Buy article online - an online subscription or single-article purchase is required to access this article.
Samples of two differently prepared zeolite rho loaded with different amounts of monomethylamine (MMA) were studied in their hydrated and dehydrated forms by X-ray and neutron diffraction. Both zeolites are partially dealuminated, as indicated by nonframework alumina, which is assumed to be Al2O3 or AlOOH. Series I was prepared from dry-calcined NH4-rho at 873 K, series II from steam-calcined NH4-rho at 773 K. The samples were loaded with different amounts of deuterated MMA, Rietveld refinements yielded the following results for series I (dry): (1) H3.8(H-MMA)5Cs0.2Al9Si39O2.Al2O3.28H2O, X-ray data collection at room temperature, Im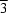 m, a = 14.9991 (2) Å, Rwp = 0.095; (2) composition as in (1), anhydrous and deuterated, neutron data collected at 5 K, disproportionation into two phases in I
m, a = 14.9991 (2) Å, Rwp = 0.095; (2) composition as in (1), anhydrous and deuterated, neutron data collected at 5 K, disproportionation into two phases in I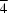 3m, with a = 14.8410 (7) and 14.5273 (11) Å, Rwp = 0.038; (3) (H-MMA)8.8Cs0.2Al9Si39O96.Al2O3.25H2O, X-ray data collection at room temperature, I
3m, with a = 14.8410 (7) and 14.5273 (11) Å, Rwp = 0.038; (3) (H-MMA)8.8Cs0.2Al9Si39O96.Al2O3.25H2O, X-ray data collection at room temperature, I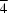 3m, a = 14.9771 (2) Å, Rwp = 0.090. Series II (steam): (4) H0.3(H-MMA)5Cs0.7Al6Si42O96.2.5Al2O3.23H2O, X-ray data collected at room temperature,Im
3m, a = 14.9771 (2) Å, Rwp = 0.090. Series II (steam): (4) H0.3(H-MMA)5Cs0.7Al6Si42O96.2.5Al2O3.23H2O, X-ray data collected at room temperature,Im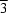 m, a = 15.0323 (2) Å, Rwp = 0.124; (5) composition as in (4), anhydrous and deuterated, neutron data collected at 5 K, disproportionation into two phases in I
m, a = 15.0323 (2) Å, Rwp = 0.124; (5) composition as in (4), anhydrous and deuterated, neutron data collected at 5 K, disproportionation into two phases in I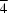 3m with a = 14.9151 (2) and 14.6475(8) Å, Rwp = 0.031. In the hydrated samples MMA resides on the center axis in the α-cage with the N atoms pointing to the single eight-ring; upon dehydration it migrates into the double eight-rings.
3m with a = 14.9151 (2) and 14.6475(8) Å, Rwp = 0.031. In the hydrated samples MMA resides on the center axis in the α-cage with the N atoms pointing to the single eight-ring; upon dehydration it migrates into the double eight-rings.

 journal menu
journal menu









 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


