Buy article online - an online subscription or single-article purchase is required to access this article.
Single crystals of strontium cobaltate, SrCoO
3-x, have been grown by the floating-zone method in an oxygen flow. The compound crystallizes with the cubic perovskite structure, with
Pm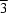 m
m symmetry, as determined by X-ray diffraction. Refinement of the O-atom site occupancy yields the chemical composition SrCoO
2.64 [
x = 0.36 (3)]. The anisotropic displacement ellipsoids of the O atoms suggest that their positional disorder occurs in a direction perpendicular to that of the Co-O bonds.
Supporting information
A single-crystal of the title compound was grown by the floating-zone method. The starting materials were powders of SrCO3 and CoO (99.9%). Stoichiometric amounts of the starting materials were mixed well, and the mixture was placed in a platinum crucible and heated at 1300 K for 10 h under a flow of oxygen. The reaction product was reground and pressed hydrostatically into a rod of 8 mm in diameter and 40 mm in length, which was then sintered at 1500 K for 1 h in an oxygen atmosphere. For the crystal growth, the sintered rod was put into an image-type floating-zone furnace, with a halogen lamp as a heat source. The crystal growth was carried out under oxygen at the flow rate of 1 l min−1. Upper and lower shafts were counter-rotated at the rate of 25 r.p.m. and the growth rate was 2 mm h−1. The number of Co4+ ions was analyzed by iodometric oxidation–reduction titration. The analytical results gave a chemical composition of 54.8 wt% SrO, 16.1 wt% CoO2 and 29.2 wt% Co2O3, with a total of 100.1 wt%, corresponding to a composition of Sr(Co4+0.34Co3+0.66)O2.67.
The maximum and minimum residual electron densities are located at the coordinates (0, 0, 0) and (0.46, 0.46, 0.46), respectively.
Data collection: WinAFC (Rigaku Corporation, 1999); cell refinement: WinAFC (Rigaku Corporation, 1999); data reduction: RADY (Sasaki, 1987); program(s) used to refine structure: RADY (Sasaki, 1987); molecular graphics: ATOMS for Windows (Dowty, 2000).
strontium cobalt(III, IV) oxide
top
Crystal data top
| SrCoO2.64 | Dx = 5.483 Mg m−3 |
| Mr = 188.79 | Mo Kα radiation, λ = 0.71069 Å |
| Cubic, Pm3m | Cell parameters from 25 reflections |
| Hall symbol: -P 4 2 3 | θ = 20.0–22.5° |
| a = 3.8530 (4) Å | µ = 30.22 mm−1 |
| V = 57.20 (2) Å3 | T = 296 K |
| Z = 1 | Sphere, black |
| F(000) = 86.12 | 0.08 mm (radius) |
Data collection top
Rigaku AFC-7R
diffractometer | Rint = 0.011 |
| ω–2θ scans | θmax = 60.0° |
Absorption correction: for a sphere
(Sasaki, 1987) | h = 0→9 |
| Tmin = 0.008, Tmax = 0.011 | k = 0→9 |
| 539 measured reflections | l = 0→9 |
| 120 independent reflections | 3 standard reflections every 100 reflections |
| 96 reflections with F > 3σ(F) | intensity decay: none |
Refinement top
| Refinement on F | σ w = 1/σ2(F) |
| R[F2 > 2σ(F2)] = 0.021 | (Δ/σ)max = 0.0001 |
| wR(F2) = 0.011 | Δρmax = 2.80 e Å−3 |
| S = 1.88 | Δρmin = −2.64 e Å−3 |
| 96 reflections | Extinction correction: isotropic Type I (Becker & Coppens, 1974a and 1974b) |
| 7 parameters | Extinction coefficient: 0.054 (2) 10-4 |
Crystal data top
| SrCoO2.64 | Z = 1 |
| Mr = 188.79 | Mo Kα radiation |
| Cubic, Pm3m | µ = 30.22 mm−1 |
| a = 3.8530 (4) Å | T = 296 K |
| V = 57.20 (2) Å3 | 0.08 mm (radius) |
Data collection top
Rigaku AFC-7R
diffractometer | 96 reflections with F > 3σ(F) |
Absorption correction: for a sphere
(Sasaki, 1987) | Rint = 0.011 |
| Tmin = 0.008, Tmax = 0.011 | 3 standard reflections every 100 reflections |
| 539 measured reflections | intensity decay: none |
| 120 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.021 | 7 parameters |
| wR(F2) = 0.011 | Δρmax = 2.80 e Å−3 |
| S = 1.88 | Δρmin = −2.64 e Å−3 |
| 96 reflections | |
Special details top
Experimental. The composition of the grown crystal was examined by means of a Jeol JCMA-733II electron microprobe analyzer and edta titration at a pH of 3. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Sr | 0 | 0 | 0 | 0.01400 (9) | |
| Co | 0.5 | 0.5 | 0.5 | 0.01009 (8) | |
| O | 0.5 | 0.5 | 0 | 0.025 (2) | 0.88 (1) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Sr | 0.0140 (2) | 0.0140 (2) | 0.0140 (2) | 0.0 | 0.0 | 0.0 |
| Co | 0.0101 (2) | 0.0101 (2) | 0.0101 (2) | 0.0 | 0.0 | 0.0 |
| O | 0.049 (4) | 0.0126 (11) | 0.0126 (11) | 0.0 | 0.0 | 0.0 |
Geometric parameters (Å, º) top
