Buy article online - an online subscription or single-article purchase is required to access this article.

One of the main phases formed at the beginning of the carbonation reaction of cementitious building materials is the calcium hemicarboaluminate (abbreviated as Hc). This AFm (shorthand for hydrated calcium aluminate phases structurally related to hydrocalumite) phase was synthesized, crystallized and then studied by synchrotron X-ray powder diffraction and micro-Raman spectroscopy. At room temperature and standard experimental conditions two major cementitious phases were detected, the Hc phase (as a major phase) and carbonated calcium hemicarboaluminate (abbreviated as cHc). By increasing the temperature the Hc form transforms into cHc. The crystal structures of these important AFm phases were successfully solved and refined in the
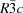
space group of the trigonal crystal system. Hc has the unit-cell parameters
a = 5.7757 (1) and
c = 48.812 (2) Å, and cHc the unit-cell parameters
a = 5.7534 (1) and
c = 46.389 (1) Å. The two crystal structures are composed of positively charged main layers, [Ca
4Al
2(OH)
12]
2+, and negatively charged interlayers, [OH
2n(CO
3)
1 - n·4H
2O]
2-. The structure of the main layers is typical of the AFm family. Conversely, the interlayer region has a characteristic structure built up from water molecules and statistically distributed anions. In the interlayer, the Hc carbonate and hydroxyl anions are distributed in a 0.25:0.5 ratio, whereas the ratio of the anions in the cHc interlayers is 0.4:0.2.
Supporting information
(cHc) Tetracalcium dialuminium dodecahydroxide hemicarbonate hydroxide n-hydrate
top
Crystal data top
| C0.4AlCa2O9.2 | Z = 6 |
| Mr = 259.14 | F(000) = 774.0 |
| Trigonal, R3c | Dx = 1.942 Mg m−3 |
| Hall symbol: -R 3 2"c | Synchrotron radiation, λ = 0.8264 (3) Å |
| a = 5.7534 (1) Å | T = 347 K |
| c = 46.389 (1) Å | white |
| V = 1329.84 (4) Å3 | ?, ? × ? × ? mm |
Data collection top
Synchrotron Diamond I11 (Great Britain)
diffractometer | |
Refinement top
| Rp = 0.030 | χ2 = 40.069 |
| Rwp = 0.050 | ? data points |
| Rexp = 0.008 | |
Crystal data top
| C0.4AlCa2O9.2 | V = 1329.84 (4) Å3 |
| Mr = 259.14 | Z = 6 |
| Trigonal, R3c | Synchrotron radiation, λ = 0.8264 (3) Å |
| a = 5.7534 (1) Å | T = 347 K |
| c = 46.389 (1) Å | ?, ? × ? × ? mm |
Data collection top
Synchrotron Diamond I11 (Great Britain)
diffractometer | |
Refinement top
| Rp = 0.030 | χ2 = 40.069 |
| Rwp = 0.050 | ? data points |
| Rexp = 0.008 | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Ca1 | 0.66667 | 0.33333 | 0.67930 (2) | 0.0231 (4)* | |
| Al1 | 0 | 0 | 0 | 0.0127 (5)* | |
| O1 | 0.3051 (3) | 0.0586 (3) | 0.52121 (3) | 0.0151 (5)* | |
| O2 | 0.66667 | 0.33333 | 0.93091 (6) | 0.0630 (10)* | |
| O3 | 0.220 (3) | 0 | 0.75 | 0.025 (2)* | 0.40 |
| C1 | 0.66667 | 0.33333 | 0.083333 | 0.025 (2)* | 0.40 |
Geometric parameters (Å, º) top
| Ca1—O1i | 2.374 (2) | Al1—O1iv | 1.890 (2) |
| Ca1—O1ii | 2.450 (2) | C1—O3v | 1.266 (4) |
| Ca1—O2iii | 2.619 (3) | | |
| | | |
| O1iv—Al1—O1vi | 180.0 | O1xiii—Ca1—O1xiv | 146.32 (5) |
| O1vii—Al1—O1viii | 180.0 | O1xiv—Ca1—O1xii | 117.25 (3) |
| O1ix—Al1—O1x | 180.0 | O1i—Ca1—O1xiv | 117.25 (3) |
| O1iv—Al1—O1vii | 95.36 (6) | O1i—Ca1—O1xii | 117.25 (3) |
| O1iv—Al1—O1ix | 95.36 (6) | O1xi—Ca1—O1xiv | 86.21 (6) |
