.4742 (3) | 0.0892 (11) | |
| H20A | 1.3000 | −0.3092 | 0.4167 | 0.107* | |
| C21 | 1.1270 (6) | −0.1114 (6) | 0.5367 (2) | 0.0797 (9) | |
| H21A | 1.2208 | −0.0209 | 0.5214 | 0.096* | |
| C22 | 0.9425 (5) | −0.0713 (5) | 0.6220 (2) | 0.0619 (7) | |
| H22A | 0.9145 | 0.0452 | 0.6642 | 0.074* |
Atomic displacement parameters (Å2) top
| U11 | U22 | U33 | U12 | U13 | U23 | |
| N1 | 0.0448 (11) | 0.0449 (10) | 0.0539 (11) | −0.0005 (9) | −0.0104 (9) | −0.0127 (9) |
| O1 | 0.0551 (10) | 0.0567 (10) | 0.0717 (11) | 0.0155 (8) | −0.0209 (9) | −0.0155 (9) |
| C2 | 0.0383 (12) | 0.0448 (12) | 0.0455 (12) | −0.0017 (10) | −0.0061 (10) | −0.0036 (9) |
| C3 | 0.0367 (11) | 0.0482 (12) | 0.0467 (12) | −0.0062 (9) | −0.0109 (9) | −0.0078 (10) |
| C4 | 0.0433 (11) | 0.0372 (11) | 0.0418 (11) | −0.0058 (9) | −0.0065 (9) | −0.0030 (9) |
| C5 | 0.0424 (12) | 0.0484 (12) | 0.0648 (15) | 0.0019 (10) | −0.0193 (11) | −0.0097 (11) |
| C6 | 0.0588 (15) | 0.0539 (14) | 0.0622 (15) | −0.0005 (12) | −0.0260 (12) | −0.0112 (12) |
| N7 | 0.0487 (11) | 0.0379 (9) | 0.0565 (11) | 0.0005 (8) | −0.0168 (9) | −0.0067 (8) |
| C8 | 0.0623 (14) | 0.0453 (12) | 0.0552 (14) | −0.0207 (11) | −0.0055 (11) | −0.0075 (10) |
| C9 | 0.133 (3) | 0.0442 (14) | 0.091 (2) | −0.0166 (17) | −0.028 (2) | −0.0201 (14) |
| C10 | 0.0462 (12) | 0.0512 (12) | 0.0478 (12) | −0.0127 (10) | −0.0027 (10) | −0.0135 (10) |
| C11 | 0.0653 (17) | 0.112 (3) | 0.077 (2) | −0.0432 (18) | −0.0171 (15) | −0.0012 (18) |
| C12 | 0.071 (2) | 0.158 (4) | 0.083 (2) | −0.029 (2) | −0.0369 (19) | −0.004 (2) |
| C13 | 0.084 (2) | 0.097 (2) | 0.0600 (18) | −0.006 (2) | −0.0152 (17) | 0.0027 (17) |
| C14 | 0.094 (2) | 0.0690 (17) | 0.0522 (15) | −0.0261 (16) | −0.0017 (15) | 0.0014 (13) |
| C15 | 0.0622 (15) | 0.0568 (14) | 0.0498 (13) | −0.0207 (12) | −0.0073 (11) | −0.0054 (11) |
| C16 | 0.0544 (14) | 0.0470 (12) | 0.0583 (14) | −0.0131 (11) | −0.0034 (11) | −0.0130 (10) |
| C17 | 0.0531 (13) | 0.0414 (11) | 0.0461 (12) | −0.0041 (10) | −0.0113 (10) | −0.0050 (9) |
| C18 | 0.094 (2) | 0.0629 (17) | 0.077 (2) | −0.0249 (16) | 0.0023 (17) | −0.0271 (15) |
| C19 | 0.129 (3) | 0.077 (2) | 0.079 (2) | −0.016 (2) | 0.013 (2) | −0.0381 (18) |
| C20 | 0.084 (2) | 0.096 (3) | 0.0637 (18) | −0.010 (2) | 0.0135 (17) | −0.0116 (18) |
| C21 | 0.0720 (19) | 0.106 (2) | 0.0581 (16) | −0.0329 (17) | −0.0007 (14) | −0.0007 (17) |