| Sr—O | 2.7245 (2) | O—Oi | 2.7245 (2) |
| Co—O | 1.9265 (2) | | |
| | | |
| O—Sr—Oii | 180.00 | O—Sr—Oix | 120.00 |
| O—Sr—Oiii | 90.00 | O—Sr—Ox | 60.00 |
| O—Sr—Oiv | 90.00 | O—Sr—Oxi | 60.00 |
| O—Sr—Ov | 120.00 | O—Co—Oi | 90.00 |
| O—Sr—Ovi | 120.00 | O—Co—Oxi | 90.00 |
| O—Sr—Ovii | 60.00 | O—Co—Oxii | 180.00 |
| O—Sr—Oi | 60.00 | O—Co—Oxiii | 90.00 |
| O—Sr—Oviii | 120.00 | O—Co—Oxiv | 90.00 |
| Symmetry codes: (i) z, x, y; (ii) −x, −y, z; (iii) −x, y, −z; (iv) x, −y, −z; (v) z, −x, −y; (vi) −z, −x, y; (vii) −z, x, −y; (viii) −y, z, −x; (ix) −y, −z, x; (x) y, −z, −x; (xi) y, z, x; (xii) x, y, z+1; (xiii) z+1, x, y; (xiv) y, z+1, x. |
Experimental details
| Crystal data |
| Chemical formula | SrCoO2.64 |
| Mr | 188.79 |
| Crystal system, space group | Cubic, Pm3m |
| Temperature (K) | 296 |
| a (Å) | 3.8530 (4) |
| V (Å3) | 57.20 (2) |
| Z | 1 |
| Radiation type | Mo Kα |
| µ (mm−1) | 30.22 |
| Crystal size (mm) | 0.08 (radius) |
| |
| Data collection |
| Diffractometer | Rigaku AFC-7R
diffractometer |
| Absorption correction | For a sphere
(Sasaki, 1987) |
| Tmin, Tmax | 0.008, 0.011 |
No. of measured, independent and
observed [F > 3σ(F)] reflections | 539, 120, 96 |
| Rint | 0.011 |
| (sin θ/λ)max (Å−1) | 1.219 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.021, 0.011, 1.88 |
| No. of reflections | 96 |
| No. of parameters | 7 |
| No. of restraints | ? |
| Δρmax, Δρmin (e Å−3) | 2.80, −2.64 |
Selected bond lengths (Å) top| Sr—O | 2.7245 (2) | O—Oi | 2.7245 (2) |
| Co—O | 1.9265 (2) | | |
| Symmetry code: (i) z, x, y. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
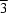 m symmetry, as determined by X-ray diffraction. Refinement of the O-atom site occupancy yields the chemical composition SrCoO2.64 [x = 0.36 (3)]. The anisotropic displacement ellipsoids of the O atoms suggest that their positional disorder occurs in a direction perpendicular to that of the Co-O bonds.
m symmetry, as determined by X-ray diffraction. Refinement of the O-atom site occupancy yields the chemical composition SrCoO2.64 [x = 0.36 (3)]. The anisotropic displacement ellipsoids of the O atoms suggest that their positional disorder occurs in a direction perpendicular to that of the Co-O bonds.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2004/05/00/bc1039/bc1039fig1thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



Strontium cobaltate, SrCoO3 − x, exists over a wide range of oxygen non-stoichiometry and several phases with different oxygen contents have been reported (Watanabe & Takeda, 1970). The cubic perovskite-type structure, with Pm3 m symmetry, is stable over the composition range x = 0.0–0.4 (Takeda et al., 1986). SrCoO3 contains low-spin Co4+ ions and is a ferromagnetic oxide, with a Curie temperature of 222 K (Taguchi et al., 1979), and a metallic conductor (Taguchi et al., 1980; Bezdicka et al., 1993). Co3+ and Co4+ ions coexist in the oxygen-deficient SrCoO3 − x phase. The magnetic and electric properties of that phase are sensitive to the oxygen deficiency (x), and the Curie temperature and the electric resistance both decrease with increasing x (Taguchi et al., 1979, 1980). Hence, investigating the structural variation of SrCoO3 − x as a function of x is important for the understanding of its physical properties. To date, only lattice parameters determined using powder samples have been reported and a full structure refinement, including displacement parameters, has not been published. The determination of these displacement parameters is important for understanding the behavior of oxygen vacancies in the structure.
The present structure refinement of SrCoO3 − x converged to an occupancy of 0.88 (1) for the O-atom site, yielding a chemical composition of Sr(Co4+0.28Co3+0.72)O2.64 [x = 0.36 (3)]. This result agrees well with the composition of Sr(Co4+0.34Co3+0.66)O2.67 obtained by chemical analysis, and also with the composition estimated from the correlation between lattice parameter and oxygen deficiency proposed by Takeda et al. (1986). The presence of oxygen vacancies is reflected in the larger displacement parameter of the O atom [Ueq = 0.025 (2) Å2]. The same observation has also been reported in other non-stoichiometric perovskites (e.g. Harrison et al., 1995). The strong anisotropy of the displacement ellipsoid in a direction perpendicular to the Co–O bonds (Fig. 1) suggests that positional disorder of the O atoms occurs in the same direction.