The chemical, electrochemical, optical and electro-optical properties of titanium oxyfluoride, TiOF2, have led to interest in this compound for a number of applications. Prior analyses have indicated that TiOF2 possesses a simple cubic structure (space group Pm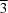 m) at room temperature. Three-dimensional nanostructured assemblies of polycrystalline TiOF2 have recently been synthesized via chemical conversion of intricate SiO2 structures by metathetic reaction with TiF4(g). Rietveld analysis has been used to evaluate the structure of the TiOF2 product formed by such reaction at 623 K. Unlike prior reports, this TiOF2 product possessed a hexagonal structure (space group R
m) at room temperature. Three-dimensional nanostructured assemblies of polycrystalline TiOF2 have recently been synthesized via chemical conversion of intricate SiO2 structures by metathetic reaction with TiF4(g). Rietveld analysis has been used to evaluate the structure of the TiOF2 product formed by such reaction at 623 K. Unlike prior reports, this TiOF2 product possessed a hexagonal structure (space group R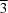 c) at room temperature. Upon heating through 333-338 K, the hexagonal TiOF2 polymorph converted into cubic (Pm
c) at room temperature. Upon heating through 333-338 K, the hexagonal TiOF2 polymorph converted into cubic (Pm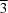 m) TiOF2. Differential scanning calorimetry and X-ray diffraction analyses have been used to evaluate this thermally induced phase transformation.
m) TiOF2. Differential scanning calorimetry and X-ray diffraction analyses have been used to evaluate this thermally induced phase transformation.
Supporting information
Crystallographic Information File (CIF) https://doi.org/10.1107/S0021889810016730/ce5082sup1.cif | |
Rietveld powder data file (CIF format) https://doi.org/10.1107/S0021889810016730/ce5082sup2.rtv |
Computing details top
Titanium oxydifluoride top
Crystal data top
| TiF2O | Z = 6 |
| Mr = 101.86 | Dx = 3.114 Mg m−3 |
| Hexagonal, R3c | Cu Kα1 radiation, λ = 1.54060 Å |
| a = 5.3325 (1) Å | T = 298 K |
| c = 13.2321 (4) Å | × × mm |
| V = 325.86 (1) Å3 |
Crystal data top
| TiF2O | V = 325.86 (1) Å3 |
| Mr = 101.86 | Z = 6 |
| Hexagonal, R3c | Cu Kα1 radiation |
| a = 5.3325 (1) Å | T = 298 K |
| c = 13.2321 (4) Å | × × mm |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top
| x | y | z | Uiso*/Ueq | Occ. (<1) | |
| Ti | 0.000000 | 0.000000 | 0.000000 | 0.010492* | |
| F | 0.5486 (5) | 0.000000 | 0.250000 | 0.000000* | 0.666667 |
| O | 0.5486 (5) | 0.000000 | 0.250000 | 0.000762* | 0.333333 |
Atomic displacement parameters (Å2) top
| U11 | U22 | U33 | U12 | U13 | U23 | |
| Ti | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
| F | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
