Crystal structures of 3,3,5,5-tetramethylglutarimide [3,3,5,5-tetramethylazacyclohexane-2,6-dione] (8) and the product of its reaction with phenyllithium, 2-hydroxy-2-phenyl-3,3,5,5-tetramethyl-6-piperidone (9), have been determined by X-ray structural methods [(8), monoclinic, P21/c, a = 7.715 (2), b = 11.136 (2), c = 11.707 (2) Å, 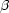 = 105.63 (3)°; (9), triclinic, P
= 105.63 (3)°; (9), triclinic, P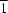 , a = 6.1685 (6), b = 11.1475 (10), c = 11.526 (2) Å,
, a = 6.1685 (6), b = 11.1475 (10), c = 11.526 (2) Å,  = 117.100 (10),
= 117.100 (10), 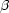 = 103.390(10),
= 103.390(10), 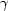 = 91.288 (7)°]. Molecular structures of three different glutarimides are discussed. Their energy optimal structures were determined with the use of ab initio calculations. The results of crystallographic and theoretical studies show that the conformational changes in glutarimide introduced by substitution with methyl groups resulted in electron-charge distribution changes. These differences in charge distribution are the reason for the observed variation in yields of the reaction of glutarimides with phenyllithium.
= 91.288 (7)°]. Molecular structures of three different glutarimides are discussed. Their energy optimal structures were determined with the use of ab initio calculations. The results of crystallographic and theoretical studies show that the conformational changes in glutarimide introduced by substitution with methyl groups resulted in electron-charge distribution changes. These differences in charge distribution are the reason for the observed variation in yields of the reaction of glutarimides with phenyllithium.

 journal menu
journal menu











