
Tetrachlorocobaltates(II) of diprotonated α,ω-diaminoalkanes with the formula [NH
3(CH
2)
nNH
3]CoCl
4, where
n = 5 (cadaverine; 1,5-pentanediammonium tetrachlorocobaltate), 8 (1,8-octanediammonium tetrachlorocobaltate) and 10 (1,10-decanediammonium tetrachlorocobaltate), were prepared. The compounds were studied by mass spectrometry, FT-IR and visible spectroscopy, magnetic susceptibility techniques and thermal analysis. The compounds contain the tetrahedral tetrachlorocobaltate(II) ion and the corresponding diprotonated diamine (cadaverine, 1,8-octamethylenediamine and 1,10-decamethylenediamine). The compound corresponding to cadaverine crystallizes in the monoclinic space group
P2
1/
c, with lattice parameters
a = 7.1633 (7),
b = 15.940 (3),
c = 11.137 (2) Å, β = 98.44 (1)° and
Z = 4. Its crystal structure contains slightly distorted tetrahedral CoCl
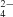
ions: the largest difference in Co—Cl bond lengths is 0.029 Å and the largest difference in Cl—Co—Cl angles is 7.76°. The compound also contains diprotonated cadaverine ions. An extensive hydrogen-bonding network connects these ions. The slightly positive deviations of the magnetic susceptibility from the Curie–Weiss law are in agreement with the
4A2 ground state for the tetrachlorocobaltate anion. The compounds with eight and ten C atoms show phase transitions in the solid state and a greater complexity is observed in their differential scanning calorimetry curves. Unfortunately, no suitable single crystals of these could be obtained.
Supporting information
 | Crystallographic Information File (CIF)
Contains datablocks comp2, global |
 | Structure factor file (SHELXL table format)
Supplementary material |
CCDC reference: 138548
Program(s) used to solve structure: SHELXS86 (Sheldrick, 1990); program(s) used to refine structure: SHELXL93 (Sheldrick, 1993); molecular graphics: ORTEP; software used to prepare material for publication: SHELXL93 (Sheldrick, 1993).
1,5-pentanediammonium tetrachlorocobaltate
top
Crystal data top
| [(NH3)2C5H10]·CoCl4 | F(000) = 620 |
| Mr = 304.93 | Dx = 1.610 Mg m−3 |
| Monoclinic, P21/c | Cu Kα radiation, λ = 1.5418 Å |
| a = 7.1633 (7) Å | Cell parameters from 54 reflections |
| b = 15.940 (3) Å | θ = 4.9–66.1° |
| c = 11.137 (2) Å | µ = 18.18 mm−1 |
| β = 98.44 (1)° | T = 293 K |
| V = 1257.9 (3) Å3 | Rectangular, blue |
| Z = 4 | 0.47 × 0.27 × 0.13 mm |
Data collection top
Seifert XRD 3000
diffractometer | 1586 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.086 |
| Graphite monochromator | θmax = 66.1°, θmin = 4.9° |
| 2θ–ω scans | h = −8→8 |
| Absorption correction: empirical (using intensity measurements) | k = 0→19 |
| Tmin = 0.073, Tmax = 0.203 | l = 0→13 |
| 2148 measured reflections | 2 standard reflections every 100 reflections |
| 2147 independent reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.063 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.178 | Iso |
| S = 1.01 | Calculated w = 1/[σ2(Fo2) + (0.123P)2]
where P = (Fo2 + 2Fc2)/3 |
| 2147 reflections | (Δ/σ)max = −0.111 |
| 157 parameters | Δρmax = 0.85 e Å−3 |
| 0 restraints | Δρmin = −0.79 e Å−3 |
Special details top
