
Both maleic and fumaric acid readily form adducts or complexes with other organic molecules. The 1:1 adduct formed by quinolin-8-ol (oxine) with maleic and fumaric acid are salts, namely 8-hydroxyquinolinium hydrogen maleate, C
9H
8NO
+·C
4H
3O
4-, (I), and 8-hydroxyquinolinium hydrogen fumarate, C
9H
8NO
+·C
4H
3O
4-, (II). The cations and anions of both salts are linked by ionic N
+-H

O
- hydrogen bonds. The maleate salt crystallizes in the space group
P2
12
12
1, while the fumarate salt crystallizes in
P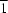
. The maleic and fumaric acids in their complex forms exist as semimaleate and semifumarate ions (mono-ionized state), respectively. Classical N-H

O and O-H

O hydrogen bonds, together with short C-H

O contacts, generate an extensive hydrogen-bonding network. The crystal structures of the maleate and fumarate salts of oxine have been elucidated to study the importance of noncovalent interactions in the aggregation and interaction patterns of biological molecules. The structures of the salts of the
Z and
E isomers of butenedioic acid (maleic and fumaric acid, respectively) with quinolin-8-ol are compared.
Supporting information
CCDC references: 724206; 724207
Equimolar quantities of maleic acid and hydroxyquinoline were dissolved in
water. The solution was stirred well and set aside to crystallize. Yellow
needle-like crystals of (I), suitable for X-ray diffraction analysis, were
obtained from the resulting solution after a week of slow evaporation.
Similarly, equimolar quantities of fumaric acid and hydroxyquinoline were
dissolved in water. The solution was stirred well and set aside to
crystallize. Yellow crystals of (II), suitable for X-ray diffraction analysis,
were obtained from the resulting solution after a week of slow evaporation.
For compound (I), which crystallized in the space group P212121,
the Friedel equivalents were merged prior to the final refinement cycles. The
O- and N-bound H atoms were located in difference Fourier maps and held fixed,
with O—H = 0.94 and 1.03 Å in (I), and 0.92 and 0.94 Å in (II), N—H =
1.00 Å in (I) and 0.89 Å in (II), with Uiso(H) =
1.5Ueq(O) and = 1.2Ueq(N). The C-bound H atoms in both
compounds were included in calculated positions and treated as riding atoms,
with C—H = 0.93 Å and Uiso(H) = 1.2Ueq(C).
For both compounds, data collection: APEX2 (Bruker, 2004); cell refinement: APEX2 and SAINT (Bruker, 2004); data reduction: SAINT and XPREP (Bruker, 2004); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008); molecular graphics: ORTEP-3 for Windows (Farrugia, 1997) and Mercury (Macrae et al., 2006); software used to prepare material for publication: PLATON (Spek, 2003).
(I) 8-hydroxyquinolinium hydrogen maleate
top
Crystal data top
| C9H8NO+·C4H3O4− | F(000) = 544 |
| Mr = 261.23 | Dx = 1.431 Mg m−3 |
| Orthorhombic, P212121 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2ac 2ab | Cell parameters from 2732 reflections |
| a = 5.3777 (3) Å | θ = 2.2–28.0° |
| b = 10.0563 (7) Å | µ = 0.11 mm−1 |
| c = 22.4243 (12) Å | T = 273 K |
| V = 1212.70 (13) Å3 | Needle, yellow |
| Z = 4 | 0.40 × 0.30 × 0.22 mm |
Data collection top
Bruker Kappa APEXII CCD
diffractometer | 1291 reflections with I > 2σ(I) |
| ω and ϕ scans | Rint = 0.034 |
Absorption correction: multi-scan
(SADABS; Bruker, 1999) | θmax = 28.3°, θmin = 2.2° |
| Tmin = 0.957, Tmax = 0.976 | h = −7→6 |
| 7633 measured reflections | k = −13→12 |
| 1764 independent reflections | l = −29→29 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.053 | H-atom parameters constrained |
| wR(F2) = 0.134 | w = 1/[σ2(Fo2) + (0.0458P)2 + 0.4725P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.16 | (Δ/σ)max = 0.001 |
| 1764 reflections | Δρmax = 0.22 e Å−3 |
| 172 parameters | Δρmin = −0.17 e Å−3 |
| 0 restraints | Absolute structure: Flack (1983), with 1220 Friedel pairs |
| Primary atom site location: structure-invariant direct methods | |
Crystal data top
| C9H8NO+·C4H3O4− | V = 1212.70 (13) Å3 |
| Mr = 261.23 | Z = 4 |
| Orthorhombic, P212121 | Mo Kα radiation |
| a = 5.3777 (3) Å | µ = 0.11 mm−1 |
| b = 10.0563 (7) Å | T = 273 K |
| c = 22.4243 (12) Å | 0.40 × 0.30 × 0.22 mm |
Data collection top
Bruker Kappa APEXII CCD
diffractometer | 1764 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker, 1999) | 1291 reflections with I > 2σ(I) |