| C22 | 0.0675 (16) | 0.0691 (17) | 0.0491 (14) | −0.0221 (14) | −0.0054 (12) | −0.0107 (12) |
Geometric parameters (Å, º) top
| N1—C2 | 1.364 (3) | C11—C12 | 1.391 (5) |
| N1—C16 | 1.447 (3) | C11—H11A | 0.9300 |
| N1—C6 | 1.461 (3) | C12—C13 | 1.364 (6) |
| O1—C2 | 1.242 (3) | C12—H12A | 0.9300 |
| C2—C3 | 1.442 (3) | C13—C14 | 1.360 (5) |
| C3—C4 | 1.360 (3) | C13—H13A | 0.9300 |
| C3—H3A | 0.9300 | C14—C15 | 1.381 (4) |
| C4—N7 | 1.350 (3) | C14—H14A | 0.9300 |
| C4—C5 | 1.501 (3) | C15—H15A | 0.9300 |
| C5—C6 | 1.500 (3) | C16—C17 | 1.520 (3) |
| C5—H5A | 0.9700 | C16—H16A | 0.9700 |
| C5—H5B | 0.9700 | C16—H16B | 0.9700 |
| C6—H6A | 0.9700 | C17—C22 | 1.375 (4) |
| C6—H6B | 0.9700 | C17—C18 | 1.381 (4) |
| N7—C8 | 1.451 (3) | C18—C19 | 1.390 (5) |
| N7—H7A | 0.90 (3) | C18—H18A | 0.9300 |
| C8—C10 | 1.522 (3) | C19—C20 | 1.354 (6) |
| C8—C9 | 1.534 (4) | C19—H19A | 0.9300 |
| C8—H8A | 0.9800 | C20—C21 | 1.360 (5) |
| C9—H9A | 0.9600 | C20—H20A | 0.9300 |
| C9—H9B | 0.9600 | C21—C22 | 1.373 (4) |
| C9—H9C | 0.9600 | C21—H21A | 0.9300 |
| C10—C11 | 1.372 (4) | C22—H22A | 0.9300 |
| C10—C15 | 1.381 (3) | ||
| C2—N1—C16 | 119.82 (18) | C15—C10—C8 | 122.2 (2) |
| C2—N1—C6 | 119.26 (18) | C10—C11—C12 | 120.8 (3) |
| C16—N1—C6 | 119.90 (19) | C10—C11—H11A | 119.6 |
| O1—C2—N1 | 119.5 (2) | C12—C11—H11A | 119.6 |
| O1—C2—C3 | 122.2 (2) | C13—C12—C11 | 120.8 (3) |
| N1—C2—C3 | 118.27 (17) | C13—C12—H12A | 119.6 |
| C4—C3—C2 | 121.65 (19) | C11—C12—H12A | 119.6 |
| C4—C3—H3A | 119.2 | C14—C13—C12 | 118.9 (3) |
| C2—C3—H3A | 119.2 | C14—C13—H13A | 120.5 |
| N7—C4—C3 | 125.4 (2) | C12—C13—H13A | 120.5 |
| N7—C4—C5 | 115.44 (18) | C13—C14—C15 | 120.5 (3) |
| C3—C4—C5 | 119.07 (18) | C13—C14—H14A | 119.7 |
| C6—C5—C4 | 111.75 (19) | C15—C14—H14A | 119.7 |
| C6—C5—H5A | 109.3 | C14—C15—C10 | 121.4 (3) |
| C4—C5—H5A | 109.3 | C14—C15—H15A | 119.3 |
| C6—C5—H5B | 109.3 | C10—C15—H15A | 119.3 |
| C4—C5—H5B | 109.3 | N1—C16—C17 | 115.6 (2) |
| H5A—C5—H5B | 107.9 | N1—C16—H16A | 108.4 |
| N1—C6—C5 | 111.6 (2) | C17—C16—H16A | 108.4 |
| N1—C6—H6A | 109.3 | N1—C16—H16B | 108.4 |
| C5—C6—H6A | 109.3 | C17—C16—H16B | 108.4 |
| N1—C6—H6B | 109.3 | H16A—C16—H16B | 107.4 |
| C5—C6—H6B | 109.3 | C22—C17—C18 | 117.6 (2) |
| H6A—C6—H6B | 108.0 | C22—C17—C16 | 123.4 (2) |
| C4—N7—C8 | 124.88 (19) | C18—C17—C16 | 118.9 (2) |
| C4—N7—H7A | 116.2 (19) | C17—C18—C19 | 120.1 (3) |
| C8—N7—H7A | 117.9 (19) | C17—C18—H18A | 119.9 |