Experimental. The large bond distances N(1)—H(112), N(2)—H(221), N(2)—H(222) and
N(2)—H(223) were distorted by the hydrogen bond involved. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Co1 | 0.38109 (15) | 0.37752 (6) | −0.19215 (9) | 0.0272 (3) | |
| Cl1 | 0.4432 (3) | 0.47207 (10) | −0.3351 (2) | 0.0392 (5) | |
| Cl2 | 0.4330 (3) | 0.24663 (10) | −0.2661 (2) | 0.0403 (5) | |
| Cl3 | 0.0821 (2) | 0.38592 (11) | −0.1477 (2) | 0.0395 (5) | |
| Cl4 | 0.5827 (2) | 0.38967 (10) | −0.0163 (2) | 0.0353 (4) | |
| N1 | 0.6059 (10) | 0.3216 (5) | 0.5061 (7) | 0.040 (2) | |
| N2 | 1.2147 (9) | 0.4502 (4) | 0.1282 (6) | 0.043 (2) | |
| C1 | 0.8109 (12) | 0.3114 (6) | 0.5539 (8) | 0.045 (2) | |
| C2 | 0.9315 (13) | 0.3705 (5) | 0.4952 (7) | 0.046 (2) | |
| C3 | 0.9344 (11) | 0.3554 (4) | 0.3613 (7) | 0.038 (2) | |
| C4 | 1.0719 (11) | 0.4118 (5) | 0.3095 (7) | 0.034 (2) | |
| C5 | 1.0692 (11) | 0.3984 (4) | 0.1769 (7) | 0.032 (2) | |
| H11 | 0.825 (18) | 0.253 (8) | 0.543 (12) | 0.12 (5)* | |
| H12 | 0.830 (12) | 0.316 (6) | 0.632 (9) | 0.06 (3)* | |
| H21 | 1.054 (13) | 0.369 (5) | 0.532 (8) | 0.05 (3)* | |
| H22 | 0.8926 | 0.4301 | 0.5102 | 0.09 (4)* | |
| H31 | 0.8044 | 0.3607 | 0.3131 | 0.10 (4)* | |
| H32 | 0.965 (14) | 0.288 (7) | 0.350 (10) | 0.09 (3)* | |
| H41 | 1.175 (19) | 0.407 (8) | 0.357 (12) | 0.12 (5)* | |
| H42 | 1.039 (12) | 0.474 (6) | 0.317 (8) | 0.06 (3)* | |
| H51 | 1.102 (12) | 0.352 (6) | 0.167 (8) | 0.05 (3)* | |
| H52 | 0.931 (13) | 0.411 (5) | 0.141 (8) | 0.06 (3)* | |
| H111 | 0.591 (8) | 0.307 (4) | 0.426 (6) | 0.018 (16)* | |
| H112 | 0.575 (13) | 0.378 (6) | 0.538 (9) | 0.07 (3)* | |
| H113 | 0.564 (17) | 0.290 (9) | 0.550 (12) | 0.11 (5)* | |
| H221 | 1.3440 | 0.4340 | 0.1704 | 0.044* | |
| H222 | 1.1931 | 0.5108 | 0.1417 | 0.12 (5)* | |
| H223 | 1.2130 | 0.4405 | 0.0382 | 0.08 (3)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Co1 | 0.0298 (6) | 0.0143 (5) | 0.0339 (6) | −0.0009 (4) | −0.0074 (4) | 0.0006 (4) |
| Cl1 | 0.0523 (11) | 0.0213 (8) | 0.0399 (9) | −0.0038 (7) | −0.0072 (7) | 0.0099 (7) |
| Cl2 | 0.0587 (12) | 0.0162 (8) | 0.0441 (10) | −0.0042 (7) | 0.0012 (8) | −0.0066 (7) |
| Cl3 | 0.0266 (9) | 0.0377 (10) | 0.0506 (11) | −0.0002 (7) | −0.0061 (7) | −0.0077 (8) |
| Cl4 | 0.0354 (9) | 0.0220 (8) | 0.0420 (9) | −0.0017 (6) | −0.0161 (7) | 0.0026 (7) |
| N1 | 0.044 (4) | 0.037 (4) | 0.036 (4) | −0.011 (3) | 0.001 (3) | −0.005 (3) |
| N2 | 0.035 (3) | 0.049 (4) | 0.043 (4) | −0.013 (3) | −0.003 (3) | −0.003 (3) |
| C1 | 0.042 (5) | 0.044 (5) | 0.044 (5) | −0.012 (4) | −0.010 (4) | 0.016 (4) |
| C2 | 0.052 (5) | 0.033 (4) | 0.045 (5) | −0.017 (4) | −0.015 (4) | 0.003 (3) |
| C3 | 0.045 (4) | 0.019 (3) | 0.044 (4) | −0.006 (3) | −0.011 (3) | 0.000 (3) |
| C4 | 0.036 (4) | 0.025 (4) | 0.038 (4) | −0.005 (3) | −0.003 (3) | −0.005 (3) |
| C5 | 0.032 (4) | 0.017 (3) | 0.044 (4) | −0.003 (3) | −0.006 (3) | −0.001 (3) |
Geometric parameters (Å, º) top
| Co1—Cl1 | 2.283 (2) | N1—H113 | 0.79 (13) |
| Co1—Cl2 | 2.293 (2) | N2—H221 | 1.009 (6) |
| Co1—Cl3 | 2.271 (2) | N2—H222 | 0.993 (7) |
| Co1—Cl4 | 2.264 (2) | N2—H223 | 1.012 (6) |
| Co1—Cl4 | 2.264 (2) | C1—H11 | 0.95 (13) |
| Co1—Cl3 | 2.271 (2) | C1—H12 | 0.86 (9) |
| N1—C1 | 1.495 (10) | C2—H21 | 0.91 (9) |