| Tmin = 0.957, Tmax = 0.976 | Rint = 0.034 |
| 7633 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.053 | 0 restraints |
| wR(F2) = 0.134 | H-atom parameters constrained |
| S = 1.16 | Δρmax = 0.22 e Å−3 |
| 1764 reflections | Δρmin = −0.17 e Å−3 |
| 172 parameters | Absolute structure: Flack (1983), with 1220 Friedel pairs |
Special details top
Geometry. Bond distances, angles etc. have been calculated using the rounded
fractional coordinates. All su's are estimated from the variances of the
(full) variance-covariance matrix. The cell e.s.d.'s are taken into account in
the estimation of distances, angles and torsion angles |
Refinement. Refinement on F2 for ALL reflections except those flagged by the user
for potential systematic errors. Weighted R-factors wR and all
goodnesses of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The observed criterion of F2 > σ(F2)
is used only for calculating -R-factor-obs etc. and is not
relevant to the choice of reflections for refinement. R-factors based
on F2 are statistically about twice as large as those based on
F, and R-factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O8 | 0.1406 (5) | 0.7132 (3) | 0.04032 (10) | 0.0613 (9) | |
| N1 | 0.0137 (5) | 0.8064 (3) | 0.14740 (10) | 0.0408 (8) | |
| C2 | −0.0545 (7) | 0.8592 (4) | 0.19918 (13) | 0.0478 (10) | |
| C3 | 0.0614 (7) | 0.8221 (4) | 0.25131 (14) | 0.0532 (13) | |
| C4 | 0.2450 (8) | 0.7301 (4) | 0.24976 (14) | 0.0553 (11) | |
| C5 | 0.5065 (9) | 0.5766 (4) | 0.19072 (17) | 0.0677 (16) | |
| C6 | 0.5679 (10) | 0.5267 (5) | 0.1361 (2) | 0.0770 (17) | |
| C7 | 0.4471 (9) | 0.5711 (4) | 0.08471 (17) | 0.0660 (16) | |
| C8 | 0.2641 (7) | 0.6632 (4) | 0.08703 (14) | 0.0482 (11) | |
| C9 | 0.1984 (6) | 0.7149 (3) | 0.14337 (13) | 0.0391 (9) | |
| C10 | 0.3206 (7) | 0.6727 (4) | 0.19540 (14) | 0.0471 (11) | |
| O1 | 0.5130 (5) | −0.0021 (3) | 0.13620 (9) | 0.0591 (9) | |
| O2 | 0.7217 (5) | −0.0866 (3) | 0.06111 (10) | 0.0544 (8) | |
| O3 | −0.0739 (7) | 0.2729 (4) | 0.09560 (13) | 0.0988 (15) | |
| O4 | 0.1655 (6) | 0.1483 (3) | 0.15096 (11) | 0.0825 (10) | |
| C11 | 0.5548 (6) | −0.0126 (3) | 0.08121 (13) | 0.0417 (10) | |
| C12 | 0.4091 (7) | 0.0649 (4) | 0.03729 (14) | 0.0475 (10) | |
| C13 | 0.2236 (7) | 0.1488 (4) | 0.04502 (15) | 0.0519 (11) | |
| C14 | 0.0958 (8) | 0.1947 (4) | 0.09934 (17) | 0.0613 (16) | |
| H1 | −0.08960 | 0.83350 | 0.11250 | 0.0490* | |
| H2 | −0.18180 | 0.92180 | 0.20020 | 0.0570* | |
| H3 | 0.01380 | 0.86000 | 0.28740 | 0.0640* | |
| H4 | 0.32230 | 0.70450 | 0.28500 | 0.0660* | |
| H5 | 0.58830 | 0.54670 | 0.22470 | 0.0810* | |
| H6 | 0.69150 | 0.46240 | 0.13300 | 0.0920* | |
| H7 | 0.49370 | 0.53650 | 0.04790 | 0.0790* | |
| H8 | 0.17400 | 0.66630 | 0.00480 | 0.0920* | |
| H4A | 0.31920 | 0.08970 | 0.14430 | 0.1240* | |
| H12 | 0.45610 | 0.05220 | −0.00220 | 0.0570* | |
| H13 | 0.16270 | 0.18570 | 0.00990 | 0.0620* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O8 | 0.0722 (17) | 0.0810 (18) | 0.0308 (11) | 0.0188 (16) | −0.0033 (12) | −0.0103 (12) |
| N1 | 0.0403 (14) | 0.0505 (15) | 0.0315 (12) | −0.0005 (13) | −0.0018 (12) | −0.0010 (11) |
| C2 | 0.052 (2) | 0.0560 (19) | 0.0353 (15) | 0.0031 (18) | 0.0030 (17) | −0.0037 (14) |
| C3 | 0.063 (3) | 0.065 (2) | 0.0315 (15) | 0.003 (2) | 0.0020 (18) | −0.0034 (15) |
| C4 | 0.064 (2) | 0.068 (2) | 0.0340 (16) | 0.000 (2) | −0.0077 (18) | 0.0053 (16) |
| C5 | 0.071 (3) | 0.075 (3) | 0.057 (2) | 0.023 (2) | −0.010 (2) | 0.004 (2) |
| C6 | 0.072 (3) | 0.078 (3) | 0.081 (3) | 0.033 (3) | −0.002 (3) | −0.002 (2) |
| C7 | 0.067 (3) | 0.075 (3) | 0.056 (2) | 0.017 (2) | 0.007 (2) | −0.012 (2) |
| C8 | 0.049 (2) | 0.0542 (19) | 0.0415 (17) | 0.0018 (18) | 0.0020 (16) | −0.0055 (15) |
| C9 | 0.0385 (17) | 0.0428 (16) | 0.0359 (14) | −0.0044 (15) | 0.0009 (14) | −0.0017 (13) |
| C10 | 0.050 (2) | 0.0511 (19) | 0.0402 (16) | 0.0014 (17) | −0.0018 (17) | 0.0033 (15) |
| O1 | 0.0590 (16) | 0.0847 (18) | 0.0335 (12) | 0.0186 (16) | 0.0016 (12) | 0.0026 (11) |
| O2 | 0.0524 (15) | 0.0720 (17) | 0.0387 (11) | 0.0144 (14) | −0.0067 (12) | −0.0075 (11) |
| O3 | 0.111 (3) | 0.114 (3) | 0.0713 (19) | 0.065 (3) | 0.013 (2) | 0.0079 (19) |
| O4 | 0.099 (2) | 0.108 (2) | 0.0404 (13) | 0.048 (2) | 0.0127 (16) | 0.0053 (15) |
| C11 | 0.0359 (17) | 0.0513 (18) | 0.0380 (17) | −0.0017 (16) | −0.0018 (14) | −0.0027 (14) |