| O1vii—Al1—O1ix | 95.36 (6) | O1xiii—Ca1—O1i | 86.21 (6) |
| O1vi—Al1—O1viii | 95.36 (6) | O1ii—Ca1—O1xii | 86.21 (6) |
| O1vi—Al1—O1x | 95.36 (6) | O1xiii—Ca1—O1xi | 83.35 (5) |
| O1viii—Al1—O1x | 95.36 (6) | O1xiii—Ca1—O1ii | 83.35 (5) |
| O1iv—Al1—O1viii | 84.64 (6) | O1xi—Ca1—O1ii | 83.35 (5) |
| O1iv—Al1—O1x | 84.64 (6) | O1xiii—Ca1—O1xii | 63.65 (8) |
| O1vii—Al1—O1vi | 84.64 (6) | O1xi—Ca1—O1i | 63.65 (8) |
| O1vii—Al1—O1x | 84.64 (6) | O1ii—Ca1—O1xiv | 63.65 (8) |
| O1ix—Al1—O1vi | 84.64 (6) | O1xii—Ca1—O2iii | 80.35 (4) |
| O1ix—Al1—O1viii | 84.64 (6) | Al1iv—O1—Ca1xv | 104.00 (7) |
| O1xi—Ca1—O1xii | 146.32 (5) | Ca1xiii—O1—Ca1xv | 93.79 (6) |
| O1ii—Ca1—O1i | 146.32 (5) | O3v—C1—O3xvi | 120 |
| Symmetry codes: (i) −y+1/3, −x+2/3, z+1/6; (ii) −x+4/3, −x+y+2/3, −z+7/6; (iii) −x+4/3, −y+2/3, −z+5/3; (iv) y, x, −z+1/2; (v) x+2/3, y+1/3, z−2/3; (vi) −y, −x, z−1/2; (vii) x−y, −y, −z+1/2; (viii) −x+y, y, z−1/2; (ix) −x, −x+y, −z+1/2; (x) x, x−y, z−1/2; (xi) x−y+1/3, −y+2/3, −z+7/6; (xii) x+1/3, x−y−1/3, z+1/6; (xiii) y+1/3, x−1/3, −z+7/6; (xiv) −x+y+4/3, y+2/3, z+1/6; (xv) −y+2/3, −x+1/3, z−1/6; (xvi) −y+2/3, x−y+1/3, z−2/3. |
(Hc) Tetracalcium dialuminium dodecahydroxide hemicarbonate hydroxide n-hydrate
top
Crystal data top
| C0.25AlCa2O9.25 | Z = 6 |
| Mr = 258.13 | F(000) = 771.0 |
| Trigonal, R3c | Dx = 1.824 Mg m−3 |
| Hall symbol: -R 3 2"c | Synchrotron radiation, λ = 0.8264 (3) Å |
| a = 5.7757 (1) Å | T = 297 K |
| c = 48.812 (2) Å | white |
| V = 1410.14 (7) Å3 | ?, ? × ? × ? mm |
Data collection top
Synchrotron Diamond I11 (Great Britain)
diffractometer | |
Refinement top
| Rp = 0.029 | χ2 = 27.384 |
| Rwp = 0.040 | ? data points |
| Rexp = 0.008 | |
Crystal data top
| C0.25AlCa2O9.25 | V = 1410.14 (7) Å3 |
| Mr = 258.13 | Z = 6 |
| Trigonal, R3c | Synchrotron radiation, λ = 0.8264 (3) Å |
| a = 5.7757 (1) Å | T = 297 K |
| c = 48.812 (2) Å | ?, ? × ? × ? mm |
Data collection top
Synchrotron Diamond I11 (Great Britain)
diffractometer | |
Refinement top
| Rp = 0.029 | χ2 = 27.384 |
| Rwp = 0.040 | ? data points |
| Rexp = 0.008 | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Al1 | 0 | 0 | 0 | 0.0127 (7)* | |
| Ca1 | 0.66667 | 0.33333 | 0.67796 (4) | 0.0214 (4)* | |
| O1 | 0.3012 (3) | 0.0484 (4) | 0.52040 (6) | 0.0224 (5)* | |
| O2 | 0.66667 | 0.33333 | 0.93875 (8) | 0.063 (2)* | |
| O3 | 0.219 (2) | 0 | 0.75 | 0.025 (4)* | 0.25 |
| O4 | 0.565 | 0.474 | 0.094 | 0.025 (7)* | 0.08333 |
| C1 | 0.66667 | 0.33333 | 0.08333 | 0.025 (4)* | 0.25 |
Geometric parameters (Å, º) top
| Ca1—O1i | 2.346 (3) | C1—O3v | 1.265 (9) |
| Ca1—O1ii | 2.467 (3) | C1—O4 | 1.324 |
| Ca1—O2iii | 2.439 (4) | O3—O4vi | 0.745 (3) |
| Al1—O1iv | 1.900 (3) | O4—O4vii | 1.456 |
| | | |
| O1iv—Al1—O1viii | 180.0 | O1i—Ca1—O1xiv | 116.50 (5) |
| O1ix—Al1—O1x | 180.0 | O1i—Ca1—O1xvi | 116.50 (5) |
| O1xi—Al1—O1xii | 180.0 | O1xiv—Ca1—O1xvi | 116.50 (5) |
| O1xiii—Ca1—O1xiv | 148.60 (8) | O1i—Ca1—O2iii | 79.08 (8) |
| O1xv—Ca1—O1xvi | 148.60 (8) | O1xiv—Ca1—O2iii | 79.08 (8) |
| O1ii—Ca1—O1i | 148.60 (8) | O1xvi—Ca1—O2iii | 79.08 (8) |
| O1xv—Ca1—O2iii | 128.85 (6) | O3v—C1—O3xvii | 120.0 |
| O1xiii—Ca1—O1i | 86.29 (9) | O3v—C1—O3xviii | 120.0 |
| O1xv—Ca1—O1xiv | 86.29 (9) | O3xvii—C1—O3xviii | 120.0 |
| O1ii—Ca1—O1xvi | 86.29 (9) | O3xix—O3—O3xx | 60 |