| N2—C5 | 1.492 (9) | C2—H22 | 1.010 (9) |
| C1—C2 | 1.492 (11) | C3—H31 | 1.008 (8) |
| C2—C3 | 1.514 (11) | C3—H32 | 1.10 (11) |
| C3—C4 | 1.510 (10) | C4—H41 | 0.85 (13) |
| C4—C5 | 1.489 (10) | C4—H42 | 1.02 (10) |
| N1—H111 | 0.92 (7) | C5—H51 | 0.79 (9) |
| N1—H112 | 1.01 (10) | C5—H52 | 1.03 (9) |
| | | |
| Cl4—Co1—Cl3 | 107.97 (8) | N1—C1—H11 | 100 (8) |
| Cl4—Co1—Cl1 | 112.44 (7) | C2—C1—H12 | 112 (6) |
| Cl3—Co1—Cl1 | 113.55 (8) | N1—C1—H12 | 111 (6) |
| Cl4—Co1—Cl2 | 105.79 (7) | H11—C1—H12 | 102 (10) |
| Cl3—Co1—Cl2 | 109.84 (8) | C1—C2—H21 | 111 (5) |
| Cl1—Co1—Cl2 | 106.96 (8) | C3—C2—H21 | 107 (6) |
| C2—C1—N1 | 112.2 (7) | C1—C2—H22 | 109.3 (9) |
| C1—C2—C3 | 115.0 (7) | C3—C2—H22 | 110.9 (7) |
| C4—C3—C2 | 112.7 (6) | H21—C2—H22 | 103 (5) |
| C5—C4—C3 | 112.3 (6) | C4—C3—H31 | 110.5 (7) |
| C4—C5—N2 | 111.9 (6) | C2—C3—H31 | 111.5 (7) |
| C1—N1—H111 | 107 (4) | C4—C3—H32 | 112 (5) |
| C1—N1—H112 | 103 (5) | C2—C3—H32 | 108 (6) |
| H111—N1—H112 | 125 (7) | H31—C3—H32 | 102 (6) |
| C1—N1—H113 | 99 (9) | C5—C4—H41 | 120 (9) |
| H111—N1—H113 | 116 (10) | C3—C4—H41 | 105 (9) |
| H112—N1—H113 | 103 (10) | C5—C4—H42 | 105 (5) |
| C5—N2—H221 | 109.4 (6) | C3—C4—H42 | 112 (5) |
| C5—N2—H222 | 110.5 (6) | H41—C4—H42 | 104 (10) |
| H221—N2—H222 | 109.3 (6) | C4—C5—H51 | 108 (6) |
| C5—N2—H223 | 111.6 (6) | N2—C5—H51 | 103 (6) |
| H221—N2—H223 | 107.4 (6) | C4—C5—H52 | 104 (5) |
| H222—N2—H223 | 108.6 (6) | N2—C5—H52 | 115 (5) |
| C2—C1—H11 | 119 (8) | H51—C5—H52 | 115 (8) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H111···Cl2i | 0.92 (7) | 2.42 (6) | 3.29 (1) | 155 |
| N1—H112···Cl1ii | 1.00 (10) | 2.3 (1) | 3.29 (1) | 157 |
| N1—H113···Cl2ii | 0.79 (13) | 2.5 (1) | 3.21 (1) | 155 |
| N2—H223···Cl3iii | 1.01 | 2.31 | 3.25 (1) | 152 |
| Symmetry codes: (i) x, −y+1/2, z+1/2; (ii) x, y, z+1; (iii) x+1, y, z. |
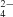 ions: the largest difference in Co—Cl bond lengths is 0.029 Å and the largest difference in Cl—Co—Cl angles is 7.76°. The compound also contains diprotonated cadaverine ions. An extensive hydrogen-bonding network connects these ions. The slightly positive deviations of the magnetic susceptibility from the Curie–Weiss law are in agreement with the 4A2 ground state for the tetrachlorocobaltate anion. The compounds with eight and ten C atoms show phase transitions in the solid state and a greater complexity is observed in their differential scanning calorimetry curves. Unfortunately, no suitable single crystals of these could be obtained.
ions: the largest difference in Co—Cl bond lengths is 0.029 Å and the largest difference in Cl—Co—Cl angles is 7.76°. The compound also contains diprotonated cadaverine ions. An extensive hydrogen-bonding network connects these ions. The slightly positive deviations of the magnetic susceptibility from the Curie–Weiss law are in agreement with the 4A2 ground state for the tetrachlorocobaltate anion. The compounds with eight and ten C atoms show phase transitions in the solid state and a greater complexity is observed in their differential scanning calorimetry curves. Unfortunately, no suitable single crystals of these could be obtained.
 journal menu
journal menu