| C12 | 0.0443 (19) | 0.065 (2) | 0.0332 (15) | 0.0027 (18) | −0.0001 (16) | 0.0001 (15) |
| C13 | 0.055 (2) | 0.060 (2) | 0.0406 (17) | 0.0075 (19) | −0.0049 (18) | 0.0051 (16) |
| C14 | 0.061 (3) | 0.070 (3) | 0.053 (2) | 0.019 (2) | 0.005 (2) | 0.0026 (19) |
Geometric parameters (Å, º) top
| O8—C8 | 1.338 (4) | C6—C7 | 1.396 (6) |
| O8—H8 | 0.9400 | C7—C8 | 1.352 (6) |
| O1—C11 | 1.258 (4) | C8—C9 | 1.411 (4) |
| O2—C11 | 1.250 (4) | C9—C10 | 1.405 (5) |
| O3—C14 | 1.208 (6) | C2—H2 | 0.9300 |
| O4—C14 | 1.303 (5) | C3—H3 | 0.9300 |
| O4—H4A | 1.0300 | C4—H4 | 0.9300 |
| N1—C9 | 1.357 (4) | C5—H5 | 0.9300 |
| N1—C2 | 1.328 (4) | C6—H6 | 0.9300 |
| N1—H1 | 1.0000 | C7—H7 | 0.9300 |
| C2—C3 | 1.376 (5) | C11—C12 | 1.480 (5) |
| C3—C4 | 1.354 (6) | C12—C13 | 1.318 (5) |
| C4—C10 | 1.409 (5) | C13—C14 | 1.473 (5) |
| C5—C10 | 1.394 (6) | C12—H12 | 0.9300 |
| C5—C6 | 1.364 (6) | C13—H13 | 0.9300 |
| | | |
| O1···C9i | 3.315 (4) | C11···N1ii | 3.407 (4) |
| O1···C2ii | 3.058 (5) | C12···O3iii | 3.398 (5) |
| O2···C2ii | 3.366 (4) | C3···H5xii | 2.9900 |
| O2···N1ii | 2.714 (4) | C11···H2ii | 3.0900 |
| O2···C8iii | 3.418 (4) | C11···H8iii | 2.5500 |
| O2···O8ii | 3.057 (4) | C11···H1ii | 2.5600 |
| O2···O8iii | 2.643 (3) | C12···H8iii | 2.8900 |
| O3···C6iv | 3.324 (6) | C12···H13iii | 3.0400 |
| O3···C12v | 3.398 (5) | C13···H7v | 3.0600 |
| O4···C2i | 3.320 (5) | C14···H3vi | 3.0900 |
| O4···C3vi | 3.057 (5) | H1···O1vii | 2.7500 |
| O4···C4vi | 3.241 (5) | H1···O2vii | 1.7300 |
| O8···N1 | 2.666 (3) | H1···C11vii | 2.5600 |
| O8···O2vii | 3.057 (4) | H1···O8 | 2.3700 |
| O8···O2v | 2.643 (3) | H2···O1vii | 2.3100 |
| O1···H1ii | 2.7500 | H2···C11vii | 3.0900 |
| O1···H2ii | 2.3100 | H3···O4x | 2.7200 |
| O1···H4viii | 2.8700 | H3···C14x | 3.0900 |
| O2···H8iii | 1.7000 | H3···O3x | 2.7800 |
| O2···H1ii | 1.7300 | H3···H6xii | 2.6000 |
| O2···H7iii | 2.8900 | H4···O1xii | 2.8700 |
| O3···H13v | 2.7900 | H4···H5 | 2.5300 |
| O3···H6iv | 2.4300 | H5···H4 | 2.5300 |
| O3···H3vi | 2.7800 | H5···C3viii | 2.9900 |
| O3···H12v | 2.7400 | H6···H3viii | 2.6000 |
| O4···H3vi | 2.7200 | H6···O3xi | 2.4300 |
| O8···H1 | 2.3700 | H7···H8 | 2.3700 |
| N1···O2vii | 2.714 (4) | H7···C13iii | 3.0600 |
| N1···O8 | 2.666 (3) | H7···O2v | 2.8900 |
| N1···C11vii | 3.407 (4) | H8···H7 | 2.3700 |
| C2···O1vii | 3.058 (5) | H8···O2v | 1.7000 |
| C2···O2vii | 3.366 (4) | H8···C12v | 2.8900 |
| C2···O4ix | 3.320 (5) | H8···H12v | 2.4900 |
| C3···O4x | 3.057 (5) | H8···C11v | 2.5500 |
| C4···O4x | 3.241 (5) | H12···O3iii | 2.7400 |
| C6···O3xi | 3.324 (6) | H12···H8iii | 2.4900 |
| C8···O2v | 3.418 (4) | H13···O3iii | 2.7900 |
| C9···O1ix | 3.315 (4) | H13···C12v | 3.0400 |
| | | |
| C8—O8—H8 | 112.00 | C2—C3—H3 | 120.00 |
| C14—O4—H4A | 108.00 | C4—C3—H3 | 120.00 |
| C2—N1—C9 | 122.1 (3) | C3—C4—H4 | 120.00 |
| C9—N1—H1 | 123.00 | C10—C4—H4 | 120.00 |
| C2—N1—H1 | 115.00 | C6—C5—H5 | 120.00 |
| N1—C2—C3 | 120.6 (3) | C10—C5—H5 | 120.00 |
| C2—C3—C4 | 119.6 (3) | C7—C6—H6 | 120.00 |
| C3—C4—C10 | 120.9 (3) | C5—C6—H6 | 120.00 |
| C6—C5—C10 | 119.7 (4) | C6—C7—H7 | 119.00 |
| C5—C6—C7 | 120.7 (5) | C8—C7—H7 | 119.00 |
| C6—C7—C8 | 121.7 (4) | O1—C11—O2 | 122.1 (3) |
| O8—C8—C9 | 116.0 (3) | O2—C11—C12 | 117.0 (3) |
| O8—C8—C7 | 126.0 (3) | O1—C11—C12 | 120.9 (3) |
| C7—C8—C9 | 118.0 (3) | C11—C12—C13 | 130.6 (3) |
| C8—C9—C10 | 121.0 (3) | C12—C13—C14 | 131.5 (3) |
| N1—C9—C8 | 119.5 (3) | O4—C14—C13 | 119.2 (4) |
| N1—C9—C10 | 119.5 (3) | O3—C14—O4 | 120.8 (4) |
| C4—C10—C9 | 117.4 (3) | O3—C14—C13 | 120.0 (4) |
| C4—C10—C5 | 123.8 (3) | C11—C12—H12 | 115.00 |
| C5—C10—C9 | 118.8 (3) | C13—C12—H12 | 115.00 |
| C3—C2—H2 | 120.00 | C12—C13—H13 | 114.00 |
| N1—C2—H2 | 120.00 | C14—C13—H13 | 114.00 |
| | | |
| C9—N1—C2—C3 | −0.1 (5) | O8—C8—C9—N1 | 2.1 (5) |
| C2—N1—C9—C8 | −179.5 (3) | O8—C8—C9—C10 | −178.1 (3) |
| C2—N1—C9—C10 | 0.8 (5) | C7—C8—C9—N1 | −179.3 (3) |
| N1—C2—C3—C4 | −0.6 (6) | C7—C8—C9—C10 | 0.5 (5) |
| C2—C3—C4—C10 | 0.6 (6) | N1—C9—C10—C4 | −0.8 (5) |
| C3—C4—C10—C5 | −179.3 (4) | N1—C9—C10—C5 | 178.7 (3) |
| C3—C4—C10—C9 | 0.1 (6) | C8—C9—C10—C4 | 179.5 (3) |
| C10—C5—C6—C7 | 0.2 (7) | C8—C9—C10—C5 | −1.1 (5) |
| C6—C5—C10—C4 | −179.9 (4) | O1—C11—C12—C13 | −1.6 (6) |
| C6—C5—C10—C9 | 0.7 (6) | O2—C11—C12—C13 | 179.4 (4) |
| C5—C6—C7—C8 | −0.8 (7) | C11—C12—C13—C14 | −0.2 (8) |