| O | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
Geometric parameters (Å, º) top
| Ti—F | 1.9112 (3) | F—F | 2.702 (2) |
| Ti—F | 1.911 (2) | F—F | 2.702 (2) |
| Ti—F | 1.911 (2) | F—F | 2.704 (2) |
| Ti—F | 1.911 (2) | F—F | 2.704 (2) |
| Ti—F | 1.911 (2) | F—F | 2.704 (1) |
| Ti—F | 1.9112 (3) | F—F | 2.704 (1) |
| F—F | 2.702 (2) | F—F | 3.442 (2) |
| F—F | 2.702 (2) | F—F | 3.442 (2) |
| F—Ti—F | 90.04 (6) | F—F—F | 140.4 (2) |
| F—Ti—F | 89.96 (7) | F—F—F | 50.43 (3) |
| F—Ti—F | 89.96 (6) | F—F—F | 93.02 (9) |
| F—Ti—F | 180.0 (2) | F—F—F | 137.9 (2) |
| F—Ti—F | 90.04 (7) | F—F—F | 50.48 (4) |
| F—Ti—F | 90.0 (1) | F—F—Ti | 45.02 (3) |
| F—Ti—F | 180.0 (4) | F—F—F | 127.1 (1) |
| F—Ti—F | 89.96 (6) | F—F—F | 169.0 (8) |
| F—Ti—F | 90.0 (1) | F—F—F | 60.05 (4) |
| F—Ti—F | 90.0 (1) | F—F—F | 59.98 (3) |
| F—Ti—F | 90.04 (7) | F—F—F | 90.00 (8) |
| F—Ti—F | 180.0 (1) | F—F—F | 124.6 (2) |
| F—Ti—F | 90.04 (6) | F—F—Ti | 94.31 (7) |
| F—Ti—F | 90.0 (1) | F—F—F | 94.62 (8) |
| F—Ti—F | 89.96 (7) | F—F—F | 140.4 (2) |
| F—F—Ti | 44.98 (3) | F—F—F | 84.55 (7) |
| F—F—F | 101.61 (8) | F—F—F | 101.61 (8) |
| F—F—F | 100.91 (7) | F—F—F | 137.9 (2) |
| F—F—F | 50.43 (3) | F—F—F | 93.02 (9) |
| F—F—Ti | 143.3 (1) | Ti—F—F | 145.9 (2) |
| F—F—F | 59.98 (3) | Ti—F—F | 124.1 (1) |
| F—F—F | 90.00 (7) | Ti—F—F | 45.02 (4) |
| F—F—F | 112.2 (1) | Ti—F—F | 44.98 (3) |
| F—F—F | 139.1 (1) | Ti—F—F | 44.98 (5) |
| F—F—F | 160.9 (4) | Ti—F—F | 122.3 (1) |
| F—F—F | 60.00 (4) | F—F—F | 60.05 (4) |
| Ti—F—F | 94.31 (7) | F—F—F | 169.0 (8) |
| Ti—F—F | 145.9 (2) | F—F—F | 100.91 (7) |
| Ti—F—F | 95.40 (8) | F—F—F | 124.6 (2) |
| Ti—F—Ti | 164.4 (1) | F—F—F | 90.00 (8) |
| Ti—F—F | 45.02 (3) | F—F—F | 114.4 (1) |
| Ti—F—F | 45.02 (4) | F—F—F | 112.2 (1) |
| Ti—F—F | 124.1 (1) | F—F—F | 79.09 (7) |
| Ti—F—F | 143.3 (1) | F—F—F | 59.98 (5) |
| Ti—F—F | 122.3 (1) | F—F—F | 90.00 (7) |
| Ti—F—F | 44.98 (5) | F—F—F | 59.98 (5) |
| F—F—F | 94.62 (8) | F—F—F | 79.09 (7) |
| F—F—F | 102.78 (9) | F—F—F | 60.00 (4) |
| F—F—Ti | 95.40 (8) | F—F—F | 160.9 (4) |
| F—F—F | 50.48 (4) | F—F—F | 100.9 (1) |
| F—F—F | 84.55 (7) |
Experimental details
| Crystal data | |
| Chemical formula | TiF2O |
| Mr | 101.86 |
| Crystal system, space group | Hexagonal, R3c |
| Temperature (K) | 298 |
| a, c (Å) | 5.3325 (1), 13.2321 (4) |
| V (Å3) | 325.86 (1) |
| Z | 6 |
| Radiation type | Cu Kα1 |
| µ (mm−1) | ? |
| Crystal size (mm) | × × |
| Data collection | |
| Diffractometer | PANalytical Alpha1 diffractometer |
| Absorption correction | ? |
| No. of measured, independent and observed (?) reflections | ?, ?, 77 |
| Rint | ? |
| Refinement | |
| R[F2 > 2σ(F2)], wR(F2), S | 9.70, 12.64, 1.64 |
| No. of reflections | ? |
| No. of parameters | ? |
| No. of restraints | ? |
| Δρmax, Δρmin (e Å−3) | ?, ? |

 journal menu
journal menu