| Symmetry codes: (i) −y+1/3, −x+2/3, z+1/6; (ii) −x+4/3, −x+y+2/3, −z+7/6; (iii) −x+4/3, −y+2/3, −z+5/3; (iv) y, x, −z+1/2; (v) x+2/3, y+1/3, z−2/3; (vi) −x+y+1/3, −x+2/3, z+2/3; (vii) −x+4/3, −x+y+2/3, −z+1/6; (viii) −y, −x, z−1/2; (ix) x−y, −y, −z+1/2; (x) −x+y, y, z−1/2; (xi) −x, −x+y, −z+1/2; (xii) x, x−y, z−1/2; (xiii) y+1/3, x−1/3, −z+7/6; (xiv) −x+y+4/3, y+2/3, z+1/6; (xv) x−y+1/3, −y+2/3, −z+7/6; (xvi) x+1/3, x−y−1/3, z+1/6; (xvii) −y+2/3, x−y+1/3, z−2/3; (xviii) −x+y+2/3, −x+1/3, z−2/3; (xix) −y, x−y, z; (xx) −x+y, −x, z. |
Experimental details
| | (cHc) | (Hc) |
| Crystal data |
| Chemical formula | C0.4AlCa2O9.2 | C0.25AlCa2O9.25 |
| Mr | 259.14 | 258.13 |
| Crystal system, space group | Trigonal, R3c | Trigonal, R3c |
| Temperature (K) | 347 | 297 |
| a, c (Å) | 5.7534 (1), 46.389 (1) | 5.7757 (1), 48.812 (2) |
| V (Å3) | 1329.84 (4) | 1410.14 (7) |
| Z | 6 | 6 |
| Radiation type | Synchrotron, λ = 0.8264 (3) Å | Synchrotron, λ = 0.8264 (3) Å |
| Specimen shape, size (mm) | ?, ? × ? × ? | ?, ? × ? × ? |
| |
| Data collection |
| Diffractometer | Synchrotron Diamond I11 (Great Britain)
diffractometer | Synchrotron Diamond I11 (Great Britain)
diffractometer |
| Specimen mounting | ? | ? |
| Data collection mode | ? | ? |
| Scan method | ? | ? |
| 2θ values (°) | 2θmin = ? 2θmax = ? 2θstep = ? | 2θmin = ? 2θmax = ? 2θstep = ? |
| |
| Refinement |
| R factors and goodness of fit | Rp = 0.030, Rwp = 0.050, Rexp = 0.008, χ2 = 40.069 | Rp = 0.029, Rwp = 0.040, Rexp = 0.008, χ2 = 27.384 |
| No. of data points | ? | ? |
| No. of parameters | ? | ? |
| No. of restraints | ? | ? |

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
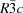 space group of the trigonal crystal system. Hc has the unit-cell parameters a = 5.7757 (1) and c = 48.812 (2) Å, and cHc the unit-cell parameters a = 5.7534 (1) and c = 46.389 (1) Å. The two crystal structures are composed of positively charged main layers, [Ca4Al2(OH)12]2+, and negatively charged interlayers, [OH2n(CO3)1 - n·4H2O]2-. The structure of the main layers is typical of the AFm family. Conversely, the interlayer region has a characteristic structure built up from water molecules and statistically distributed anions. In the interlayer, the Hc carbonate and hydroxyl anions are distributed in a 0.25:0.5 ratio, whereas the ratio of the anions in the cHc interlayers is 0.4:0.2.
space group of the trigonal crystal system. Hc has the unit-cell parameters a = 5.7757 (1) and c = 48.812 (2) Å, and cHc the unit-cell parameters a = 5.7534 (1) and c = 46.389 (1) Å. The two crystal structures are composed of positively charged main layers, [Ca4Al2(OH)12]2+, and negatively charged interlayers, [OH2n(CO3)1 - n·4H2O]2-. The structure of the main layers is typical of the AFm family. Conversely, the interlayer region has a characteristic structure built up from water molecules and statistically distributed anions. In the interlayer, the Hc carbonate and hydroxyl anions are distributed in a 0.25:0.5 ratio, whereas the ratio of the anions in the cHc interlayers is 0.4:0.2.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig4thm.gif)
![[Figure 5]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig5thm.gif)
![[Figure 6]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig6thm.gif)
![[Figure 7]](http://journals.iucr.org/b/issues/2012/05/00/eb5017/eb5017fig7thm.gif)

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