| C6—C7—C8—O8 | 178.9 (4) | C12—C13—C14—O3 | −179.5 (5) |
| C6—C7—C8—C9 | 0.5 (6) | C12—C13—C14—O4 | −0.6 (7) |
| Symmetry codes: (i) x, y−1, z; (ii) x+1, y−1, z; (iii) x+1/2, −y+1/2, −z; (iv) x−1, y, z; (v) x−1/2, −y+1/2, −z; (vi) −x, y−1/2, −z+1/2; (vii) x−1, y+1, z; (viii) −x+1, y−1/2, −z+1/2; (ix) x, y+1, z; (x) −x, y+1/2, −z+1/2; (xi) x+1, y, z; (xii) −x+1, y+1/2, −z+1/2. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O2vii | 1.00 | 1.73 | 2.714 (4) | 167 |
| N1—H1···O8 | 1.00 | 2.37 | 2.666 (3) | 96 |
| O4—H4A···O1 | 1.03 | 1.40 | 2.427 (4) | 174 |
| O8—H8···O2v | 0.94 | 1.70 | 2.643 (3) | 177 |
| C2—H2···O1vii | 0.93 | 2.31 | 3.058 (5) | 137 |
| C6—H6···O3xi | 0.93 | 2.43 | 3.324 (6) | 160 |
| Symmetry codes: (v) x−1/2, −y+1/2, −z; (vii) x−1, y+1, z; (xi) x+1, y, z. |
(II) 8-hydroxyquinolinium hydrogen fumarate
top
Crystal data top
| C9H8NO+·C4H3O4− | Z = 2 |
| Mr = 261.23 | F(000) = 272 |
| Triclinic, P1 | Dx = 1.451 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 7.3282 (3) Å | Cell parameters from 6825 reflections |
| b = 7.4363 (3) Å | θ = 2.8–33.0° |
| c = 11.5680 (5) Å | µ = 0.11 mm−1 |
| α = 79.349 (2)° | T = 273 K |
| β = 74.994 (2)° | Block, yellow |
| γ = 89.337 (2)° | 0.30 × 0.20 × 0.16 mm |
| V = 597.97 (4) Å3 | |
Data collection top
Bruker Kappa APEXII CCD
diffractometer | 3049 reflections with I > 2σ(I) |
| ω and ϕ scans | Rint = 0.025 |
Absorption correction: multi-scan
(SADABS; Bruker, 1999) | θmax = 33.3°, θmin = 2.8° |
| Tmin = 0.967, Tmax = 0.982 | h = −10→11 |
| 15682 measured reflections | k = −11→11 |
| 4437 independent reflections | l = −17→17 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.047 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.152 | H-atom parameters constrained |
| S = 1.07 | w = 1/[σ2(Fo2) + (0.0668P)2 + 0.170P]
where P = (Fo2 + 2Fc2)/3 |
| 4437 reflections | (Δ/σ)max < 0.001 |
| 172 parameters | Δρmax = 0.45 e Å−3 |
| 0 restraints | Δρmin = −0.24 e Å−3 |
Crystal data top
| C9H8NO+·C4H3O4− | γ = 89.337 (2)° |
| Mr = 261.23 | V = 597.97 (4) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 7.3282 (3) Å | Mo Kα radiation |
| b = 7.4363 (3) Å | µ = 0.11 mm−1 |
| c = 11.5680 (5) Å | T = 273 K |
| α = 79.349 (2)° | 0.30 × 0.20 × 0.16 mm |
| β = 74.994 (2)° | |
Data collection top
Bruker Kappa APEXII CCD
diffractometer | 4437 independent reflections |
Absorption correction: multi-scan
(SADABS; Bruker, 1999) | 3049 reflections with I > 2σ(I) |
| Tmin = 0.967, Tmax = 0.982 | Rint = 0.025 |
| 15682 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.047 | 0 restraints |
| wR(F2) = 0.152 | H-atom parameters constrained |
| S = 1.07 | Δρmax = 0.45 e Å−3 |
| 4437 reflections | Δρmin = −0.24 e Å−3 |
| 172 parameters | |
Special details top
Geometry. Bond distances, angles etc. have been calculated using the rounded
fractional coordinates. All su's are estimated from the variances of the
(full) variance-covariance matrix. The cell e.s.d.'s are taken into account in
the estimation of distances, angles and torsion angles |
Refinement. Refinement on F2 for ALL reflections except those flagged by the user
for potential systematic errors. Weighted R-factors wR and all
goodnesses of fit S are based on F2, conventional
R-factors R are based on F, with F set to zero for
negative F2. The observed criterion of F2 > σ(F2)
is used only for calculating -R-factor-obs etc. and is not
relevant to the choice of reflections for refinement. R-factors based
on F2 are statistically about twice as large as those based on
F, and R-factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O8 | 0.35340 (14) | 0.84094 (17) | 0.53120 (9) | 0.0439 (3) | |
| N1 | 0.25392 (15) | 0.85725 (15) | 0.32353 (9) | 0.0314 (3) | |
| C2 | 0.2160 (2) | 0.86895 (19) | 0.21670 (12) | 0.0368 (4) | |
| C3 | 0.0373 (2) | 0.8185 (2) | 0.21063 (13) | 0.0423 (4) | |
| C4 | −0.0991 (2) | 0.7580 (2) | 0.31579 (14) | 0.0399 (4) | |
| C5 | −0.1962 (2) | 0.6898 (2) | 0.54225 (14) | 0.0412 (4) | |
| C6 | −0.1473 (2) | 0.6891 (2) | 0.64845 (13) | 0.0423 (4) | |
| C7 | 0.0359 (2) | 0.7400 (2) | 0.64921 (12) | 0.0371 (4) | |
| C8 | 0.17279 (18) | 0.79266 (17) | 0.54205 (11) | 0.0311 (3) | |
| C9 | 0.12277 (17) | 0.79750 (16) | 0.43155 (10) | 0.0280 (3) | |
| C10 | −0.06041 (18) | 0.74554 (17) | 0.43036 (12) | 0.0322 (3) | |
| O1 | 0.58773 (14) | 0.06213 (12) | 0.27497 (8) | 0.0348 (3) | |