| N7—C8—C10 | 114.32 (19) | C19—C18—H18A | 119.9 |
| N7—C8—C9 | 107.9 (2) | C20—C19—C18 | 121.2 (3) |
| C10—C8—C9 | 109.7 (2) | C20—C19—H19A | 119.4 |
| N7—C8—H8A | 108.3 | C18—C19—H19A | 119.4 |
| C10—C8—H8A | 108.3 | C19—C20—C21 | 118.9 (3) |
| C9—C8—H8A | 108.3 | C19—C20—H20A | 120.5 |
| C8—C9—H9A | 109.5 | C21—C20—H20A | 120.5 |
| C8—C9—H9B | 109.5 | C20—C21—C22 | 120.7 (3) |
| H9A—C9—H9B | 109.5 | C20—C21—H21A | 119.6 |
| C8—C9—H9C | 109.5 | C22—C21—H21A | 119.6 |
| H9A—C9—H9C | 109.5 | C21—C22—C17 | 121.4 (3) |
| H9B—C9—H9C | 109.5 | C21—C22—H22A | 119.3 |
| C11—C10—C15 | 117.5 (2) | C17—C22—H22A | 119.3 |
| C11—C10—C8 | 120.3 (2) | ||
| C16—N1—C2—O1 | −0.2 (3) | C15—C10—C11—C12 | −0.4 (5) |
| C6—N1—C2—O1 | −168.7 (2) | C8—C10—C11—C12 | 178.2 (3) |
| C16—N1—C2—C3 | −178.6 (2) | C10—C11—C12—C13 | 1.0 (6) |
| C6—N1—C2—C3 | 13.0 (3) | C11—C12—C13—C14 | −1.1 (6) |
| O1—C2—C3—C4 | −164.6 (2) | C12—C13—C14—C15 | 0.5 (5) |
| N1—C2—C3—C4 | 13.7 (3) | C13—C14—C15—C10 | 0.1 (4) |
| C2—C3—C4—N7 | 169.0 (2) | C11—C10—C15—C14 | −0.2 (4) |
| C2—C3—C4—C5 | −7.1 (3) | C8—C10—C15—C14 | −178.7 (2) |
| N7—C4—C5—C6 | 160.0 (2) | C2—N1—C16—C17 | −82.3 (3) |
| C3—C4—C5—C6 | −23.6 (3) | C6—N1—C16—C17 | 86.1 (3) |
| C2—N1—C6—C5 | −43.3 (3) | N1—C16—C17—C22 | 13.3 (3) |
| C16—N1—C6—C5 | 148.3 (2) | N1—C16—C17—C18 | −170.5 (2) |
| C4—C5—C6—N1 | 46.8 (3) | C22—C17—C18—C19 | 1.1 (4) |
| C3—C4—N7—C8 | 6.6 (3) | C16—C17—C18—C19 | −175.3 (3) |
| C5—C4—N7—C8 | −177.2 (2) | C17—C18—C19—C20 | −1.4 (6) |
| C4—N7—C8—C10 | −64.0 (3) | C18—C19—C20—C21 | 0.6 (6) |
| C4—N7—C8—C9 | 173.7 (2) | C19—C20—C21—C22 | 0.4 (5) |
| N7—C8—C10—C11 | 155.3 (2) | C20—C21—C22—C17 | −0.7 (5) |
| C9—C8—C10—C11 | −83.3 (3) | C18—C17—C22—C21 | −0.1 (4) |
| N7—C8—C10—C15 | −26.2 (3) | C16—C17—C22—C21 | 176.1 (3) |
| C9—C8—C10—C15 | 95.1 (3) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N7—H7A···O1i | 0.90 (3) | 1.96 (3) | 2.850 (3) | 171 (3) |
| Symmetry code: (i) x−1, y+1, z. |

 Subscribe to Acta Crystallographica Section E: Crystallographic Communications
Subscribe to Acta Crystallographica Section E: Crystallographic Communications
The full text of this article is available to subscribers to the journal.
- Information on subscribing
- Sample issue
- If you have already subscribed, you may need to register

 journal menu
journal menu











 Alert level C
Alert level C
 Alert level G
Alert level G



