Magnesium hydroxide sulfate [2MgSO4.Mg(OH)2; MHS] is tetragonal with a = 7.454 (1), c = 12.885 (2) Å, V = 716.0 Å3, space group P43212, Z = 4 and Dx 2.774 g cm−3. The structure (single-crystal X-ray, R = 0.025, wR = 0.023) comprises spiral (43) single chains of corner-shared Mg(2) octahedra cross-linked by SO4 tetrahedra and face-shared Mg(1) octahedra. A linear ternary group of face-shared Mg octahedra [Mg(2)—Mg(1)—Mg(2)] alternates with an unoccupied octahedral position in rows along [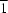 10]. A crystal of MHS was grown hydrothermally (0.15 GPa, 673 K) from gold-bearing Mg—S—O—H fluid. The MHS structure, with Mg(1) octahedra sharing two octahedral faces with Mg(2) octahedra, revises the structure assumed for the complex solid solution magnesium hydroxide sulfate hydrate and the related ocean-floor mineral caminite [2MgSO4.xMg(OH)2.(2 − 2x)H2O; 0.5 < x < 1.0]. The substitution reaction appears to be Mg(1)2+ ⇋ 2H+. The H-substituted MHS structure is distinguished from that of kieserite (MgSO4.H2O), which has straight single chains of corner-shared Mg octahedra.
10]. A crystal of MHS was grown hydrothermally (0.15 GPa, 673 K) from gold-bearing Mg—S—O—H fluid. The MHS structure, with Mg(1) octahedra sharing two octahedral faces with Mg(2) octahedra, revises the structure assumed for the complex solid solution magnesium hydroxide sulfate hydrate and the related ocean-floor mineral caminite [2MgSO4.xMg(OH)2.(2 − 2x)H2O; 0.5 < x < 1.0]. The substitution reaction appears to be Mg(1)2+ ⇋ 2H+. The H-substituted MHS structure is distinguished from that of kieserite (MgSO4.H2O), which has straight single chains of corner-shared Mg octahedra.

 journal menu
journal menu