| O2 | 0.66839 (18) | 0.07780 (12) | 0.07461 (9) | 0.0461 (3) | |
| O3 | 0.8069 (3) | 0.74935 (16) | −0.07584 (12) | 0.0793 (5) | |
| O4 | 0.65185 (16) | 0.73101 (12) | 0.11737 (8) | 0.0390 (3) | |
| C11 | 0.63628 (18) | 0.14898 (15) | 0.16694 (11) | 0.0290 (3) | |
| C12 | 0.65203 (18) | 0.35315 (15) | 0.14914 (11) | 0.0302 (3) | |
| C13 | 0.7118 (2) | 0.45877 (17) | 0.04200 (12) | 0.0390 (4) | |
| C14 | 0.7287 (2) | 0.66130 (17) | 0.02162 (12) | 0.0361 (4) | |
| H1 | 0.36510 | 0.90390 | 0.32450 | 0.0380* | |
| H2 | 0.30930 | 0.91120 | 0.14530 | 0.0440* | |
| H3 | 0.01090 | 0.82580 | 0.13550 | 0.0510* | |
| H4 | −0.21900 | 0.72460 | 0.31200 | 0.0480* | |
| H5 | −0.31800 | 0.65380 | 0.54380 | 0.0490* | |
| H6 | −0.23790 | 0.65390 | 0.72220 | 0.0510* | |
| H7 | 0.06490 | 0.73820 | 0.72310 | 0.0450* | |
| H8 | 0.36890 | 0.86930 | 0.60210 | 0.0660* | |
| H4A | 0.65960 | 0.85920 | 0.09920 | 0.0590* | |
| H12 | 0.61770 | 0.40760 | 0.21750 | 0.0360* | |
| H13 | 0.74600 | 0.40310 | −0.02580 | 0.0470* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O8 | 0.0366 (5) | 0.0657 (7) | 0.0315 (5) | −0.0089 (4) | −0.0105 (4) | −0.0115 (4) |
| N1 | 0.0337 (5) | 0.0318 (5) | 0.0290 (5) | −0.0024 (4) | −0.0094 (4) | −0.0044 (4) |
| C2 | 0.0434 (7) | 0.0379 (7) | 0.0288 (6) | 0.0004 (5) | −0.0107 (5) | −0.0035 (5) |
| C3 | 0.0482 (8) | 0.0484 (8) | 0.0356 (7) | 0.0015 (6) | −0.0209 (6) | −0.0070 (6) |
| C4 | 0.0377 (7) | 0.0431 (7) | 0.0442 (7) | 0.0015 (5) | −0.0196 (6) | −0.0093 (6) |
| C5 | 0.0302 (6) | 0.0486 (8) | 0.0436 (7) | −0.0007 (5) | −0.0070 (5) | −0.0093 (6) |
| C6 | 0.0359 (7) | 0.0512 (8) | 0.0340 (6) | −0.0013 (6) | −0.0003 (5) | −0.0060 (6) |
| C7 | 0.0398 (7) | 0.0440 (7) | 0.0275 (6) | 0.0014 (5) | −0.0079 (5) | −0.0078 (5) |
| C8 | 0.0337 (6) | 0.0322 (6) | 0.0291 (5) | 0.0011 (4) | −0.0098 (4) | −0.0078 (4) |
| C9 | 0.0314 (5) | 0.0255 (5) | 0.0279 (5) | 0.0017 (4) | −0.0085 (4) | −0.0060 (4) |
| C10 | 0.0319 (6) | 0.0312 (6) | 0.0355 (6) | 0.0027 (4) | −0.0117 (5) | −0.0077 (5) |
| O1 | 0.0468 (5) | 0.0293 (4) | 0.0273 (4) | −0.0067 (4) | −0.0099 (4) | −0.0020 (3) |
| O2 | 0.0856 (8) | 0.0225 (4) | 0.0293 (5) | −0.0018 (5) | −0.0117 (5) | −0.0068 (3) |
| O3 | 0.1402 (14) | 0.0302 (5) | 0.0404 (6) | −0.0044 (7) | 0.0205 (7) | −0.0007 (5) |
| O4 | 0.0610 (6) | 0.0217 (4) | 0.0323 (5) | 0.0022 (4) | −0.0079 (4) | −0.0060 (3) |
| C11 | 0.0392 (6) | 0.0210 (5) | 0.0272 (5) | −0.0015 (4) | −0.0096 (4) | −0.0039 (4) |
| C12 | 0.0409 (6) | 0.0213 (5) | 0.0297 (5) | 0.0002 (4) | −0.0094 (5) | −0.0075 (4) |
| C13 | 0.0639 (9) | 0.0221 (5) | 0.0298 (6) | 0.0016 (5) | −0.0084 (6) | −0.0071 (4) |
| C14 | 0.0535 (8) | 0.0222 (5) | 0.0300 (6) | 0.0001 (5) | −0.0068 (5) | −0.0040 (4) |
Geometric parameters (Å, º) top
| O8—C8 | 1.3434 (17) | C6—C7 | 1.402 (2) |
| O8—H8 | 0.9200 | C7—C8 | 1.3719 (19) |
| O1—C11 | 1.2578 (15) | C8—C9 | 1.4124 (17) |
| O2—C11 | 1.2459 (15) | C9—C10 | 1.4054 (19) |
| O3—C14 | 1.1956 (19) | C2—H2 | 0.9300 |
| O4—C14 | 1.3020 (16) | C3—H3 | 0.9300 |
| O4—H4A | 0.9400 | C4—H4 | 0.9300 |
| N1—C9 | 1.3658 (15) | C5—H5 | 0.9300 |
| N1—C2 | 1.3227 (17) | C6—H6 | 0.9300 |
| N1—H1 | 0.8900 | C7—H7 | 0.9300 |
| C2—C3 | 1.389 (2) | C11—C12 | 1.4957 (16) |
| C3—C4 | 1.364 (2) | C12—C13 | 1.3084 (18) |
| C4—C10 | 1.412 (2) | C13—C14 | 1.4823 (18) |
| C5—C6 | 1.365 (2) | C12—H12 | 0.9300 |
| C5—C10 | 1.408 (2) | C13—H13 | 0.9300 |
| | | |
| O1···C8i | 3.3969 (16) | C6···O3xii | 3.234 (2) |
| O1···O8ii | 3.1729 (14) | C7···O3xii | 3.2019 (19) |
| O1···N1ii | 2.7711 (15) | C8···C10xiii | 3.5744 (18) |
| O1···C2ii | 3.3672 (18) | C8···O1i | 3.3969 (16) |
| O1···C4iii | 3.2636 (18) | C10···C8xiii | 3.5744 (18) |
| O1···O8i | 2.6151 (14) | C11···C2ii | 3.597 (2) |
| O2···C2iv | 3.2042 (17) | C11···N1ii | 3.4494 (17) |
| O2···C3iv | 3.3980 (18) | C11···O4ii | 3.2587 (15) |
| O2···C14ii | 3.2701 (16) | C13···C13iv | 3.501 (2) |
| O2···O4ii | 2.5327 (13) | C14···O2viii | 3.2701 (16) |
| O3···C7v | 3.2019 (19) | C3···H13iv | 3.0500 |
| O3···C3vi | 3.372 (2) | C11···H1ii | 2.7500 |
| O3···C6v | 3.234 (2) | C11···H7i | 3.0000 |
| O3···C2vi | 3.0267 (19) | C11···H2ii | 3.0800 |
| O4···C2 | 3.3217 (19) | C11···H4Aii | 2.4200 |
| O4···C4vii | 3.3148 (19) | C11···H8i | 2.6400 |
| O4···O2viii | 2.5327 (13) | C12···H8i | 3.0100 |
| O4···C3vii | 3.3832 (19) | C12···H6xiv | 3.0100 |
| O4···C11viii | 3.2587 (15) | C12···H7i | 2.8500 |
| O8···O8ix | 3.0687 (17) | C14···H3vii | 3.0900 |
| O8···O1i | 2.6151 (14) | H1···O1viii | 1.9200 |
| O8···O1viii | 3.1729 (14) | H1···C11viii | 2.7500 |
| O8···N1 | 2.6673 (14) | H1···O8 | 2.3300 |
| O1···H4iii | 2.8900 | H2···O2viii | 2.7800 |
| O1···H4Aii | 2.6900 | H2···C11viii | 3.0800 |
| O1···H1ii | 1.9200 | H2···H4A | 2.5200 |
| O1···H8i | 1.7000 | H2···O2iv | 2.4900 |
| O2···H3iv | 2.9000 | H2···O3vi | 2.7000 |
| O2···H2iv | 2.4900 | H2···O4 | 2.8000 |
| O2···H2ii | 2.7800 | H3···O4x | 2.8000 |
| O2···H13 | 2.4900 | H3···C14x | 3.0900 |
| O2···H4Aii | 1.6000 | H3···O2iv | 2.9000 |
| O3···H7v | 2.6000 | H4···O4x | 2.6500 |
| O3···H2vi | 2.7000 | H4···O1xi | 2.8900 |
| O3···H6v | 2.6600 | H4···H5 | 2.5400 |
| O4···H12 | 2.4500 | H4A···H2 | 2.5200 |
| O4···H2 | 2.8000 | H4A···C11viii | 2.4200 |
| O4···H3vii | 2.8000 | H4A···O2viii | 1.6000 |
| O4···H4vii | 2.6500 | H4A···O1viii | 2.6900 |
| O8···H5vii | 2.7900 | H5···O8x | 2.7900 |
| O8···H8ix | 2.9000 | H5···H4 | 2.5400 |
| O8···H1 | 2.3300 | H6···C12xiv | 3.0100 |
| N1···C11viii | 3.4494 (17) | H6···O3xii | 2.6600 |
| N1···O1viii | 2.7711 (15) | H7···H8 | 2.4100 |
| N1···O8 | 2.6673 (14) | H7···O3xii | 2.6000 |
| C2···O4 | 3.3217 (19) | H7···C11i | 3.0000 |
| C2···O3vi | 3.0267 (19) | H7···C12i | 2.8500 |
| C2···C11viii | 3.597 (2) | H8···H7 | 2.4100 |
| C2···O2iv | 3.2042 (17) | H8···O1i | 1.7000 |
| C2···O1viii | 3.3672 (18) | H8···O8ix | 2.9000 |
| C3···O3vi | 3.372 (2) | H8···C11i | 2.6400 |
| C3···O4x | 3.3832 (19) | H8···C12i | 3.0100 |
| C3···O2iv | 3.3980 (18) | H12···O4 | 2.4500 |
| C4···O1xi | 3.2636 (18) | H13···O2 | 2.4900 |
| C4···O4x | 3.3148 (19) | H13···C3iv | 3.0500 |
| | | |
| C8—O8—H8 | 112.00 | C4—C3—H3 | 120.00 |
| C14—O4—H4A | 111.00 | C2—C3—H3 | 120.00 |
| C2—N1—C9 | 122.71 (12) | C3—C4—H4 | 120.00 |
| C9—N1—H1 | 119.00 | C10—C4—H4 | 120.00 |
| C2—N1—H1 | 118.00 | C10—C5—H5 | 120.00 |
| N1—C2—C3 | 120.29 (13) | C6—C5—H5 | 120.00 |
| C2—C3—C4 | 119.43 (13) | C5—C6—H6 | 119.00 |
| C3—C4—C10 | 120.78 (14) | C7—C6—H6 | 119.00 |
| C6—C5—C10 | 119.36 (14) | C8—C7—H7 | 120.00 |
| C5—C6—C7 | 121.69 (13) | C6—C7—H7 | 120.00 |
| C6—C7—C8 | 120.63 (13) | O1—C11—O2 | 124.89 (11) |
| O8—C8—C7 | 126.08 (12) | O2—C11—C12 | 118.08 (11) |
| C7—C8—C9 | 118.16 (12) | O1—C11—C12 | 117.01 (10) |
| O8—C8—C9 | 115.76 (11) | C11—C12—C13 | 123.11 (11) |
| C8—C9—C10 | 121.38 (11) | C12—C13—C14 | 124.28 (12) |
| N1—C9—C10 | 119.22 (11) | O3—C14—C13 | 121.29 (13) |
| N1—C9—C8 | 119.39 (12) | O4—C14—C13 | 114.37 (11) |
| C4—C10—C5 | 123.64 (13) | O3—C14—O4 | 124.34 (13) |
| C4—C10—C9 | 117.58 (12) | C11—C12—H12 | 118.00 |
| C5—C10—C9 | 118.74 (12) | C13—C12—H12 | 118.00 |
| N1—C2—H2 | 120.00 | C12—C13—H13 | 118.00 |
| C3—C2—H2 | 120.00 | C14—C13—H13 | 118.00 |
| | | |
| C9—N1—C2—C3 | −0.4 (2) | O8—C8—C9—N1 | −2.98 (18) |
| C2—N1—C9—C8 | −178.60 (13) | O8—C8—C9—C10 | 178.31 (12) |
| C2—N1—C9—C10 | 0.14 (19) | C7—C8—C9—N1 | 177.09 (12) |
| N1—C2—C3—C4 | 0.5 (2) | C7—C8—C9—C10 | −1.63 (19) |
| C2—C3—C4—C10 | −0.2 (2) | N1—C9—C10—C4 | 0.08 (19) |
| C3—C4—C10—C5 | 177.92 (14) | N1—C9—C10—C5 | −177.97 (12) |
| C3—C4—C10—C9 | 0.0 (2) | C8—C9—C10—C4 | 178.80 (12) |
| C10—C5—C6—C7 | −1.0 (2) | C8—C9—C10—C5 | 0.75 (19) |
| C6—C5—C10—C4 | −177.38 (14) | O1—C11—C12—C13 | 175.86 (14) |
| C6—C5—C10—C9 | 0.6 (2) | O2—C11—C12—C13 | −5.5 (2) |
| C5—C6—C7—C8 | 0.0 (2) | C11—C12—C13—C14 | 179.94 (14) |
| C6—C7—C8—O8 | −178.69 (14) | C12—C13—C14—O3 | 170.10 (18) |
| C6—C7—C8—C9 | 1.2 (2) | C12—C13—C14—O4 | −10.1 (2) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) x, y−1, z; (iii) x+1, y−1, z; (iv) −x+1, −y+1, −z; (v) x+1, y, z−1; (vi) −x+1, −y+2, −z; (vii) x+1, y, z; (viii) x, y+1, z; (ix) −x+1, −y+2, −z+1; (x) x−1, y, z; (xi) x−1, y+1, z; (xii) x−1, y, z+1; (xiii) −x, −y+2, −z+1; (xiv) −x, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O1viii | 0.89 | 1.92 | 2.7711 (15) | 159 |
| N1—H1···O8 | 0.89 | 2.33 | 2.6673 (14) | 102 |
| O4—H4A···O2viii | 0.94 | 1.60 | 2.5327 (13) | 177 |
| O8—H8···O1i | 0.92 | 1.70 | 2.6151 (14) | 175 |
| C2—H2···O2iv | 0.93 | 2.49 | 3.2042 (17) | 133 |
| C12—H12···O4 | 0.93 | 2.45 | 2.7657 (15) | 100 |
| C13—H13···O2 | 0.93 | 2.49 | 2.7988 (16) | 100 |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (iv) −x+1, −y+1, −z; (viii) x, y+1, z. |
Experimental details
| | (I) | (II) |
| Crystal data |
| Chemical formula | C9H8NO+·C4H3O4− | C9H8NO+·C4H3O4− |
| Mr | 261.23 | 261.23 |
| Crystal system, space group | Orthorhombic, P212121 | Triclinic, P1 |
| Temperature (K) | 273 | 273 |
| a, b, c (Å) | 5.3777 (3), 10.0563 (7), 22.4243 (12) | 7.3282 (3), 7.4363 (3), 11.5680 (5) |
| α, β, γ (°) | 90, 90, 90 | 79.349 (2), 74.994 (2), 89.337 (2) |
| V (Å3) | 1212.70 (13) | 597.97 (4) |
| Z | 4 | 2 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 0.11 | 0.11 |
| Crystal size (mm) | 0.40 × 0.30 × 0.22 | 0.30 × 0.20 × 0.16 |
| |
| Data collection |
| Diffractometer | Bruker Kappa APEXII CCD
diffractometer | Bruker Kappa APEXII CCD
diffractometer |
| Absorption correction | Multi-scan
(SADABS; Bruker, 1999) | Multi-scan
(SADABS; Bruker, 1999) |
| Tmin, Tmax | 0.957, 0.976 | 0.967, 0.982 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 7633, 1764, 1291 | 15682, 4437, 3049 |
| Rint | 0.034 | 0.025 |
| (sin θ/λ)max (Å−1) | 0.668 | 0.772 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.053, 0.134, 1.16 | 0.047, 0.152, 1.07 |
| No. of reflections | 1764 | 4437 |
| No. of parameters | 172 | 172 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.22, −0.17 | 0.45, −0.24 |
| Absolute structure | Flack (1983), with 1220 Friedel pairs | ? |
Hydrogen-bond geometry (Å, º) for (I) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O2i | 1.0000 | 1.7300 | 2.714 (4) | 167.00 |
| N1—H1···O8 | 1.0000 | 2.3700 | 2.666 (3) | 96.08 |
| O4—H4A···O1 | 1.0300 | 1.4000 | 2.427 (4) | 174.00 |
| O8—H8···O2ii | 0.9400 | 1.7000 | 2.643 (3) | 177.00 |
| C2—H2···O1i | 0.9300 | 2.3100 | 3.058 (5) | 137.00 |
| C6—H6···O3iii | 0.9300 | 2.4300 | 3.324 (6) | 160.00 |
| Symmetry codes: (i) x−1, y+1, z; (ii) x−1/2, −y+1/2, −z; (iii) x+1, y, z. |
Hydrogen-bond geometry (Å, º) for (II) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O1i | 0.8900 | 1.9200 | 2.7711 (15) | 159.00 |
| N1—H1···O8 | 0.8900 | 2.3300 | 2.6673 (14) | 102.00 |
| O4—H4A···O2i | 0.9400 | 1.6000 | 2.5327 (13) | 177.00 |
| O8—H8···O1ii | 0.9200 | 1.7000 | 2.6151 (14) | 175.00 |
| C2—H2···O2iii | 0.9300 | 2.4900 | 3.2042 (17) | 133.00 |
| C12—H12···O4 | 0.9300 | 2.4500 | 2.7657 (15) | 100.00 |
| C13—H13···O2 | 0.9300 | 2.4900 | 2.7988 (16) | 100.00 |
| Symmetry codes: (i) x, y+1, z; (ii) −x+1, −y+1, −z+1; (iii) −x+1, −y+1, −z. |
 O- hydrogen bonds. The maleate salt crystallizes in the space group P212121, while the fumarate salt crystallizes in P
O- hydrogen bonds. The maleate salt crystallizes in the space group P212121, while the fumarate salt crystallizes in P O and O-H
O and O-H O hydrogen bonds, together with short C-H
O hydrogen bonds, together with short C-H O contacts, generate an extensive hydrogen-bonding network. The crystal structures of the maleate and fumarate salts of oxine have been elucidated to study the importance of noncovalent interactions in the aggregation and interaction patterns of biological molecules. The structures of the salts of the Z and E isomers of butenedioic acid (maleic and fumaric acid, respectively) with quinolin-8-ol are compared.
O contacts, generate an extensive hydrogen-bonding network. The crystal structures of the maleate and fumarate salts of oxine have been elucidated to study the importance of noncovalent interactions in the aggregation and interaction patterns of biological molecules. The structures of the salts of the Z and E isomers of butenedioic acid (maleic and fumaric acid, respectively) with quinolin-8-ol are compared.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2009/02/00/su3028/su3028fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2009/02/00/su3028/su3028fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2009/02/00/su3028/su3028fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/c/issues/2009/02/00/su3028/su3028fig4thm.gif)




Oxine (8-hydroxyquinoline, 8-HQ), a monoprotic bidentate chelating agent, and its derivatives, are widely used as analytical reagents and potential anti-amoebic agents (Bambury, 1979). The complexes and the heterocycle itself exhibit antiseptic, disinfectant and pesticide properties (Phillips, 1956). Its solution in alcohol [Ethanol?] is used as a liquid bandage. 8-HQ was also once of interest as an anticancer drug (Shen et al., 1999). In this paper, we present the maleic and fumaric acid complexes of 8-HQ, compounds (I) and (II), respectively.
Maleic acid, the Z isomer of butenedioic acid, has been used as a simple building block in supramolecular architectures in two and three dimensions (Bowes et al., 2003; Jin et al., 2003). The maleic acid anion can exist in the fully deprotonated form, or as hydrogen maleate with one of the carboxylic acid groups protonated (Lah & Leban, 2003). Several singly dissociated maleate salts are reported in the Cambridge Structural Database (Version?; Allen, 2002). Fumaric acid, the E isomer of butenedioic acid, is of interest since it is known to form supramolecular assemblies with N-aromatic compounds (Batchelor et al., 2000). It tends to form infinite chains arranged in a nearly coplanar manner via pairs of strong O—H···O hydrogen bonds. This organic dicarboxylic acid crystallizes in two polymorphic forms; one is monoclinic, space group P21/c (Brown, 1966) and the other triclinic, space group P1 (Bednowitz & Post, 1966). In both crystal structures the acid molecules are linked by carboxylic acid R22(8) (Bernstein et al., 1995) hydrogen-bond pairs, forming one-dimensional supramolecular tapes. An extensive network of hydrogen bonds is observed in the majority of the crystal structures of salts of fumaric acid (Shan et al., 2003; Alagar, Krishnakumar et al., 2003; Videnova-Adrabińska, 1996; Smith et al., 1997; Li et al., 2007; Büyükgüngör et al., 2004). The title compounds, (I) and (II), are the salts of oxine with the cis and trans pair of dicarboxylic acids, maleic and fumaric acids, the Z and E isomers of butenedioic acid, respectively. They have been studied to explore their hydrogen-bonding patterns.
The structures of compounds (I) and (II) are shown in Figs. 1 and 2, respectively. The asymmetric unit of (I) consists of a 1:1 complex of a semimaleate anion and the hydroxyquinolinium cation. Compound (II) consists of a 1:1 complex of a semifumarate anion and the hydroxyquinolinium cation. The 8-HQ molecules in both compounds are protonated at atom N1. Proton transfer observed between the 8-HQ N atom and the carboxylic acid groups of both (I) and (II) is confirmed by the increase in the internal angles at N1, which are 122.1 (3) and 122.71 (12)°, respectively, compared with 119° observed in neutral 8-HQ (Roychowdhury et al., 1978).
The C8—O8 bond lengths in the 8-HQ cation in (I) and (II) are 1.338 (4) and 1.3434 (17) Å, respectively. These distances are shorter than the bond lengths observed in the neutral 8-HQ molecule (1.39 Å). Furthermore, the exocyclic angles, C7—C8—O8 and C9—C8—O8, in the cation of (I) are 126.0 (3) and 116.0 (3)°, respectively, and those in the cation of (II) are 126.08 (12) and 115.76 (11)°, respectively. These angles are significantly different from those observed in neutral 8-HQ (118.2 and 120.3°). This difference in the bond lengths and angles may be attributed to the presence of weak intramolecular N—H···O hydrogen bonds. Similar hydrogen bonds have been observed to result in an enhancement of the internal angle at the N centre in the crystal structures of the 8-HQ complexes quoted above. The O1—C11 and O4—C14 bonds of the semimaleate anion are longer than O2—C11 and O3—C14, showing differences between the C—O and C═O bond types of the carboxylic acid group (Borthwick, 1980). Similarly, the O1—C11 and O4—C14 bonds of the semifumarate anion are longer than the O2—C11 and O3—C14 bonds.
The 8-HQ cations in both (I) and (II) are essentially planar. The dihedral angles between the fused rings are 0.77 and 2.23°, respectively, and the total puckering amplitudes (Cremer & Pople, 1975) Q are 0.026 (4) and 0.059 Å, respectively. The semimaleate and semifumarate anions are almost planar. The angles between the planes of the half-semimaleate anion in (I) and those of the semifumarate anion in (II) (O1/O2/C11/C12 and O3/O4/C13/C14) are 1.58 and 14.83°, respectively. The semimaleate anion is planar with a cis conformation about the central C═C bond [C11—C12—C13—C14 = -0.2 (8)°], while the semifumarate anion is planar with a trans conformation about the central C═C bond [C11—C12—C13—C14 = 179.94 (14)°]. Both the cations and anions of (I) and (II) are individually planar, but with dihedral angles of 10.92 and 73.17°, respectively.
The crystal packings of compounds (I) and (II) are stabilized by an extensive network of hydrogen bonds, which are summarized in Tables 1 and 2. Parts of the crystal structures of (I) and (II), depicting the hydrogen-bonding interactions and the formation of hydrogen-bonded motifs, are shown in Figs. 3 and 4, respectively. In both compounds, a strong intermolecular N+—H···O- hydrogen bond exists between the deprotonated carboxylate group of the anion and the hydroxyquinolinium N atom of the cation. The presence of an intramolecular N—H···O hydrogen bond, represented by a graph-set motif of S(5), in the 8-HQ cations of (I) and (II) has been observed in most of the 8-hydroxyquinolinium and related cation compounds (Balasubramanian & Muthiah, 1996; Balasubramanian & Thomas Muthiah, 1996; Jebamony & Thomas Muthiah, 1998; Banerjee et al., 1984; Smith et al., 2004). This hydrogen bond was observed to result in an enhancement of the internal angle at the N centre. An intramolecular hydrogen bond between atoms O1 and O4 in the semimaleate ion of (I) is found to be asymmetric, as in the crystal structures of maleic acid itself (James & Williams, 1974) and several maleate salts of various amino acids (Alagar, Krishnakumar et al., 2001, Alagar et al., 2002; Alagar, Subha Nandhini et al., 2003; Rajagopal et al., 2001, 2002). However, in the crystal structures of the complexes of maleic acid with DL- and L-arginine (Ravishankar et al., 1998), L-hystidine and L-lysine (Pratap et al., 2000), and L-phenylalaninium (Alagar, Krishnakumar & Natarajan, 2001), this intramolecular hydrogen bond is symmetric and generates an S(7) motif. C—H···O intramolecular interactions generate an S(5) motif in the semifumarate anion in molecule (II).
Intermolecular hydrogen-bonding interactions between the semimaleate anions in (I) are not observed. The hydroxyquinolinium cation is connected to the anion by N1—H1···O2 and C2—H2···O1 interactions to form a ring motif described by a graph-set motif of R22(7). In (II), however, intermolecular hydrogen-bonding interactions between the semifumarate anions, as well with the cations, are observed. The anions are linked by chains propagating perpendicularly to the ac plane via O4—H4A···O2 hydrogen bonds and can be described by a graph-set motif of C(7). Hydroxyquinolinium cations form infinite stacks propogating perpendicularly to the ac plane, the distance between adjacent molecules within a stack being 7.44 Å. A pair of cations and a pair of anions are linked through O—H···O and N—H···O hydrogen bonds to form a ring motif, represented by a graph-set motif of R22(8). The cations and anions in (I) aggregate through hydrogen bonds to form an extensive three-dimensional network, while in (II), they aggregate through hydrogen bonds to form a two-dimensional network.